Q: Identify how the compounds below are related to one another. Select the single best answer.…
A: Answer: (A) EnantiomersExplanation:Answer: (A) Enantiomers: Stereoisomers that are…
Q: Ph3P
A: An ylide is a molecule that is electrically neutral and has both positive and negative charges…
Q: Select the missing reagent to achieve the following transformat +? cat. FeCl3 9) Litcp b) Naci c)…
A: Chlorobenzene can be formed from benzene through a substitution reaction called electrophilic…
Q: What is the [HA] (in mol L-1) of a solution containing 0.846 mol L-1 of a diprotic acid with PKA₁ =…
A: A diprotic acid dissociates to produce two H3O+ ions per acid molecule when dissolved in water.
Q: Macmillan Learning For a particular isomer of CH₁g, the combustion reaction produces 5108.7 kJ of…
A: The isomer is of C8H18 The combustion reaction is as follows:C8H18(g)+25/2…
Q: 1 A chemist dissolves 441. mg of pure barium hydroxide in enough water to make up 90. mL of…
A: The values are provided in questionmass of barium hydroxide = 441 mgmass of barium hydroxide = 441…
Q: Complete the table: Element Mass Moles Number of Atoms Na 45.5 mg C 1.56 212 V Hg 1.16 kg
A: Atomic mass of Na = 22.99 g/molAtomic mass of C = 12.01 g/molAtomic mass of V = 50.94 g/molAtomic…
Q: Suppose a 500. mL flask is filled with 0.90 mol of CO, 1.8 mol of CO2 and 1.4 mol of H2. The…
A: Volume of flask = 500 mL = 0.5 LMoles of CO = 0.9 molMoles of H2= 1.4 molMoles of CO2 = 1.80 molK =…
Q: The number of spin allowed electronic transition states for the complex [Ni(en),] are O 2 3 0
A: The objective of the question is to find the number of spins allowed for electronic transition for…
Q: Using the tables for water, determine the specified property data at the indicated states. (a) At p…
A: Given:Pressure P = 3 barSpecific volume m3/kgTo find:Temperature, in °C Specific internal energy,…
Q: Calculate the value of the equlibrium constant Kc for this reaction rounding to two significant…
A: The objective of the question is to determine the equilibrium constant for the reaction.
Q: A molecule analogous to camphor is 4-t-butyl- cyclohexanone, seen below. Speculate on whether the…
A: In a cyclohexene conformation the tertiary butyl group is bulkier group present at equatorial…
Q: Name this alcohol according to the IUPAC system. Include stereochemistry. CH3 OH
A: Given,The molecule is:
Q: IV. Draw chemical structures and equations to show how the following transformation below may be…
A: Benzene undergoes nitration in the presence of strong acids like to form nitrobenzene.Nitrobenzene…
Q: Product ? Give correct typing answer
A: The final product of the given reaction is 3-chloro-3-propyl cyclohexene.Explanation:Step 1 :…
Q: How do I convert Hepta-5-yne-2,4-dione to 4-hydroxy-2-butanone?
A: To convert hepta-5-yne-2,4-dione to 4-hydroxy-2-butanone, we would carry out the following…
Q: A 45.5-g sample of copper at 99.8 °C is dropped into a beaker containing 152 g of water at 18.5 °C.…
A: Given: Mass of copper = 45.5gMass of water = 152gInitial temperature of copper = Initial temperature…
Q: HO H HO H C HO HO V/! H , כול HO | H C HO-> HO HO H /! ! H₂C
A: The carbohydrate that can react with Tollens reagent ( Ammoniacal Silver nitrate solution) , Fehling…
Q: Which option is the correct line-bond structure of the given molecule? CH3 H-C-CC-H CH3
A: Answer:-This question is answered by using the simple concept of writing the bond line structure of…
Q: 7 With the Polygon in a Circle method, which one has a configuration? ⑦With the shell closed shell
A: Open shell configuration means there is the presence of unpaired electrons, i.e., there are some…
Q: CH3 Ph. steps OH H3C 'N' Ph CH3 H
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Tooth enamel consists of hydroxyapatite, Ca5(PO4)3OH (Ksp = 8 × 10−37). Fluoride ion added to…
A: The objective of this question is to calculate the solubility of hydroxyapatite (Ca5(PO4)3OH) and…
Q: Which element below has two unpaired electrons? Se Be Ca He
A: It refers to a singly occupied electron in an orbital rather than an electron pair.
Q: What is the relationship between the parent structure on the left and the structure on the right? Me…
A: Enantiomers : Stereoisomers which are non-superimposable mirror images of each other and rotate the…
Q: Suppose a 500. mL flask is filled with 1.5 mol of Cl₂, 1.3 mol of HCl and 0.60 mol of CCI. The…
A: Answer:A reaction is said to at equilibrium if the forward reaction rate is equal to the rate of…
Q: 3. Please show the mechanism for the following reaction steps. 2. HCl/H₂O MgBr
A:
Q: Enzymes function most efficiently at the temperature of a typical cell, which is 37 degrees Celsius.…
A: The question is asking about the significance of temperature regulation in organisms and how this…
Q: A2B A2BA AB AB2 Number of Electrons Given the data below, if atoms of A would react with atoms of B,…
A: The molecular formula will be: AB2Explanation:Thank You.
Q: What is the rate law? ( Use k for the rate constant. ) Rate = K [NO]2 [Cl2] b…
A: Rate Law: The relationship between a chemical reaction's rate and the reactant concentrations…
Q: Draw the structure(s) of the major product(s) of the following reaction after workup to neutralize…
A: “Since you have posted a question with multiple questions, we will provide the solution only to the…
Q: Calculate the K₂ value for the following acids. (Enter your answer to three significant figures.) a)…
A: Since,Ka is the dissociation constant of acid.The negative logarithm of Ka value is known as pKa…
Q: What are the planes of symmetry and proper rotation of [Co(en)2Cl2]+?
A: The objective of the question is to identify the planes of symmetry and proper rotation of the…
Q: A sample of a gas in a cylindrical chamber with a movable piston occupied a volume of 0.00256 cubic…
A: Given: Initial volume, V1 = 0.00256 m3 = 2.56 L [ 1000 L = m3…
Q: csco (h) CH,CO CO,H
A: The given equation is a chemical reaction between Cesium Carbonate (CsCO3) and Acetic Acid…
Q: 19. What is the major organic product obtained from the following reaction? PCC OH CH2Cl2 OH OH 2 HO…
A: The objective of the question is to identify the major organic product obtained from the reaction of…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. 1 H3O+ Drawing a
A: Given,The reaction is:
Q: for the uncatalyzed reaction O3 (g) + O (g) -----> 2O2 (g) is 14.0 kJ. Eafor the same reaction…
A: The objective of this question is to find the ratio of the rate constant for the catalyzed reaction…
Q: CE 3500 Copyright 1994 300 2500 1501.53 2H 3H 2H zi mat 91-1221 254 740.55 Эн порнороско 8.4 8.2 9.0…
A: The characteristic bands can be tabulated as; Wavenumber (cm-1)Bondvibration shape intensity…
Q: Problem 1 Explain the symmetry elements of the crystals that have the following space groups: P4"21…
A: The objective of the question is to explain the symmetry elements of the crystals that have the…
Q: Determine the number of grams of NH Br needed to prepare an aqueous solution at a specific pH by…
A:
Q: Question Content Area The reaction of NO2 (g) and CO (g) is thought to occur in two steps: Step 1…
A: The objective of the question is to find the overall stoichiometric reaction from the given two-step…
Q: 19. What is the major organic product obtained from the following reaction? Η ΟΗ 1 2 PCC ΟΗ CH2C12 3…
A: PCC Pyridinium chlorochromate is a weak oxidizing agent, it cannot oxidize the primary alcohols…
Q: Draw the major 1,2- and 1,4-addition products formed when this diene reacts with H3O+. Do not…
A: ->Dienes have nucleophilic character which can give electrophilic addition reaction to…
Q: For the molecule below: 1. Identify all CHIRAL centers if any are present: 2. Name each CHIRAL…
A: we have to find chiral centres and name the chiral centres.
Q: Me HO H H. HO
A: We have given a sequence of reaction in which alcoholic functional group is first converted into…
Q: Balance the following gas-phase reaction and write its reaction quotient, Qc: SClF5 (g) + H2(g) ⇌…
A: We have to balance the following gas-phase reaction and write its reaction quotient, Qc: SClF5 (g) +…
Q: At a certain temperature, the equilibrium constant K for the following reaction is 0.86: Br2(g) +…
A:
Q: A chemist is studying the following equilibrium, which has the given equilibrium constant at a…
A:
Q: Which of the statements most accurately describes the following electron configuration? 1 b 1 a. It…
A: The objective of the question is to identify the correct option.
Q: (c) 100.9 mL of 0.49 M HBr titrated with 0.25 M KOH The acid is: The base is: The halfway point pH…
A: Answer:During acid-base titration, where acid is the analyte and base is titrant, half way point is…
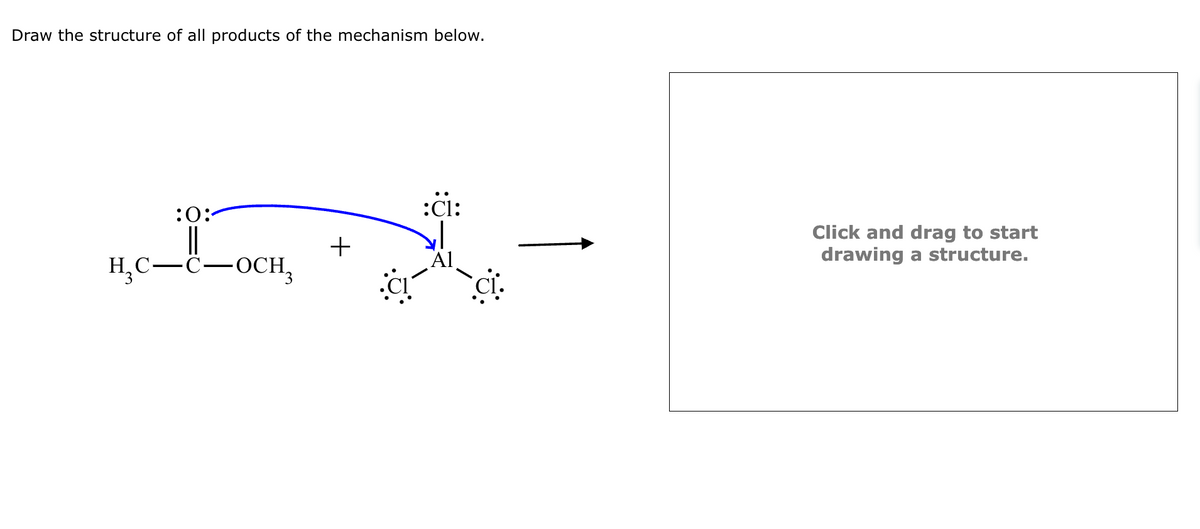
Step by step
Solved in 1 steps with 2 images

- Identify F in the following reaction sequence. F was converted in several steps to the antidepressant paroxetine (trade name Paxil; see also Problem 9.9).Please draw the mechanism (assuming an SN1pathway) for the reaction of cyclopentyl bromide with the cyanide anion (-CN) to yield cyclopentyl cyanide as the organic product Thank youDraw a stepwise mechanism for the attached reaction that illustrateshow two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reactionconditions, even though it is a 1 ° alkyl halide.
- Complete the reaction by drawing the reactions major organic product and propose a detailed mecanism for it's formationPlease Answer the following question with proper explanation and mechanism with Chemdraw!Draw the product of this reaction and states its IUPAC name, and states what type of mechanism is occuring (eg. SN1, SN2, E1, E2, etc)?
- . Complete the following reaction schemes using appropriate reagent(s) and predictproduct eGive the complete mechanism with arrows and the molecules below.The reaction of cyclohexene with iodobenzene under Heck conditions forms E, a coupling product with the new phenyl group on the allylic carbon, but none of the “expected” coupling product F with the phenyl group bonded directly to the carbon–carbon double bond. Draw a stepwise mechanism that illustrates how E is formed.
- Please draw the complete mechanism for the reaction below(A) Provide the major organic product for the reaction below (B) Would the product be optically active of optically in active?I need an explanation on the mechanism, linked bellow 1 paragraph explanation on the steps and mechanism. and what the final product is called

