A 50.0 mL of 0.02 F ammonia solution is added to a solution containing 0.004 g Ni2, 0.003 g Mn2, and 0.002 g Cr2 per 750 ml. a calculate whether or not a precipitate will torm b. if answer to (aj is yes, formulas of precipitates C. which ion wil precipitate first d what pH iS recessary to prevent precipitation of all cations
A 50.0 mL of 0.02 F ammonia solution is added to a solution containing 0.004 g Ni2, 0.003 g Mn2, and 0.002 g Cr2 per 750 ml. a calculate whether or not a precipitate will torm b. if answer to (aj is yes, formulas of precipitates C. which ion wil precipitate first d what pH iS recessary to prevent precipitation of all cations
Chapter12: Gravimetric Methods Of Analysis
Section: Chapter Questions
Problem 12.32QAP
Related questions
Question
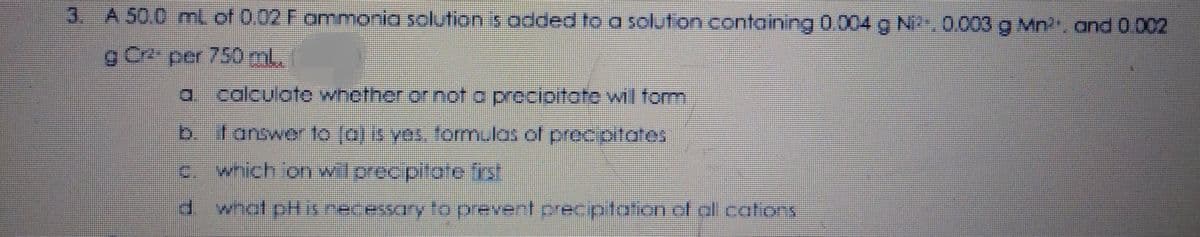
Transcribed Image Text:3. A 50.0 mL of 0.02 F ammonia solution is added to a solution containing 0.004 g Ni.0.003 g Mn. and 0.002
g Cr2 per 750 ml.
calculote whether or not o procioitate wil form
b. fanswer to (o) is ves. formulos of precipitates
C which onwil prec pitate fist
d what pHis necessary to pevent prec ptaion of al catons
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 8 steps with 8 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole