Q: Draw the Lewis dot structure of the conjugate base of cyclopentane by assuming all the atoms in the…
A: Lewis’s structure: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a…
Q: GC2.3.6 An evacuated vessel containing a small amount of powdered graphite is heated to 1080 K, and…
A: Given,Pressure of CO2 = 0.458 atmAt equilibrium, total pressure inside the vessel =0.757…
Q: . An unknown substance has a van’t Hoff factor of 3.4. Into exactly 1 kg of water, you dissolve 16.2…
A:
Q: Which of the following is a conjugate acid- base pair? A) HCN, H3O+ B) H₂O, HCN C) HCN, CN- D) H₂O*,…
A: Conjugate acid -base pair - When an acid lose a proton then it forms an anion (base). This base is…
Q: Be sure to answer all parts. A gas expands from 252 mL to 936 mL at a constant temperature.…
A: The question is based on the concept of chemical thermodynamics. we need to calculate work done in…
Q: Please write the chemical reaction in fluorescence Riboflavin (Vitamin B2) + Acetic Acid C17H20N4O6…
A: In the given question, we have to complete the given reaction.
Q: Write the IUPAC name for the following molecule.
A: The given compound is a hydrocarbon, cycloalkane. The IUPAC name of a cycloalkane can be written on…
Q: Critical Thinking. If you add NaOH to a solution with pH as in question 1 and stay within the buffer…
A: This question belongs to Equilibrium.
Q: 12. The described electrochemical cell (concentration cell) was set up: Cu | CuSO4 (c1) || CuSO4…
A: The question is based on the concept of redox reactions reactions. Reactions in which…
Q: Be sure to answer all parts. A gas expands from 252 mL to 936 mL at a constant temperature.…
A: Given : V1 = 252 mL = 0.252 L = INITIAL VOLUME V2 = 936 mL = 0.936 L = FINAL VOLUME.…
Q: Enter your answer in the provided box. 0 From the standard enthalpies of formation, calculate AH…
A: We have given the reaction C6H12(l) + 9O2(g) → 6CO2(g( + 6H2O(l) To calculate ∆H°rxn we have to…
Q: be 1 2 3.5 3.5 5 7 3 4 10 5 I need help finding Concentration and here are my volume readings 6…
A: To calculate the concentration of the copper solution, we can use the Beer-Lambert law, which…
Q: What spectroscopic terms arise from the configuration 1s22s22p63s23p63d1? What is the degeneracy?
A: In electronic spectroscopy, an atomic term symbol specifies the certain electronic state of an atom.…
Q: CI CF3 Na, NH3 CH3OH NaNH, A NaNH, A NH₂ 1. HC1/NaNO₂ 2. KI
A: All transformations are shown below.
Q: 3. Predict the product for both steps shown below. HO 1 eq Ho H OH H* OH 1. NaBH 2. H₂O* 1. LAH 2.…
A: We have given the organic reaction and we have to find the major product of reaction.
Q: Which of the reagents listed below would efficiently accomplish the transformation of…
A:
Q: Which of the following would indicate the strongest intermolecular forces in a solid? a low melting…
A: To solve this problem we have to know about effect of intermolecular forces in solid.
Q: --T- ABC 2. The TLC plate (left) shows three lanes; the lower dotted line is where the original…
A: Thin layer chromatography(TLC) is used to separate the compounds based on the relative affinity of…
Q: Nick and Kel reacted 2.80g of NiCl2 x 6H20 with 7.00 ml of 4.00 M C2H8N2. They recovered 2.40g of…
A: In the given question we have to calculate the theoretical yield and actual yield of the following…
Q: Sn4+ (aq) + 2 Br (aq) → Sn2+ (aq) + Br₂(1) Sn²+ (aq) Pb(s) + Fe2+ (aq) → Pb²+ (aq) + Fe(s) Co2+ (aq)…
A: The question is based on the concept of redox reactions reactions. Reactions in which simultaneous…
Q: Direction: Determine the polarity and type/s of intermolecular forces that exist in the following 1)…
A: Intermolecular forces are the attractive forces that arise between the molecules. Types of…
Q: How many moles of oxygen atoms are present in 5.20 moles of Al2(SO4)3? 15.6 moles O 20.8 moles 62.4…
A: Given, moles of Al2(SO4)3 = 5.20 moles moles of Oxygen (O) atoms = ?
Q: Please, I need help to do question 3 for the post-Lab questions. The question is, Which was the…
A: Solution: To calculate the limiting reagent in any chemical reaction we must know about the reaction…
Q: Question 6 I and III The statements numbered above that refer to reduction are III and IV II and III…
A:
Q: The activation energy of a reaction is 56.0 kJ/mol and the frequency factor is 1.5×1011/s .…
A:
Q: calculate the molar mass of potassium hydrogen phthalate
A: Molecular formula of potassium hydrogen phthalate is KHC8H4O4 .
Q: 3. Draw the skeletal structure of 3-methylhexanoic acid. What is the geometry at the carbonyl…
A: Given here the name of a organic acid and we are asked to draw the skeletal structure for it.
Q: 3. Arrange the following intermolecular forces in order of increasing strength (least to highest).…
A: According to the Bartley guidelines for the multiple questions, I can solve only first one. So I'm…
Q: a Calculate the molar concentration of H3O+ in a solution that contains 4.1x10-12 M OH". (Enter your…
A: Autoionization of water takes place as follows: In pure water, the concentration of the hydronium…
Q: 24.7 g of lithium nitrate reacts with 11.5 g of barium sulfate. Find the mass of the projects.
A:
Q: A scientist measures the standard enthalpy change for this reaction to be -739.4 kJ/mol: 2CO(g) + 2…
A: The question is based on the concept of chemical thermodynamics. we need to calculate enthalpy of…
Q: 5. When a particle of mass 9.1 X 10-28 g in a certain one-dimensional box goes from the n = 5 level…
A: Since you have posted multiple questions, we will provide the solution only to the first question as…
Q: НО OH HO Ph
A: The question is based on the concept of organic reactions. we need to convert reactant into product…
Q: What is the pH of an aqueous solution of 0.0464 M barium hydroxide? pH =
A:
Q: Which one of the following statements is true? A) A solution with a pOH of 12.50 will have a higher…
A: pH : pH can be defined as the negative logarithm of hydrogen ion concentration pH = -log[H+] when…
Q: Enter your answer in the provided box. From the following heats of combustion, 3 CH₂OH() + O₂(g) →…
A: Given that, We have to calculate the enthalpy of formation of methanol from the given data.
Q: For a gaseous reaction, standard conditions are 298 K and a partial pressure of 1 bar for all…
A: Given , Reaction : 2 NO(g) + O2(g) ⇌2 NO2(g)
Q: Click in the answer box to activate the palette. Determine the empirical formula of a compound…
A: For determination of empirical formula, first calculate moles of each element and divide it by…
Q: Phthalic acid (H₂CH₂O4) is a diprotic acid with Kal = 1.12 × 10-³ and K₁2 = 3.90 × 10-6. Determine…
A:
Q: ço NH 8 + B1₂ HNO₂ B(OH)₂ 5.0 FeBra AICI₂ H₂SO4 AICI HNO3 Pd(PPh₂) Na₂CO₂ Pd(PPhale Na₂CO₂
A: According to the bartleby guidline, for sub multiple parts I can solve only first three questions.…
Q: In the electrolysis of an acid solution, oxygen can be produced by the following half-reaction:…
A:
Q: Question 8 Use the following information to answer the next question. Equations of Redox Reactions…
A:
Q: The pH of a basic solution is 11.27. What is [H*]?
A: pH = 11.27 [H+] = ?
Q: A sample of solid zinc is heated with an electrical coil. If 70.6 J is added to a 14.8-gram sample…
A:
Q: Be sure to answer all parts. Industrially, vanadium metal, which is used in steel alloys, can be…
A:
Q: Explosive exnon trioxide, XeO3, is prepared as in the below unbalanced equation. In one expriment,…
A:
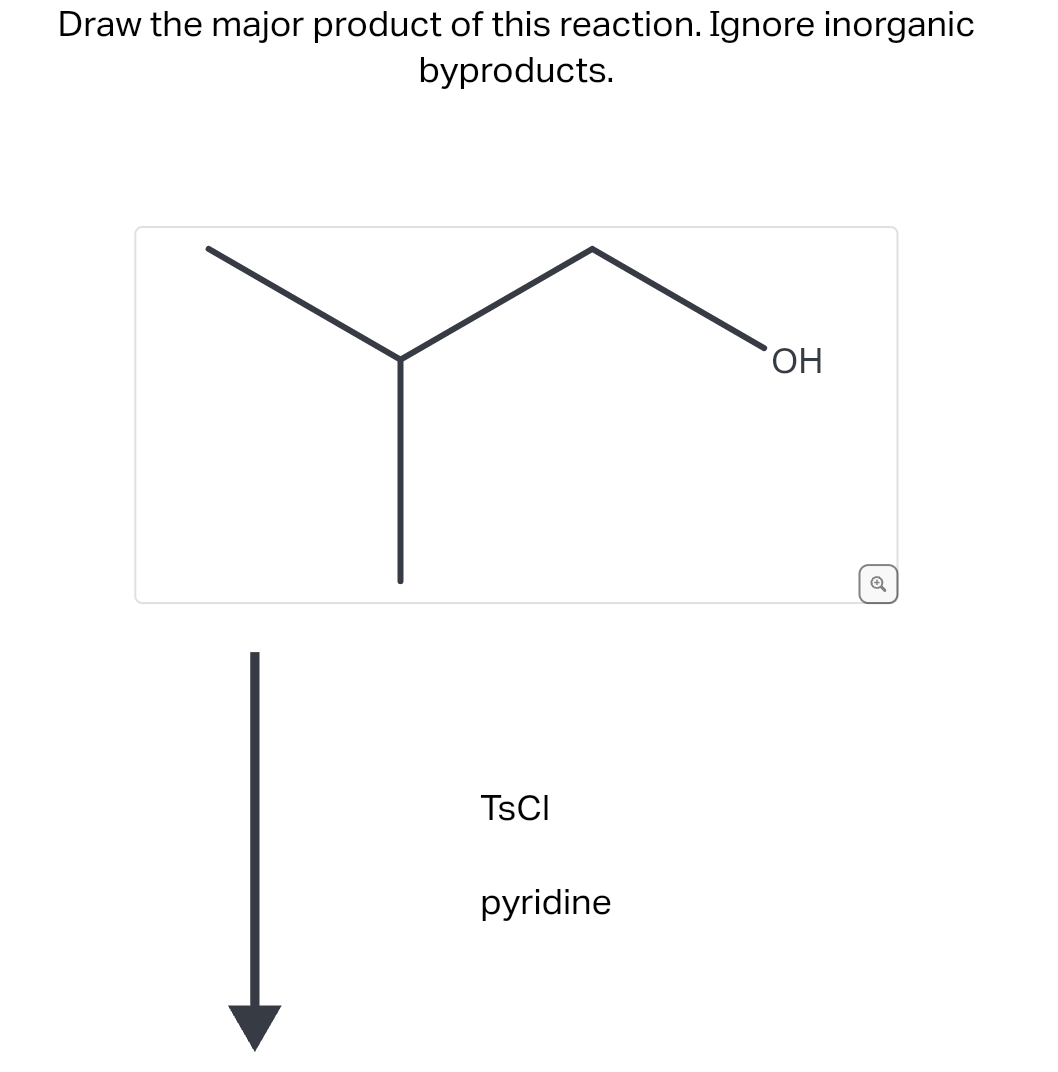
Q: Draw the major product of this reaction. Include stereochemistry if applicable. Ignore byproducts.…
A: E2 elimination: Simultaneous, removal of the group and hydrogen atom in presence of a base to form…
Q: How many polar bonds are represented in this structure? H O 5- O 3 02 H None of the bonds are polar.
A:
Q: 3. Calculate the pressure (in Pa) of argon gas required to produce a collision rate of 4.5 x 1020…
A: Collision rate is defined as the average rate in which two reactants collide and used to express…
Q: The point group for the molecule GaI3 is D3h. Which vibrational modes are infrared active? Raman…
A: Solutions- Raman Active, Infrared active
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Draw the product of the reaction below. Ignore inorganic byproducts.Draw structures to correspond with the following common and systematic names: (e) N,N-dimethylformamide (f) benzoic propionic anhydrideGiven the information pictured, The final step of the reaction sequence uses PhLi. Provide a reason and possible mechanism by which this final elimination and hydrolysis reaction would occur.
- Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA. a. Explain why it can be used only under slightly acidic conditions. b. Explain why it can cross-link DNA.Triethylenemelamine (TEM) is an antitumor agent. Its activity is due to its ability to cross-link DNA. a. Explain why it can be used only under slightly acidic conditions.b. Explain why it can cross-link DNA.An aqueous solution of a primary or secondary amine reacts with an acyl chloride to form an amide as the major product. However, if the amine is tertiary, an amide is not formed. What product is formed? Explain.
- The reaction of an imine with sodium methylsulfide (NaSCH3) is shown below.please DRAW the full mechanism of the Na2S2O4 reduction reaction belowANSWER ALL AND WILL GIVE YOU THUMBS UP AND RATE YOU GOOD Classify the following examples to the given agents. CHOICES: Suspending agent Emulsifying agent QUESTIONS: Bentonite Kaolin Lecithin Carbomer Polysorbate 20 Carrageenan Methyl cellulose Ceteareth 20 Tragacanth Mustard

