Q: b) OTf N-Pt-OTf + PMe3 OTf + 3. Predict the products of the following reactions: a) CI 72- CI-Pt-CI…
A: The objective of this question is to predict the products of reactions involving coordination…
Q: 2. To warm 400.0 grams of water, 0.050 mol of ethanol (C2H5OH) is burned. The water warms from…
A: To find the heat absorbed by water, use the formula:q=mcΔTWhere:q is the heat absorbed by water (in…
Q: Label the chiral center ( R or S) , then draw the structures enantiomer.
A: As shown, the configuration of the chiral centers in the given compound was R and R. An enantiomer…
Q: 122. * Predict the products of the following reactions. In (a) and (d), give the intermediate…
A: In (a) and (d), the reaction is allylic and benzylic bromination, respectively. Their free radicals…
Q: Longifolene is a terpene which has the structure A. a) Identify which class of terpenes it belongs…
A: Terpenes belong to the biggest class of secondary metabolites and consist of five carbon isoprene…
Q: Draw the product of the following reaction. CH3 CH3CHCH2CH2CO₂CH3 1. DIBAH 2. H3O+ • Draw only the…
A: Step 1: Step 2: Step 3: Step 4:
Q: Which of the following is best described as an acid–base reaction? Cu(s) + H2S(g) → CuS(s) + H2(g)…
A: The reaction that best fits the description of an acid-base reaction is:…
Q: raw the major product of this reaction. Ignore inorganic byproducts. 1. excess Br2, NaOH 2.…
A: The objective of the question is to predict the product formed in the given reaction.The given…
Q: Which of these ionic solids would have the largest lattice energy?A) NACIB) NaFC) CaBrD) CsIE) CaO
A: Lattice energy is the energy released when gaseous ions combine to form an ionic solid. The greater…
Q: 2. Complete the road map by providing the structures of the products or the reagents as needed in…
A: Approach to solving the question: The given question aims to complete the given road map by…
Q: (a) The 1H-NMR spectrum of compound A, C7H15 Cl, consists of the following signals: 8 1.1 (s, 9H)…
A: Step 1: To determine, we have to determine the structure of the molecule based on the given dataThis…
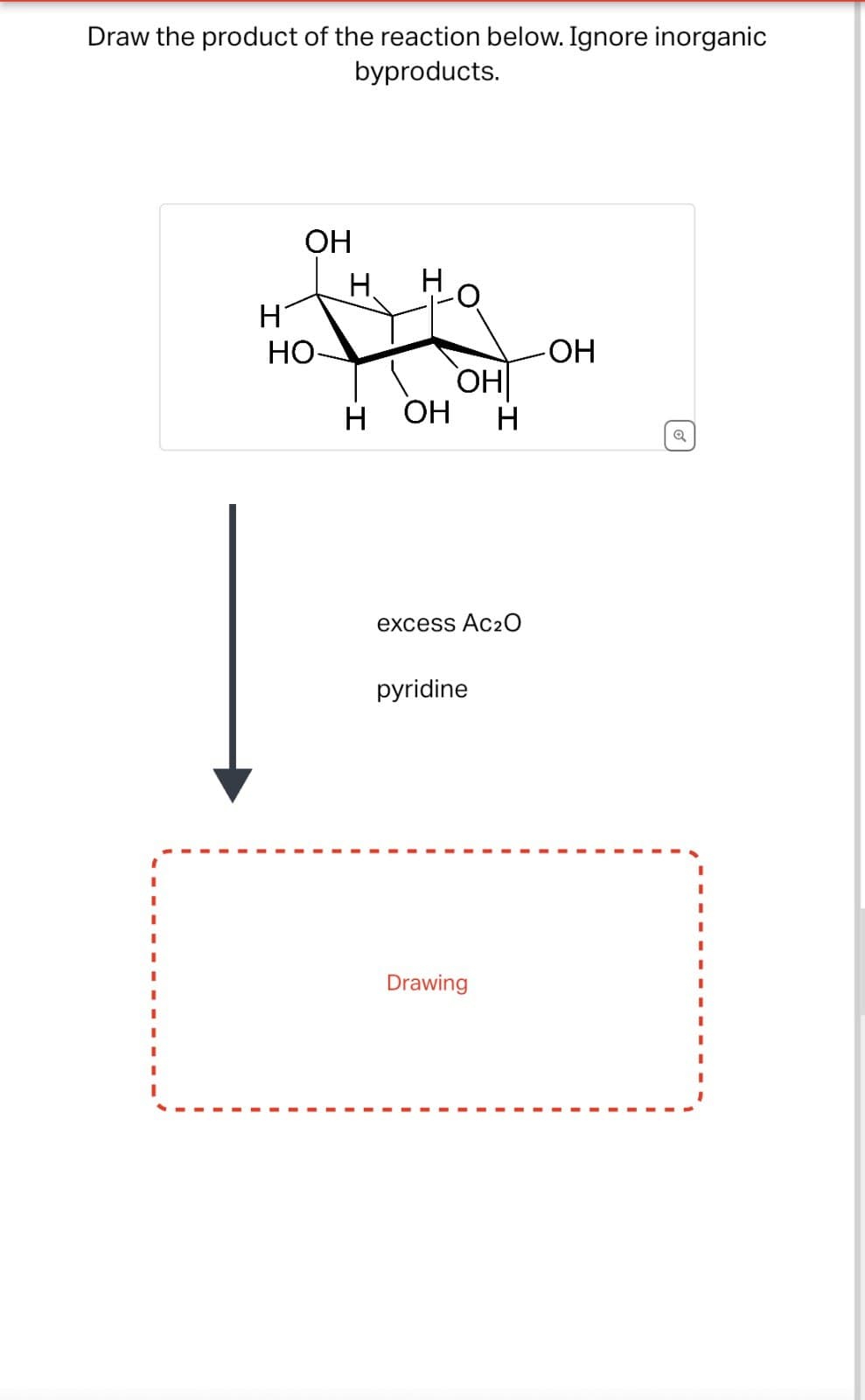
Q: Provide the major product for the following reaction? H HO OH
A: The objective of the question is to find out the product of the given question.In the question, the…
Q: What is the solubilty of Al(OH)3 in a solution that contains 0.500 AI+ ions? Ksp of Al(OH)3 is 8.89X…
A: Given: Ksp=8.89x10−17;[Al3+]=0.500MStep 1: Write the dissociation of…
Q: How would you synthesize the following substances from benzaldehyde and reagents from the table? In…
A: The given target molecules are shown below.The given reagents are shown below.We have to synthesize…
Q: Consider the phase diagram of carbon dioxide below. Starting from the triple point, the carbon…
A: A question based on phase diagram. For the given CO2 phase diagram the probable change of phase at…
Q: 9. Propose the mechanism for the reaction below using curved arrow: OH HCI + H₂O
A:
Q: v Draw a skeletal ("line") structure of this molecule: OH CH3C-CH2-CH=CH2 OH
A: In a skeletal line structure, the covalent bonds are shown in the form of lines. The atoms other…
Q: The starting material that completes the following reaction is: NaOH, H₂O ? Draw Your Solution H
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 122. * Predict the products of the following reactions. In (a) and (d), give the intermediate…
A: This is allylic halogenation.When N-bromosuccinimide (NBS) is used in presence of carbon…
Q: Sort the following compounds according to their ability to undergo a haloform reaction.
A:
Q: Calculate the molar solubility of Fe(OH)3 (Ksp = 4.0 x 10-38) in each of the following solvents.…
A:
Q: 11. Write the chemical reaction for the acid hydrolysis of each amide sample. Include the condensed…
A: Acid hydrolysis of amide gives carboxylic acid and salt of ammonia .
Q: Но -OH
A: Ketone react with HCN to give cyanohydrin then further hydrolysis to give alpha hydroxy acid
Q: 6) What starting materials (no fewer than four carbon atoms) would be necessary to prepare the…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: How many atoms in the following molecule are likely to participate in hydrogen bonding with another…
A: The objective of the question is to determine the number of atoms in the given molecule that are…
Q: Check the box under each molecule in the table below that is an isomer of this molecule: If there…
A: The objective of the question is to find the isomer of the given molecule from the given option.The…
Q: Your answer is partially correct. The enediol tautomerizes to a ketose sugar before the next…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the aldol…
A: Aldol condensation reaction is a reaction between two carbonyl compounds to form an alpha-beta…
Q: 7. For each of the following compounds, indicate the ring carbon that would be nitrated if the…
A: To determine which carbon atom in 3-nitroanisole would be nitrated when treated with HNO3/H2SO4,…
Q: Calculate the solubility at 25 °C of CuBr in pure water and in 0.33 M KCN. You'll probably find some…
A: Given:Concentration of KCN =0.33 MWe have to calculate the solubility of CuBr in pure water and in…
Q: A student prepares a 1.1 mM aqueous solution of 4-chlorobutanoic acid (C3H6CICO₂H). Calculate the…
A: Please comment down for any doubts. I hope my answer helps you.
Q: A chemist makes 460. mL of barium chloride (BaCl2) working solution by adding distilled water to…
A:
Q: 4. The decomposition of a certain element was observed over a period of 30 hours and the results…
A: The objective of the question is to determine the order of the reaction and the half-life of the…
Q: This question has multiple parts. Work all the parts to get the most points. For the reaction below:…
A: When one mole alcohol adds to an aldehyde or ketone the result is called a hemiacetal.
Q: The base protonation constant K of 1-H-imidazole (C3H4N2) is 9.0 × 108. Calculate the pH of a 1.3 M…
A: Step 1:Step 2: Step 3: Step 4:
Q: Please correct answer and don't use hend raiting
A:
Q: 11. Balance each of the following chemical equations: 1. BC13 (s)+ H2O(1) H3BO3 (aq) + HCl (aq) 2.…
A: To balance the equation the number of atoms on both sides of the given chemical equations should be…
Q: (c) [5 pts] Sketch a graph of the entropy of the van der Waals gas and the entropy of the perfect…
A: The objective of the question is to understand the behavior of entropy for both van der Waals gas…
Q: Predict the major product(s) of the reaction below. OH 2) OH H 1) BH,⚫ THF major product 2) H₂O₂,…
A: Step 1: Hydroboration-Oxidation of alkeneAnti Markovnikov addition of Boron means boron adds to the…
Q: Using ethyl bromide and formaldehyde as your only source of carbons, show the complete synthesis of…
A: The reactants are ethyl bromide and formaldehyde.Approach:We need to consider the functional group…
Q: Draw the skeletal structure of the disulfide formed when the thiol shown is oxidized. SH
A: Thiols, strong nucleophiles with -SH functional groups, are used to protect aldehydes or ketones by…
Q: Show the products of these reactions: + a) PPh3 1) 2) H3O+
A: In organic chemistry, a ylide is a species with opposite formal charges on adjacent atoms.In the…
Q: The solubility-product constants, Kp, at 25 °C for two compounds [iron(II) carbonate, FeCO3, and…
A: Step 1: FeCO3FeCO3 → Fe2+ + CO32-Ksp = [Fe2+][CO32-]2.10 x 10-11 = [1.03 x 10-2][CO32-][CO32-] =…
Q: A 1250 mL solution contains 0.400 M KCгO, and 0.400 M Na₂SO. A 4 concentrated Ba(NO3), solution is…
A: Given thatThe volume of solution is V =1250mLThe molarity of K2CrO4 and Na2SO4 is 0.400M.Where,Ksp…
Q: a) According to the Bohr model for a hydrogen atom, what is the energy di erence between the ground…
A: The objective of the question is to calculate the energy difference between the ground state and the…
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the phthalic acid…
A: The objective of the question is to predict the product formed in the given reaction.The given…
Q: (3). Count the number of electrons for the metals in the following complexes. (you are recommended…
A: The objective of this question is to calculate the electron count for metal centers and ligands in…
Q: None
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: Carvone is the major constituent of spearmint oil. Draw the major organic product of the reaction of…
A:
Q: Give the IUPAC name for this molecule. Do not forget stereochemistry - place in the front, followed…
A: To name an organic compound, the longest C chain of the compound is identified and numbered in such…
Step by step
Solved in 2 steps with 1 images

- Give the following reaction A+3B --->2C.Complete the reaction schemes below providing the products of the reaction schemes below.Which compounds from 5a-d will react with hydroxide in the fashion shown. Which reaction will be the fastest and slowest? Explain using knowledge of carbonyl chemistry, pka and leaving groups.
- Ochem help with reactions... See the attached image... what would be the major product(s) of the following reactions? ThanksDraw themajor product of this reaction. Ignore inorganic byproducts. PPC, CH2Cl2Propose a detailed mechanism for the reaction below (in the attached picture), showing the structure of thestable intermediate and using curved arrows to indicate electron flow in each step



