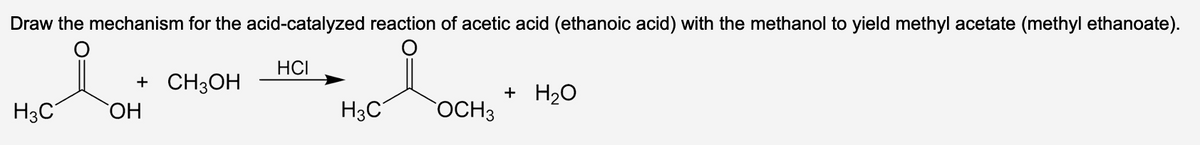
Draw the mechanism for the acid-catalyzed reaction of acetic acid (ethanoic acid) with the methanol to yield methyl acetate (methyl ethanoate). HCI + CH3OH H3C OH H3C OCH3 + H₂O
Q: 35) Identify the transition metal that is used in oxygen transport in the human body. A) zinc B)…
A: Metals are present in our body in trace amount these metals are very useful to functioning properly…
Q: An iodine clock experiment was performed and the following data was recorded. Determine the initial…
A:
Q: ▪ In a blood alcohol analysis by GC-FID, methanol elutes at 1 min, ethanol at 2 min, IPA at 3 min,…
A: Answer : Relative retention time = retention time of analyte / retention of known peak based on…
Q: A student conducts a titration using NaOH (strong base) as the titrant to determine the…
A:
Q: Draw the structures of the TWO Reactants that will be needed to synthesized the desired product from…
A:
Q: MISSED THIS? Read Section 18.8. You can click on the Review link o access the section in your e…
A: Given reactions are : Part. A :- N2O4 (g) --------> 2NO2 (g) Part. B :- NH4I (s)…
Q: Draw the Lewis structure of the missing product. Make sure to include lone pairs and non-zero formal…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: 41. Potassium permanganate, KMnO4, a common oxidizing agent, is made from various ores that contain…
A: The balanced chemical equation for the formation of KMnO4 is: 2MnO2 + 2KOH + O2 → 2KMnO4 + H2 O2 is…
Q: small.unt of sodium chloride (NaCl) is dissolved in a large amount of water. Imagine separating this…
A:
Q: What is/are the reagent(s) and conditions for the following reaction? Problem viewing the image.…
A:
Q: A student mixes hydrobromic acid and aluminium solid and observes the formation of a gas. a. Write…
A:
Q: C. Trehalose and isomaltose are both dimers of glucose. However, they have considerably different…
A: #C: Trehalose is formed by the 1→1 glycosidic bond between two α-glucose units. Isomaltose is…
Q: لله - od This B-diketone can be synthesized from cyclohexene and cyclopentanone using an enamine…
A:
Q: equilibrium), Flavylium cation +H2O + heat ↔ Hydrated flavylium + OH- (colored)…
A: Here we have to determine the true statement among the following for the given equilibrium reaction.…
Q: 5) Given the following substances in order of increasing acid strength, (AOCT (99) (HC2H302 (99)k…
A:
Q: Calculate the pH of a 0.08 M NaOH solution. Compare to the pH found in problem 3. pll = 12.90 4.
A: Given, Concentration of NaOH solution = 0.08 M pH of the solution = ? Note: NaOH is a strong base…
Q: Cowen l'e if approximately 5 kJ I mod Stronger than a and a G-D bond is C-H bond C-H bond thas 의 an…
A: We know that wave number = 1/2πc * √(K/b) Where, K = force constant while b = reduced mass Or, 1/b…
Q: What is the correct IUPAC name of the following compound? Br e Problem viewing the image, Click…
A: Here we are required to write the correct IUPAC name for the given compound
Q: One of the steps to sweeten sour gas using the Claus process is reacting hydrogen sulfide gas with…
A:
Q: environmental advantages and disadvantages
A: Polymer – A polymer is a large atom made up of smaller molecules linked together. These small…
Q: For the Lewis dot structure shown below, predict: Y=X=Y (b) what is the bond angle?
A:
Q: OH isopropyl 2-hydroxy-4-methoxybenzoate OH NaOH o hydroxy(isopropoxy)(4-methoxyphenylmethanolate OH…
A: Esters on basic hydrolysis to give carboxylic acid, which undergo decarboxylation. In the Birch…
Q: Consider the following reversible reaction equation: MgCO₂(aq) + H+(aq) → Mg²+ (aq) + HCO¸¯(aq) K =…
A:
Q: In titrating a H2SO4 solution, it was found that 23.66 mL of 0.2137 M NaOH would neutralize 22.04 mL…
A:
Q: Consider the effects of electronegativity and atomic sizes on the deviation from ideality of…
A:
Q: What is the major organic product of the following reaction? (Assuming excess reagents) A. B. C.…
A: Organic reaction are those in which organic reactant react to form organic products.
Q: Imagine there is a bulla, a large blister or sack of gas, in the lungs of a person who is climbing…
A: Introduction : According to Boyle's law the absolute pressure exerted by given mass of ideal gas is…
Q: Balance the equation above. If you perform this reaction with 30.0 mL of 4.50 M HCl and 20.5 grams…
A:
Q: 29) Which of the following compounds exhibits geometric isomerism? A) CH₂=CH₂ B) CH₂=CCl2 C) 1₂C=CHI…
A: Since, In geometrical isomerism, rotation is restricted. It means that, two different types of group…
Q: A sample of O2 gas ws collected over water at a total pressure of 620 mm Hg and 25 degrees celsius.…
A:
Q: Using the data given, fill in the following table of ion concentrations and pH values. Remember that…
A: pH greater than 7 means solution is basic and pH less than 7 means solution is acidic.
Q: 2). Which of the following reactions will not produce 2,3-dimethylpentan-3-ol? A) C) + + 2. H3O+…
A: The Grignard reagent reacts with a ketone to form tertiary alcohol. The reaction is proceeded by the…
Q: Given K+ values: NiY2 = 4.2 x 10¹8, FeY2 = 2.1 x 10¹4, CoY2 = 2.0 x 10¹6. In the separate…
A: Complexometric titration is used for the determination of a mixture of different metal ions in…
Q: 4. Draw the structures for the oxidation of the following carbohydrates. C-H H-L OH +애 CH₂OH…
A:
Q: Rank the following substances in order in increasing intermolecular forces: a. CH3F b. CH3NH2 c.…
A:
Q: In coal, sulfur typically exists in the form of iron pyrites, FeS₂, or in organic compounds. Burning…
A:
Q: Draw the structures of the TWO Reactants and the Reagents/Conditions that will be needed to…
A:
Q: The standard way of quantifying or measuring matter is by its a. mass b. weight c. volume d. length…
A: A substance that has mass and occupies volume is known as a matter. There are three states of matter…
Q: Consider the following reactions and choose the correct structures from the pool of choices below.…
A:
Q: Consider the molecule und build it using the simulation provided. Complete each section provided…
A:
Q: What are the products of the following reaction? O OH OH concentrated HI and and and and HO K HO
A: -> In presence of HI ether can break and form alcohol and alkyl iodide .
Q: -24. The following data are for a liquid chromatographic column: Length of Packing Flow rate VM Vs A…
A: Given, Chromatic mixture of species A,B,C and D. Length of packing , L =24.7 cm
Q: For each chemical reaction in the table below, decide whether the highlighted reactant is a…
A: The answer to the following question is-
Q: Christian tried to sabotage Hazelle’s experiment by pouring 10 ml of 1M ammonia into Hazelle’s 10 ml…
A: Christian tried to sabotage Hazelle’s experiment by pouring 10 ml of 1M ammonia into Hazelle’s 10 ml…
Q: relationship between concentration and time
A: Given statement is : What is the relationship between concentration and time ? Concentration is…
Q: 26) Name the following compound CH₂CH₂CH3 .CH3CH₂CCH₂CH3 CH3 A) 3-ethyl-3-methylhexane B)…
A:
Q: Synthesis #1 CO₂CH3 from mod H CH3 juvabione skeleton Br H and any other reagents and sources of…
A: Note - Since the given question is a multiple question, hence I solved first question according to…
Q: OH 3-pentanol OH diphenylmethanol pentanol benzophenone OH Test: Positive Sign: Test: Positive Sign:
A: Disclaimer “Since you have asked multiple question, we will solve the first question for you. If…
Q: How many hydrogen and carbon are present in the molecule according to the HNMR and CNMR
A: Number of carbon atoms and hydrogen atoms are found using 13CNMR spectra and 1HNMR spectra. The…
Q: What is the numerical value of the autoionization constant for water, Kw?
A: numerical value of the autoionization constant for water, Kw?
5
Step by step
Solved in 2 steps with 1 images

- Describe the reaction mechanism for the hydrolysis of 2-bromo-2-methylpropane [CH3C(CH3)2Br] with aqueous hydroxide ions (OH-).When S-2-bromobutane reacts with hydroxide ions (OH-) it forms R-2-butanol. The accepted reaction mechanism of this reaction is shown below. This is an SN2 mechanism.1,4-Dioxane is made commercially by the acid-catalyzed condensation of an alcohol. Propose a mechanism for this reaction.
- complete total synthesis of Delta9 THC (all the steps of the mechanism).) Propose a mechanism for the acid-catalyzed hydrolysis of cyclohexanone ethylene acetal.Imagine that the aldehyde 2 methylbutanal reacts with ethane-1,2-diol under acidic conditions. Draw a balanced reaction formula for the reaction and Draw the reaction mechanism for the reaction.
- What type of mechanisms will the following reactants undergo? (vii )Propanal : …………………………………………………………. (viii) Propene ……………………………………………………….. (ix)Methylbenene ………………………………………………….Early chemists could envision three possible mechanisms for hydroxide-ion-promoted ester hydrolysis. Devise an experiment that would show which of the three is the actual mechanism. 1. a nucleophilic acyl substitution reaction 2. an SN2 reaction 3. an SN1 reactionDraw curved arrows to illustrate the mechanism for the reaction of 3‑ethyl‑3‑pentanol and HCl.
- Draw reaction mechanism for week 2 A wittig reaction experiment: Week 1 -》 preparation of phosphonium salt Materials : triphenylphosphine (5.3g),methyl Bromoacetate 3.36g (2.1ml) , ethanol 30ml . The yield obtained was 8.93g .The percentage yield was 106.4% Week 2 -》 Formation of the ylide and wittig reaction Materials :Phosphonium salt (5g), napthyl-2- carboxaldehyde( 2.65g), 20 ml of water and 5 nl of Nahco3. Yield obtained was 0.51g . The percentage yield was 14.2%. Week 3: Solvent free wittig reaction Materials: Benzyltriphenylphosphonium chloride (0.5g ), 4 - Bromoabenzaldehyde (0.24g), Potassium phosphate (tribasi c) 0.275g .The yield obtained was 1.21g. The percentage yield was 35.6%.show the mechanism for the alkylation of benzene by 2-butene + HF.Propose a mechanism for the acid-catalyzed hydrolysis of cyclohexanone dimethyl acetal.

