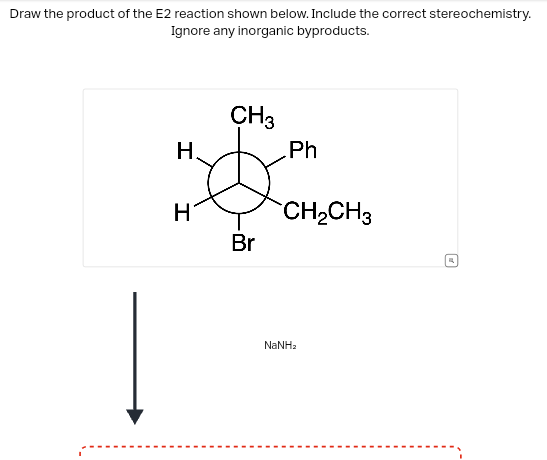
Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any inorganic byproducts. H CH3 H Br Ph CH2CH3 NaNha M
Q: None
A: Approach to solving the question:1. Calculate the moles of NaH2PO4 and Na2HPO4.2. Determine the…
Q: Identify the most acidic solution. options: 0.001 M solution of HNO3 10 M solution of CH3COOH 0.001…
A: Solution: To identify the most acidic solution, we can compare the strength of the acids. The…
Q: 6. What is an advantage of using pyridinium tribromide instead of bromine? (1 point) 7. Draw the…
A: The advantage of using pyridinium tribromide over bromine is that it is a solid at room temperature,…
Q: Arrange the following from lowest to highest entropy: H2O (l) , H2O2 (l), H2O (g) , CuO (s). Assume…
A: The objective of the question is to arrange the given substances in the order of increasing entropy.…
Q: Fill in the missing information for each of the 4 reactions
A: The answer image is attached here
Q: Draw a stepwise mechanism for the following reaction, a key step in the synthesis of ticlopidine, a…
A: The objective of the question is to find the stepwise mechanism of the following reaction of…
Q: Payalben
A: The objective of the question is to estimate the concentration of E. coli bacteria at the end of the…
Q: Please answer both! Confused on how to slove them!
A: Step 1: you can also prove this by using trigonometric identity: sin2θ+cos2θ=1Square and add above…
Q: The Kp for the reaction A(g) ⇌ 2B(g) = 0.0450. So, what is Kp for the reaction 2 A (g) ⇌ 4 B (g)
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: A gas-filled weather balloon with a volume of 60.0 L is held at ground level, where the atmospheric…
A: Step 1:Step 2:
Q: Please don't use hend raiting and step by step solutions
A: Step 1: SN2 ReactionRate of the SN2 reaction:Rate = k[RX][nucleophile] SN2 reaction follow…
Q: None
A: Step 1: Retrosynthetic analysis is a method used in organic chemistry to plan the synthesis of…
Q: How many moles of potassium fluoride should be dissolved in 1.98 L of solvent to produce a 10.2 M…
A: Given: V=1.98L;[KF]=10.2MStep 1: What is the number significant figures for the final answer.The…
Q: Select to Draw CI Select to Draw NaCCCH₂ THE ☑
A:
Q: T(C) 1200 Cu - Li Data from FScopp alloy databases (2019) 1000 800 600 400 200 0 FCC-A1…
A: The objective of the given question is to determine the solubility limit of and the melting…
Q: A 10.0 L sample of carbon dioxide gas has a temperature of 500.0 K. If the volume of the gas is…
A: The objective of this question is to find the final temperature of the carbon dioxide gas when the…
Q: Please re do over the work and check it it correct
A: This reaction is hydroboration-oxidation of an alkyne. The reagent does the same thing with BH3…
Q: Identify the molecular geometry around the carbon atom highlighted red in the given organic…
A: The molecule you're describing, geometry around the carbon atom highlighted red in the given organic…
Q: None
A: Correct name of molecule is 2-methylpent-1-en-3-yne step-by-step process and explanation of each…
Q: Provide the mechanism of the transformation below. 1. H3O+ is
A: Step 1: Given the reaction, Now the above reaction mechanism is,
Q: Give the major product of the following reaction. excess OCH₂CH 1. CH, CHO CH3CH₂ OCH2CH3 2. HCI…
A: Step 1: The mechanism followed by the reaction is explained below Step 2: Step 3: Step 4:Hope it…
Q: Predict the major product of the following reaction:
A: When morpholine combines with an alkyl bromide, it usually undergoes a substitution…
Q: 17. Calculate F = F(T,V,N) and H = H(S, p, N) of the ideal gas, using the function of the internal…
A: Understanding he Information:The image you sent contains an equation and a table related to the…
Q: The great expert Hand written solution is not allowed.
A: Step 1:Step 2:
Q: I need answer expert solutions I need answer step by step
A: Please Rate if the solution helps you.Thank You
Q: Calculate the atom densities for U-235, U-238, F, O, and H for a UO2F2 water solution. The U is…
A: U-235: 235g/molU-238: 238.03g/molF (Fluorine): 18.998g/molO (Oxygen): 15.999g/molH (Hydrogen):…
Q: Calculate the pH of a 0.061 M solution of sodium ascorbate, Na2C6H6O6. For ascorbic acid, H2C6H606,…
A: The objective of this question is to calculate the pH of a 0.061 M solution of sodium ascorbate,…
Q: (8) The cell: SCE//HA(0.250M), NaA(0.170M)/H2(1.00bar), Pt has a poten- tial of -0.721V at 298K.…
A: The objective of the question is to Sketch the cellCalculate the Potential of the CellCalculate the…
Q: None
A:
Q: A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.554 M…
A:
Q: Problem 1. Fill in the missing products in this synthetic sequence. 1. s-BuLi, THF, -30 C 2. ZnCl2,…
A: The objective of the question is to find the missing products of the reaction.“Since you have posted…
Q: None
A: This is the required image of solution.
Q: 8. You are titrating 50.0 mL of 0.200 M propanoic acid with 0.100 M NaOH. Use the data in the table…
A:
Q: Which aqueous solution has the LOWEST freezing point? 3.0 M LiS 4.0 M KBr O2.0 M MgC₁₂ 2.0 M AlCl3
A: The objective of this question is to determine which among the given aqueous solutions has the…
Q: many grams of solid sodium fluoride should be added to 0.500 L of 0.124 M hydrofluoric acid solution…
A: The objective of this question is to determine the amount of solid sodium fluoride (NaF) that should…
Q: 6. Draw the most likely major cyclic product, assuming the reaction goes to completion. 1. H3O+ HO H…
A: The major product of this reaction is a cyclic acetal. At first in presence of H+ cyclic hemiacetal…
Q: Determine the molar concentration of a solution of NaF that has a pH of 8.51 at 25 °C. The K₂ of HF…
A: The objective of this question is to find the molar concentration of a solution of NaF given the pH…
Q: 1. What kind of titration is this? Strong acid, strong base strong base, weak acid strong acid,…
A:
Q: 18. What is the relationship between the following pairs? (Choices: enantiomers, diastereomers,…
A: Step 1: Types of isomersStep 2:the atoms are connected differently. And this is the simplest class…
Q: please explain answer...
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: How many grams of Kr are in a 3.84 L cylinder at 55.0 °C and 1.76 atm? Enter numeric value mass: g
A: The objective of the question is to find out the mass of Kr (Krypton) in a cylinder with given…
Q: None
A: Step 1: Step 2: Once you know the working of reagent you have to follow the arrow pushing i…
Q: 0.9583 g of sample was dissolved in 80 mL of xylene and extracted with three 15 mL quantities of…
A: The objective of the question is to calculate the free chloramphenicol content of the sample in…
Q: Question 2 (2 points) Which of the following reactions is most likely to be an SN2 reaction? mm CI…
A: Step 1: Step 2: Step 3: Step 4:
Q: A scientist wants to look into the kinetics of the degradation of atrazine (A). There are 2 ways for…
A: Using a series of differential rate equations based on the reaction kinetics, it can be utilised to…
Q: Predict the major product for each of the following reactions. PPh3 ? Draw Your Solution eTextbook…
A:
Q: I need answer expert solutions Need answer step by step
A: The standard Gibbs free energy change for a reaction at a specified temperature is the energy…
Q: Sally obtains a standard calibration curve for their assigned food dye by plotting absorbance versus…
A: The objective of this question is to determine the concentration of the food dye in the original…
Q: None
A:
Q: None
A: Step 1: When 1-chloropropane reacts with Li and Cu, it forms Gilman reagent as shown below. This is…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the major and minor products of the E2 elimination shown below. Ignore any inorganic byproducts.Draw the major organic product of this E1 elimination reaction. Ignore byproducts.Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any inorganic byproducts.
- Which of A to E correctly explains why iodocyclohexane rather than chlorocyclohexane is a better substrate for the E1 reaction shown below?Draw the Zaitsev and Hofmann products that are expected when each of the following compounds is treated with a strong base to give an E2 reaction.Identify the major and minor products for these E1 reactions. Please explain also how it is done, how to get these products, is there any technique?

