Q: What alkenes are formed from attached alkyl halide by an E2 reaction? Use the Zaitsev rule to…
A:
Q: 9. Which SN1 reaction in each pair is faster? a) b) (CH3)3CCI + H2O → H,O (CH3)3CB +H0→ .CI H,O
A: SN1 is a unimolecular Nucleophilic substitution reaction. The reaction involves a carbocation…
Q: Which is the better leaving group in each pair?a. Cl-, I-b. NH3, -NH2c. H2O, H2S
A: Generally good leaving group is weak base. This weak base will have stronger conjugate acids with…
Q: Which is the most stable: 3,4-dimethyl-2-hexene, 2,3-dimethyl-2-hexene, or 4,5-dimethyl-2-hexene? a.…
A: The stability of an alkene increases as the number of alkyl substituents bonded to its sp2 carbon…
Q: Draw the products of each reaction, and state whether the reaction is faster or slower than a…
A:
Q: Draw the product formed by treating each compound with excess CH3I, followed by Ag2O, and then heat.
A: The complete reaction for a is shown below.
Q: C. CH;OH Is this reaction E1 or E2? elimination product(s) d. NaOH Is this reaction SN1 or SN2?…
A:
Q: Draw the organic product of each reaction. a. Br "OH + "OCH(CH3)2 С. b. + "OCH3 d. Br + "OCH,CH3
A: The reactions given are,
Q: Draw the products formed when the following diene is treated with Og followed by CH,SCH3.
A: Ozone is a very strong oxidizing agent. It is used for production of ketone or aldehyde from alkene.…
Q: Which is the most stable carbocation? C B. A OA OB
A: The species in which the carbon atom bonded with three sigma bonds and has a positive charge is…
Q: Draw the product formed by treating each compound with excess CH3I, followed by Ag2O, and then heat.
A: Hoffmann elimination It is a type of elimination reaction of an alkene where least stable alkene…
Q: Which SN2 reaction is each pair is faster? А. Br H20 Br HO, OH Br Br OCH3 Oc .CI HO, B.
A: In a SN 2 reaction, the nucleophile attacks the alkyl halide from backside, and the bond breaking…
Q: Draw the products of each reaction. KMNO4 Br2 FeBra a. CH3 -C(CH3)3 d. Br2 NH,NH2 b. hv "OH NO2 CH3…
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be…
Q: Draw the product formed from cyclohex-2-enone in each of three reactions. Reaction 1 Reaction 2…
A: Introduction: In the first reaction, cyanide ion act as a nucleophile attacks on the carbonyl…
Q: OCHS H,C-S но 1 4 3.
A: The leaving group ability of a species depends on the stability of the anion. As the stability of…
Q: Draw the products formed when each diene is treated with one equivalent of HCI. а. CH;CH=CH-CH=CHCH3…
A: Since you are posted with multiple sub-parts. As per the rule, I answering the first three questions…
Q: Br Pool of choices: OCH₂ ABC CH3OH DEF major substitution product GHI OCH 3 major elimination…
A:
Q: Which halide is most reactive in SN2 reactions? Select one: CH2CH2CI ÇI CI H3C CI CI CH3
A: The reactivity order of SN2 : 1° > 2° > 3° alkyl halides.
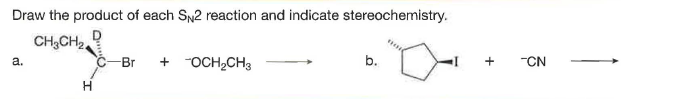
Q: Draw the product of each SN2 reaction and indicate stereochemistry.
A: Please find below the SN2 reaction product formed. Since the attack is from the back side. Hence the…
Q: Draw an energy diagram for the following SN2 reaction. Label the axes, the starting materials, and…
A: The reaction is assumed to be exothermic and so, the energy of the reactants is more than the energy…
Q: Which carbocation in each pair is more stable?
A: a) Since in compound 1 the carbocation can do resonance with double bond as it is in conjugation…
Q: The product in the first reaction is a ? H,SO, of R =R H20
A: The product of a first reaction has to be identified below.
Q: which of the following SN2 reactions will be faster? (1) Reactions of CN- (cynaide ion) with…
A: We have to choose for following given SN2 reactions will be faster. (1) Reactions of CN- (cynaide…
Q: Draw the product of an SN2 reaction shown below. Include all lone pairs. Use wedge and dash bonds to…
A: SN2 (Substitution Nucleophilic bimolecular) is a bimolecular reaction. It means reaction take place…
Q: CN ČN CO2CH3 CO2CH3 ? (HạC),sid + ?
A: To find out diels alder reactants we can do retro Diels-Alder reaction. Let's see,
Q: What alkenes are formed from each alkyl halide by an E2 reaction? Use the Zaitsev rule to predict…
A: Given compounds,
Q: Which is the best leaving group for an SN2 reaction? O F- O C- O Br-
A: To identify: The best leaving group for an SN2 reaction.
Q: What type of intermediate is present in the SN2 reaction of cyanide (CN) with bromoethane?…
A: What type of intermediate present on reaction as given via SN2 reaction---
Q: Draw the products of each SN1 reaction and indicate the stereochemistry of any stereogenic centers.
A: Figure 1
Q: Which halide is most reactive in SN2 reactions? Select one: -CH2CH2CI ÇI CI H3C ÇI CI CH3
A: Nucleophilic substitution biomolecular reaction (SN2):The bimolecular nucleophilic substitution…
Q: Draw an energy diagram for the following SN2 reaction. Label the axes, the starting materials, and…
A: The mechanism of the reaction with the intermediate that is formed would be given as,
Q: Draw an energy diagram for the following SN2 reaction. Label the axes, the starting materials, and…
A: The reaction mechanism along with the intermediate formed can be shown as follows,
Q: Draw the product of an SN2 reaction shown below. Use wedge and dash bonds to indicate…
A:
Q: Draw the products of attached Diels–Alder reaction, and indicate the stereochemistry.
A: To draw the products of the below Diels–Alder reaction and to indicate the stereochemistry.
Q: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary.
A:
Q: Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2
A: Introduction: NaNH2 is a strong base. It is often used for deprotonation in weak acids or also for…
Q: Draw the products of attached reaction, including stereochemistry.
A: Stereochemistry is defined as the branch of chemistry which deals with the arrangement of atoms in…
Q: Which halide is most reactive in SNl reactions? Select one: O H;C-CH2-CH2CI CH3 H3C-CH-CH2CI CH3…
A:
Q: ? Br What is the best reagent for the transformation shown above? B E „Mgl A D PPH3
A: We know that R-Li gives exclusively gives 1,2 product.
Q: What are the products of this NBS bromination reaction? CD2=CH-CH3 + N-Br hv (A) CHD=CD-CH,-Br and…
A: The answer is given as follows
Q: Draw the products, including stereochemistry, of attached reaction.
A: In electrophilic addition reaction, unsaturated alkenes are converted to saturated hydrocarbons with…
Q: Draw the products of the attached reaction, including stereochemistry.
A: During Bromination of alkenes, the π-elcetrons of the alkene attack the Br2 from downward position…
Q: Draw the products of each SN1 reaction and indicate the stereochemistry of any stereogenic centers.
A: To draw the products of each SN1 reaction and to indicate the stereochemistry of any stereogenic…
Q: Consider two Sn2 reactions: (1) a reaction between NaOH and 2-iodopropane and, (2) a reaction…
A:
Q: Which of the following SN2 reactions occur faster and why? a CH3 H2 -C-Br CH;CH,CHBr + OH or H,C-…
A:
Q: Which halide is most reactive in SN2 reactions? Select one: CH3 H3C-C-CH2CI CH3 CH3 H3C-CH-CI O…
A: Which halide is most reactive in SN2 reation?
Q: Draw the products of each SN1 reaction and indicate the stereochemistry when necessary.
A: 1) Since in SN1 reaction, formation of carbocation takes place. And since the carbocation can be…
Q: Why is - CN a weaker leaving group than - NHCH3.
A: Weak Bases are the Best Leaving Groups. (Recall that the stronger the acid, the weaker the conjugate…
Q: 2) Which of the following would react fastest in an SN2 type of reaction? d) -CI b) c) a) CI
A: SN2 means bi molecular nucleophilic substitution reaction Reactivity order CH3Cl > 1° > 2°…
Q: 25.38 Draw the product of each intramolecular cycloaddition. а. hv [2 + 2] b. H. [4 + 2] OSIR3 С. A…
A:
Step by step
Solved in 3 steps with 2 images

- Draw the product of attached SN2 reaction and indicate stereochemistry.Draw the products of each SN1 reaction and indicate the stereochemistry when necessary.Draw an energy diagram for the following SN2 reaction. Label the axes, the starting materials, and the product. Draw the structure of the transition state.
- a.What product(s) (excluding stereoisomers) are formed when Y is heated with Cl2? b.What product(s) (excluding stereoisomers) are formed when Y is heated with Br2? c.What steps are needed to convert Y to the alkene Z?What alkene is the major product formed from each alkyl halide in an E1 reaction?Draw the products formed when the following diene is treated with O3 followed by CH3SCH3.

