Q: 2a. Draw the mechanism and major product of the synthesis reaction below. b. Give the reagent(s)…
A: Given reaction is, E1 reaction I.e. unimolecular elimination reaction.
Q: NH2
A: The given reagent is Di-tert-butyl dicarbonate which is commonly known as Boc anhydride. It is a…
Q: Provide the mechanism for the conversion shown below. CH3Li is effectively H3C-Li+. What type of…
A: Given : reaction Step 1: put partial charge on reactant
Q: OH H2SO4 HOCH,CH3 Doubling [CH3CH,OH] doubles the rate of rxn.
A: Given : Doubling concentration of CH3CH2OH doubles the rate of reaction.
Q: 2. Show the B-cleavage mechanism and product for following reaction -R R'
A: The given problem deals with β - cleavage reactions which are generally initiated in the presence of…
Q: Both of the following reactions will give the same SN2 product. Draw the mechanism for each of these…
A: SN2 reaction is nuclephilic substitution reaction of second order where a nucleophile attack at a…
Q: NH2 Br ETONO + COOH Br Heat
A: The given reaction is an example of [4+2]-cycloaddition reaction. Firstly, the two reactants will be…
Q: CN .CN NH3 CH3OH Br Br `NH2
A:
Q: Make a table with the main conditions for each mechanism: E1, E2, SN1, SN2.
A: The main conditions for E1, E2, SN1, SN2 mechanism have to be given.
Q: Explain the mechanism of the following reactions and give the name of product? CH,CH(OH)C(CH,), –…
A:
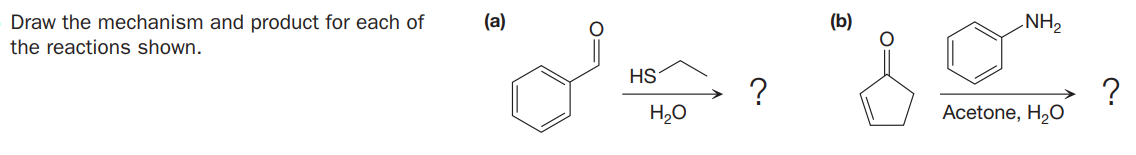
Q: Draw the mechanism and predict the major product for each of the following reactions. (a) (b) CH3…
A: Concept introduction: Reduction: It is the process in which hydrogen is added to the reactant by…
Q: Both compounds A and B below give the same carbocation intermediate C in the SN1 reaction. However,…
A: In this question, we will Identify which Compound ( A or B ) give carbocation intermediate ( C )…
Q: Draw a mechanism for this reaction: CH3 H,C-OH Br CH3 ČH3
A: The above Reaction is the Nucleophilic substitution reaction of the allylic Halide . In this above…
Q: CH3 CH3 CHO OSIME;
A:
Q: Draw a step-wise mechanism for each of the following reactions. Show all arrows, proton transfers,…
A:
Q: 6.) Provide a reasonable mechanism, including all curvy arrows and intermediates, for the following…
A: Fluroine when attached with aromatic ring becomes a better leaving group. Fluorine is having…
Q: 3. Draw the mechanism R [M]=/
A:
Q: Q5. Draw the all products and a detailed mechanism for the following reaction? -OCH2CH3 Br CH;CH2OH
A: This reaction will proceed through SN1 -mechanism . The slvent is polar protic and reactant is…
Q: Provide the product and mechanism for the reaction: ОН CrO3, H,SO, H,0
A: This reaction is an example of Jone oxidation. Mechanism of Reaction:
Q: OH H2SO4 (cat.) H20
A:
Q: Provide a curved-arrow mechanism for the following reaction. Be sure to use your mechanism and a few…
A: So, in this reaction, the alkene acts as a nucleophile, attacking the electrophilic bromine, giving…
Q: Draw curved arrows to illustrate the mechanism for the reaction of 3‑methylbutan‑1‑ol and HBrHBr.…
A:
Q: H2NNH2, KOH Hао, heat
A:
Q: A B C heat Na NH3 1) 03 2) DMSO reductive workup OH O H
A: Since, you have multiple subparts questions. So, I will solved first three subparts questions for…
Q: 2. Draw the step-by-step reaction mechanism and predict product(s) of the following reaction?
A:
Q: (a) PHOOC-CH,-C-CH,-COOP. (1) KCN, A (2) H,0® (o) СООН Onl
A:
Q: Show a detailed mechanism of the following reaction OH OH ATP H,0
A:
Q: H2N. Br N° H2N
A:
Q: Show the detailed mechanism for the following reactions. a. он b. H H OCH3 CH;OH H
A:
Q: Draw the mechanism for the following reaction:
A: Please go through the reaction mechanism.
Q: 3. Explain, by means of a detail mechanism for each, why the two reactions give the same product.…
A: ->In presence of base elimination reaction occur and alkene formed.it removed antiperiplanar…
Q: (b) Show the mechanism of this reaction using proper arrow push notation. You mu intermediates,…
A: We will write the mechanism of the given conversion
Q: Will each of the following reactions follow an El or E2 mechanism? CI HO Он
A:
Q: 5. Provide the product that will result for the following reaction and provide the complete…
A: Diazomethane (CH2N2) is a source of carbene and shows carbene addition across the double bond to…
Q: Could the reactions shown here take place by a concerted mechanism?
A:
Q: Draw the mechanism and the major organic product(s) for each of the following reactions. (a) (b) 1.…
A:
Q: 3. Provide the complete mechanism for each of the following reaction. Н-Br
A: Provide the complete mechanism for each of the following reaction.
Q: Show the detailed mechanism of the following reaction. Include the electron-push arrow. Fischer…
A: Let us first prepare the reagent for the reaction and then provide a mechanism of fisher reaction.
Q: 1. Give a mechanism of following conversion CH,OH
A:
Q: .CI .CI aq. HBr 65 °C HO. Br
A: The reaction taking place is given as,
Q: CH3 CH, H3C-¢* + :Br: H,C Br : CH3 CH,
A: The movement of electrons are shown by curved arrows. There are two types of arrows to show the…
Q: Draw the structure of all products of the mechanism below. H-C=C -OH + H-Ö-CH, H. it
A: Due to presence of lone pair electrons in the O atom, alcohols like methanol can act as nucleophiles…
Q: Draw the mechanism and predict the major product for each of the following reactions. (a) (b) O (c)…
A: The Reagents NaBH4 and LiAlH4 is a very good reducing agents which converts aldehyde or ketones into…
Q: Choose the mechanism that forms the main product of the following reaction. Er ONa CH3CHOH calor
A:
Q: Draw the mechanism of the reaction of 1 mole of methanol with 1 mole of cyclohexanone, in the…
A: Ketones reacts with alcohols in presence of hydronium ion to produce hemiacetals.
Q: 2. Answer ALL parts. a) Write a detailed reaction mechanism for the following conversion. ОН HO…
A: We have to give the mechanism of the given reaction.
Q: Please show a plausible mechanism. This question is related to Pericyclic reactions CH3 CH3 550 °C…
A: First step of the reaction involves reversible electrocyclic sing opening to a ketene aldehyde.…
Q: Draw the mechanism and predict the major product for each of the following reactions. (a) (b) CH3…
A: The reduction reactions in organic chemistry indicate that the conversion of an unsaturated…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Between E1 and E2, which reaction mechanism is most efficient to synthesize the (E) stereoisomer of this product on an industrial scale?Provide the mechanism for the conversion shown below. CH3Li is effectively H3C-Li+. What type of mechanism is occuring?What is the mechanism and product for the following reaction?
- Can you provide a mechanism for this reaction to show the major diastereoisomer that would be formed?Between E1 and E2, which reaction mechanism is more efficient to synthesize the (E) stereoisomer of this product on a large scale?Azo coupling reaction. Provide a mechanism with intermediates and arrows
- Please draw the mechanism for the following reaction. The first step in the reaction mechanism is the formation of a carbocation intermediate (shown), followed by a carbocation rearrangement. Which bond moves in the carbocation rearrangement? Which compound shown below has that appropriate bond in pink?( complete the reaction with mechanism , do 2st one )is this an E1 or E2 mechanism for this reaction? What is the major product and mechanism for it?

