Q: Draw the (most likely) major elimination product for the reaction of 2-bromo-2-methylbutane with...…
A: Small nucleophile abstract hydrogen from more substituted side. Bulky nucleophile abstract hydrogen…
Q: Two gases X and Y are found in the atmosphere in only trace amounts because they decompose quickly.…
A:
Q: A chemistry graduate student is studying the rate of this reaction: NH,OH(aq) →NH, (aq)+H,O(aq) She…
A: In the given question, data for the reaction is given and we have to find rate law and rate…
Q: A certain drug has a half-life in the body of 3.5 h. Suppose a patient takes one 300. mg pill at…
A: The half-life is the time required for the substance to become half of its original concentration of…
Q: What is the major product of the following reaction? Cl (CH3)2CuLi CH3 II Li III Cu IV HO CH3 CH3
A: Product of the reaction
Q: Determine the pH during the titration of 21.3 mL of 0.307 M hypochlorous acid (K,= 3.5x108) by 0.391…
A:
Q: The reactant concentration in a zero-order reaction was 0.100 M after 100 s and 4.00x10-2 Mafter 400…
A:
Q: д O2 Cu(bpy) (NMI) cal TEMPO cat acetone х OH ОН но он H₂SO4 cat toluene де НО H₂SO4 cat HO Н OH Ба…
A: A protecting group is a chemical functional group that is temporarily attached to a reactive site on…
Q: Using 4-bromotoluene as starting material, make 4-methylbenzoic acid
A: We have to synthesized 4-Methylbenzoic acid from 4-Bromotoluene.
Q: A. Please provider your answer below. 0₂ ☐☐ At 50 °C, a neutral solution has a pOH = 6.33. What is…
A: pOH = 6.33 at temperature 50°C The autoionization of water is given by the equation: H2O(l) ⇌…
Q: How many electrons in an atom could have these sets of quantum numbers? n = 2 n = 5, f = 1 n = 6,=…
A: we have to calculate the number of electrons for the given quantum numbers
Q: V 1. Identify which of the following are aromatic and explain your reasoning. Ö: H B :0: b. f. :Ö-Ň…
A: This question belong to General Organic chemistry that is Concept of Aromaticity. Aromatic Compound:…
Q: Draw the skeletal structure of the ester formed when carboxylic acid CH₂CH(CH₂)CH₂CH₂CO₂H is treated…
A: Carboxylic Acids on reaction with alcohols in the presence of small amount of acid yield esters.
Q: 5. Devise at least 2 methods of generating a benzene containing molecule from non-aromatic starting…
A:
Q: Calculate the vapor pressure of Hg at 49 °C (in atm). Hg (1) - Hg (g) AH° = 61.32 kJ and AS° = 98.83…
A:
Q: What is the reagent for the following reaction? NH2 SOCI عليه من مي شه NH2 NHSOCI IV NH₂
A:
Q: A sample of xenon gas at a pressure of 0.709 atm and a temperature of 112°C, occupies a volume of…
A: According to Charle's law, volume of a gas is directly proportional to temperature at constant…
Q: Using 4-bromotoluene as starting material, make P-bromophenylethanoic acid
A: The given transformation involves three major steps such as, 1. Bromination at benzylic position…
Q: Strontium chloride and sodium fluoride react to form strontium fluoride and sodium chloride,…
A: Given, SrCl2(aq) + 2 NaF(aq) →SrF2(s) + 2…
Q: Why was it important to keep the dioxolane derivative (an acetal) cold and dry after it was formed?
A: The dioxolane derivative, also known as an acetal, is formed from the reaction of an aldehyde or…
Q: An aqueous solution contains 0.309 M ethylamine (C₂H5NH₂). How many mL of 0.211 M hydroiodic acid…
A: A buffer is a solution which resist any change in pH on adding a small amount of acid or base . it…
Q: The decomposition of a generic diatomic element in its standard state is represented by the equation…
A: Solution- At 2000K ∆G0f =4.43 kJ/mol = 4430 kJ/molWe know, ∆G0f =– RT In Kor, In K=∆G0f-RThere, In…
Q: The pH of solution A1, solution A2, solution A3 and solution A4 are 1.0, 2.0, 3.0, and 4.0,…
A:
Q: 13. Provide the reagents necessary to carry out the following conversion. ocor CN
A:
Q: Solid CO₂ (0.35 mol) is reacted with CH3MgBr (0.20 mol) in ether solvent. The product of the…
A:
Q: A 5.65 L vessel contains 22.0 g of PC13 and 3.35 g of O₂ at 15.0 °C. The vessel is heated to 215 °C,…
A: Mass of PCl3 given = 22 g and mass of O2= 3.35 g
Q: Write a balanced net ionic equation for the reaction of [Cr(C₂H3O2)2(H₂O)]2 with air in the presence…
A: We have find out the answer.
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free…
A:
Q: A voltaic cell is constructed in which the cathode is a standard hydrogen electrode and the anode is…
A:
Q: What are the products of the following reactions: a) ● b) c) 1. AICI3 2. H₂O 1. AICI3 2. H₂O 1.…
A: In Friedel Crafts reactions, the lewis acid ( example ; AlCl3) is used to generate electrophile.
Q: 6. What is the mass, in g, of a piece of iron that has a volume of 1.50 x 10² and a density of 7.20…
A: The mass of a substance is equal to its density multiplied by its volume. In this case, the volume…
Q: An iron nail rusts when exposed to oxygen. For the following reaction, 4.91 grams of oxygen gas are…
A:
Q: я NaBH4 OCH₂CH3 CH3CH₂OH
A: NaBH4 is reduces only ketone not ester.
Q: Which of the following has polar covalent bonds but is nonpolar? SN₂ O SC1₂ O CH3CH3 O HCI O CH,O O…
A: Molecules
Q: 8. When treated with hydroxide, trans-A yields B. However, when cis-A is treated with hydroxide, no…
A:
Q: In a 0.73 mM aqueous solution of trimethylacetic acid (C₂H,CO₂H), what is the percentage of…
A:
Q: Find the pH during a titration of 1.00 ml of 0.600 M analyte when a) 60.00 ml b) 120.00 ml of 0.0100…
A:
Q: By first working out how many stereogenic centers are present in the following molecules, determine…
A: Stereogenic center:- A caron which have four different groups or atoms attached is called as chiral…
Q: a. b. Consider the reaction between nitrogen dioxide and carbon monoxide: NO2(g) + CO(g) NO(g) +…
A: Rate law for the reaction Rate constant
Q: If 25.00 mL of a 0.100 M solution of hydrochloric acid are titrated with 0.100 M sodium hydroxide…
A: Molarity of HCl = 0.100 M Volume of HCl = 25.00 mL Molarity of NaOH = 0.100 M Volume of NaOH =…
Q: a)What is an isomer? b) What is a polymer, and a monomer? Please give examples. (2 points).…
A: This answer provides definitions and examples of important chemical concepts. It begins by defining…
Q: Consider the following data on some weak acids and weak bases: name hydrofluoric acid hydrocyanic…
A: The question is based on the concept of salt hydrolysis. We need to arrange given salts in…
Q: Develop a likely mechanism of the formation: A) Diethylketone B) 4 - hydroxypentanal C) 4,5…
A: Write the mechanism of A to E
Q: Convert 5.0 qt to: a. b. C. gal L mL
A:
Q: A student wishes to determine the chloride ion concentration in a water sample at 25 °C using a…
A: Silver Half cell : AgCI (s) +e-→Ag(s) + CI-(aq) E°red=0.2223 V Copper electrode : Cu2++2e- →…
Q: 9) NH3 + H2O ------> NH4++OH- 0.1 M NH3 ionizes 1% in water according to the above reaction.…
A: Given -: 0.1 M NH3 ionizes 1 % in Water Kb for NH3 = 1.8 × 10-5 NH3 + H2O…
Q: The weak acid HX has a pKa = 4.22. If 20.00 mL of 0.100 M HX are titrated with 0.100 M sodium…
A: The question is based on the titrations. we are titrating a weak acid with a strong base sodium…
Q: Specify the number of tertiary carbons in the molecule below. l 04 01 02 3
A: A tertiary carbon atom is a carbon atom which bound to three other carbon atoms.tertiary carbon…
Q: How many Calories should there be in three grams of pure sucrose? A.4 B. 12 C. 18 D. 8 E. None of…
A: Mass of pure sucrose = 3 grams
Q: An old sample of concentrated sulfuric acid to be used in the laboratory is approximately 97.5…
A:
help #5
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- The 1,2‑dibromide is synthesized from an alkene starting material. Draw the alkene starting material. Clearly, show stereochemistry of the alkene.Fill in the missing reagents below.1. When you add H-Br in a terninal alkene, the product has the rule od Markovnikov? 2. The type pf intermetary thag forms when we add HgSO4/H2SO4/H2O in an alkyl is? Please solve my both questions.thank you.
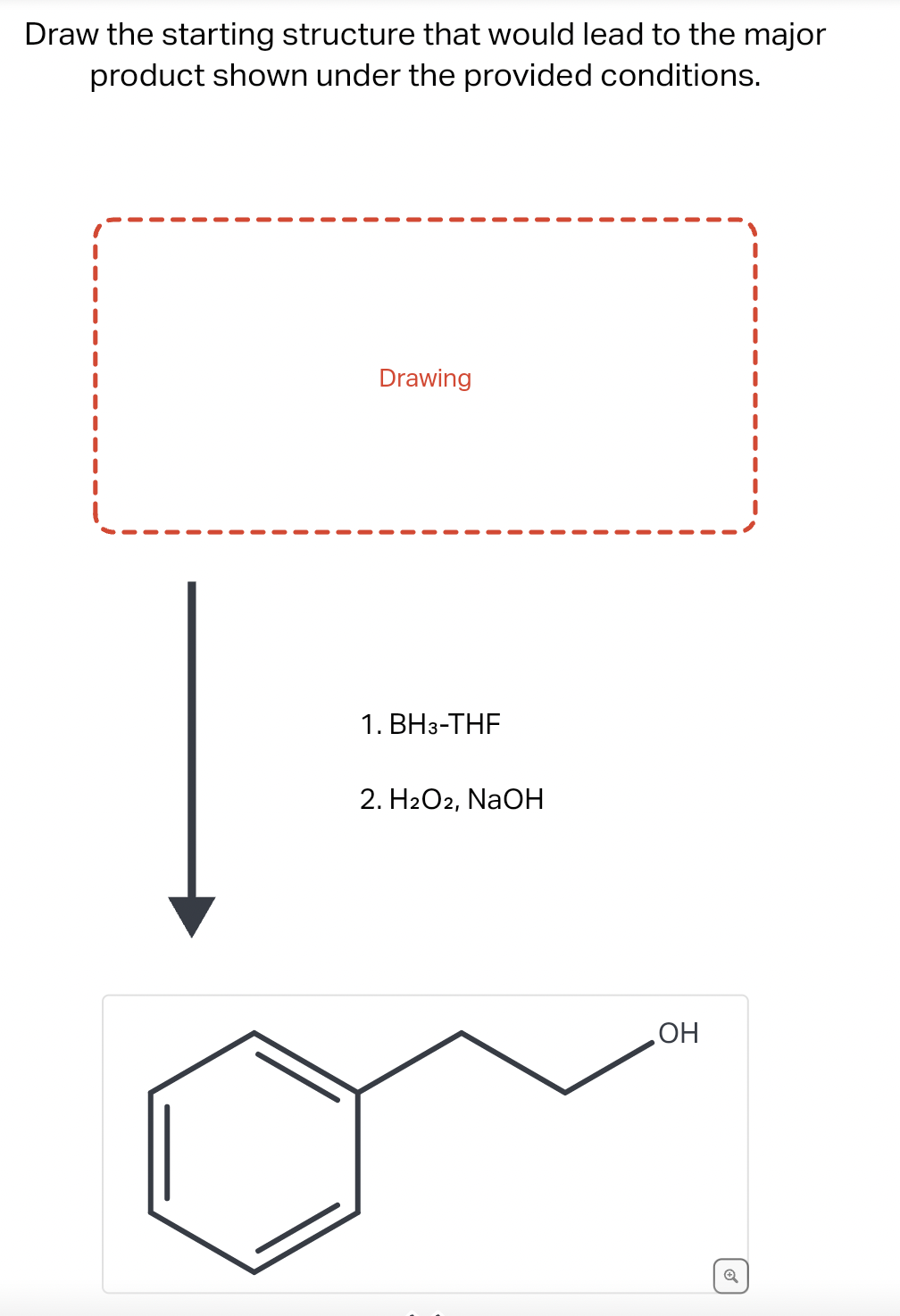
- Supply the missing reagent in the following reaction: (see attachment) A. Lindlar, H2, mCPBA B. O3 or KMnO4 C. fused KOH D. NH3 E. NaNH2Complete the following reaction equations :(i) C6H5Cl + CH3COCl →(ii) C2H5NH2 + C6H5SO2Cl →(iii) C2H5NH2 + HNO2 →Draaw the starting structure that would lead to the major product shown under the provided conditions
- A hydrocarbon of unknown structure has the formula C8H10. On catalytichydrogenation over the Lindlar catalyst, 1 equivalent of H2 is absorbed. Onhydrogenation over a palladium catalyst, 3 equivalents of H2 are absorbed.(a) How rnany degrees of unsaturation are present in the unknown?(b) How many triple bonds are present?(c) How many double bonds are present?(d) How many rings ar e present?(e) Draw a structure that fits the data.Which structure will not yield cis- or trans- isomers after a reaction with H2/Lindlar catalyst or Na/NH3?KMnO4, warm, conc'd reacts with hept-1-ene to yield __________. CO2, hex-1-ene CO2, hexanoic acid Formic acid, pentanoic acid Ethanoic acid, pentanal Formic acid, hexanone
- How to draw the potential energy diagram for this reaction? C₃H₈O (l) → C3H6 (g) + H2O (l) propan-1-ol → propene + waterWhat is the main product formed from the reaction below?When toluene is treated with sulfuric and nitric acids under special conditions, three nitro (NO2) groups are substituted for hydrogens at the 2, 4 and 6 positions on the ring (the next section discusses why the 2, 4, and 6 positions are substituted). The product is a highly explosive substance called 2,4,6-trinitrotoluene. This subastance is commonly known by a three letter name. What is it?

