Chapter14: Conjugated Compounds And Ultraviolet Spectroscopy
Section14.1: Stability Of Conjugated Dienes: Molecular Orbital Theory
Problem 1P
Related questions
Question
6
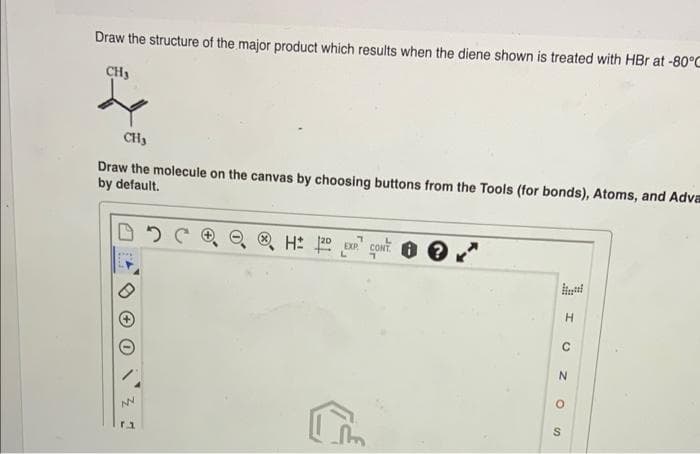
Transcribed Image Text:Draw the structure of the major product which results when the diene shown is treated with HBr at -80°C
CH3
CH3
Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Adva
by default.
O, H: E cONT.
EXP
I O Z O

Transcribed Image Text:When (S)-3-bromopent-1-ene is heated in water, which of the following compounds is not produced?
O (E)-pent-2-en-1-ol
O (S)-pent-1-en-3-ol
O (R)-pent-1-en-3-ol
pent-4-en-1-ol
O (Z)-pent-2-en-1-ol
Submit
Request Answer
Provide Feedback
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning