Q: 1. Predict the product of the reaction below. Co,CH3 Product A Diels-Alder Product (unstable) H.
A: In diels-alder reaction there is no intermediate is formed.
Q: 12. Show how to convert cyclopentene into these compounds: ams bos Br (b) (a) Br (c) (d) OH Br
A: We have to predict the reagents for given conversions.
Q: Consider the following scheme: Br CH3 H3C-CH CH-CH2-CH-CH CH3 H-CEC-H B (Acetylene) Diels-Alder…
A: A chemical reaction between a conjugated diene and a substituted alkene, which is known as…
Q: 3. What diene and dienophile should be used to synthesize the following compounds in a Diels-Alder…
A:
Q: Draw the major products A, B, and C for the Diels-Alder reactions below. N- A EtO2C. B +
A:
Q: HC! OH H20
A:
Q: Fill in the final products for the following Diels-Alder reactions: CH₂ H CN (1) CN (2) \\_// CH₂…
A: In this question we have to tell the product of the reaction.
Q: Draw resonance structures for the following compounds: CH2 Why does the molecule below fail to…
A: we have to draw the resonating structure of compound benzylic carbocation.
Q: Draw the products of the Diels-Alder reaction of 1,3-butadiene with cis-1,2-dibromoethene. Use…
A: Introduction: Diels-Alder reaction is a cycloaddition reaction of diene and dienophile to form a…
Q: Draw the π MO energy diagram for [10]annulene, similar to those in Figures 14-19 and 14-21. Fill up…
A: [10] annulene is also called cyclodecapentaene. Its formula is C10H10. The structure of [10]…
Q: 14. What is the pro 15. What is the nar A) Fischer B) Friedel-c C) Diels-Ald D) Wittig E) Grignarc…
A:
Q: Diels Alder product of Buta-1,3-diene and 1-Chloropent-2-ene is: CH2CH3 A *CH2CI B None of the given…
A:
Q: Which structure represents (Z)-4-bromohexa-1,3-diene? Br Br Br Br D
A:
Q: Consider the Diels-Alder reaction shown. Draw the structures of the diene and dienophile that yield…
A: Diel Alders reaction is a cycloaddition reaction involving a reaction between diene reacts with a…
Q: Which statement about the Diels-Alder reaction below is true ? CHO CHO CHO CHO (A) CHO (B) Product…
A: Diels Alder reaction is also termed as (4+2) cycloaddition reaction which occurs under thermal…
Q: d) The Diels Alder reaction between diene 4 and dienophile 5 can give four products, draw the four…
A:
Q: 4. Propose a synthesis plan for these molecules (a) (b) Br. OEt from a Wittig reaction from benzene…
A: Concept : Retrosynthesis 1) it is also called as back synthesis or reverse synthesis. 2) it is an…
Q: What diene and dienophile are used in the Diels-Alder route to the compound shown? CN H CN H
A:
Q: 2) Predict the major product for each Diels-Alder reaction. Include stereochemistry where…
A: All these are examples of Diels alder reaction. If there is possibility of formation of exo and endo…
Q: Which diene is more reactive in a Diels–Alder reaction?
A:
Q: Draw the product formed when each diene and dienophile react in a Diels-Alder reaction. соон CH3 a.…
A: Given reactions,
Q: In a Diels-Alder reaction, compound will react the fastest and _ will react the slowest. CN NH2 CN…
A: Dienes react with dienophiles in Diels alder reactions. Dienophile should have electron withdrawing…
Q: 3) Predict the major product for each Diels-Alder reaction. Include stereochemistry where…
A:
Q: 4.) For the following dienes A-G, please circle which organic molecules would not function as dienes…
A: Diela-Alder reactions are a type of cyclic reactions. The reaction is taking place between a…
Q: What combination of reactants would give the product P in a Diels-Alder reaction? COOH "COOH HOOC,…
A: the detailed answer is attached below.
Q: Select the best answer that identifies the dienes that would react with an alkene in a Diels-Alder…
A:
Q: CH,CH,CH, CH3 C=C (CH32CH H. H. CH3 C=C C=C H,C CH,CH3 H CHCH,CH,CH3 CH3 CH3 CH3 C Which of the…
A: Stability of an alkene is governed by Saytzeff's rule. It says that more alkylated the alkene the…
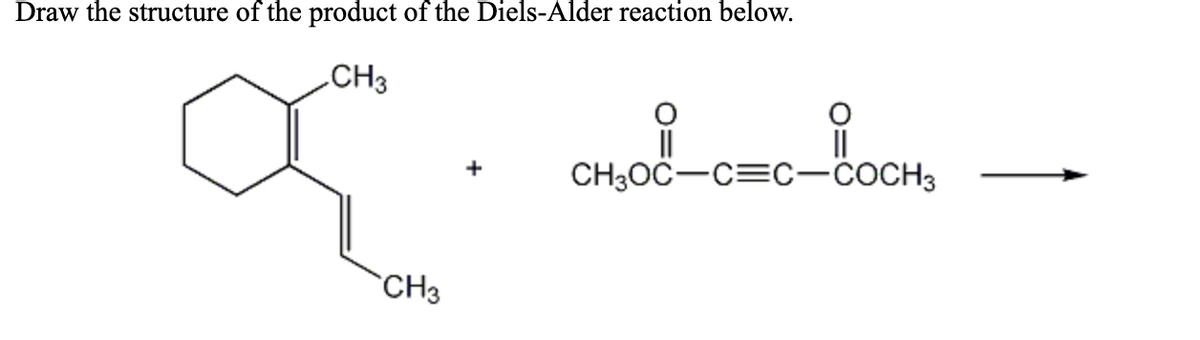
Q: coOCHy +. COOCH3 Predict the praducts of the following Diels-Alder reactions?
A: Welcome to bartleby !
Q: H ОНС H. H. CH3 H CHO Hi CH3 H CN NC H a b d Rank these dienophiles in decreasing order of…
A: The reaction of conjugated dienes with alkenes to form 6 membered cyclic adduct is named as…
Q: Considering both 5-methylcyclopenta-1,3-diene (A) and 7-methylcyclohepta-1,3,5-triene (B), which…
A: On losing Hb, the compound gain aromaticity. Thus, Hb will be the most acidic, and Ha will be the…
Q: 8. Which factors affect the reaction rate in Diels-Alder reactions? OA. Electron donating group on…
A:
Q: Gibberellic acid is a plant hormone that controls the development of plants. At various points in…
A: diels-Alder reactions are highly stereospecific reactions between dienes and dienophiles with the…
Q: Diels Alder does the product is correct? CH2CH3 product CHECH3 H3 CHC CHECH3
A: Given: Diel Alder reaction To find: To check the product of the above reaction
Q: Draw the starting material necessary to prepare the following product by a Diels-Alder reaction. Br…
A: Here the product contains a six membered ring. Therefore, this product can be made by using [4+2]…
Q: Draw the products of the following Diels-Alder Reactions. Remember to show the correct…
A: In Diels alder reaction, diene react with dienophile to form 4+2 cycloaddition product.
Q: 5. Show how you would prepare each of the following compounds using a Diels-Alder reaction: CH3 CN…
A: Given here two Diels alder reaction adduct and we are asked to write the mechanism for the formation…
Q: mpis leit eck my Click the "draw structure" button to launch the drawing utility. Draw the product…
A:
Q: Considering both 5-methylcyclopenta-1,3-diene (A) and 7-methylcyclohepta-1,3,5-triene (B), which…
A: In case of A, if we remove the Hb+ then we will have a -ve charge on carbon which is in conjugation…
Q: Draw the correct product for the following Diels-Alder reaction: 4.0-
A: The correct product of the given Diels-Alder reaction is to be determined.
Q: Compounds containing triple bonds are also Diels–Alder dienophiles. With this in mind, draw the…
A:
Q: Draw one enantiomer of the major product of the following Diels- Alder reaction. (Use wedge/dash…
A: Concerted reaction of diene with dienophile gives Diels-Alder product. The presence of electron…
Q: Heat HBr 40 C
A: In presence of heat, 1,3- butadiene system undergo diels alder reaction with dienophile .
Q: Considering both 5-methylcyclopenta-1,3-diene (A) and 7-methylcyclohepta-1,3,5-triene (B), which…
A: In case of A, if we remove the Hb+ than we will have a -ve charge on carbon which is in conjugation…
Q: Which diene is more reactive in a Diels–Alder reaction? CH2=CHCH=CHOCH3 or CH2=CHCH=CHCH2OCH3
A: Diels-Alder reactions are very important reactions in organic synthesis. In this reaction, the diene…
Q: Which diene would be the most reactive in a Diels-Alder Reaction? ob. Oc.
A: Diels–Alder reaction can be described as a chemical reaction between the substituted alkene (i.e.…
Q: What is the major product of the following Diels-Alder reaction? CN NC CN CN CN CN CN CN CN A. C D.…
A: This reaction is an example of pericyclic reaction. Here two molecule reacts to form a single…
Q: Predict the products of the following Diels-Alder reactions. Include stereochemistry where…
A: Diels–Alder reaction is the cycloaddition of a diene and a dienophile by a concerted mechanism. A…
Q: _7 Which of the following dienes would react fastest in a Diels-Alder reaction? ¿ ¿ ¿ он NO2 CN I II…
A: A multiple choice question about diene in Diels-Alder reaction, which has to be answered.
Q: NC 기 CN NC 소 CN NC
A: Intrdouction : We have to make the products .These are pericylic reactions .
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Diels–Alder reaction of a monosubstituted diene (such as CH2=CH–CH=CHOCH3) with a monosubstituted dienophile (such as CH2=CHCHO)gives a mixture of products, but the 1,2-disubstituted product oftenpredominates. Draw the resonance hybrid for each reactant, and use thecharge distribution of the hybrids to explain why the 1,2-disubstitutedproduct is the major product.Kk.41. please give the diene and dieneophile for these Diels-Alder productsDiels–Alder reaction of a monosubstituted diene (such as CH2=CH– CH=CHOCH3) with a monosubstituted dienophile (such as CH2=CHCHO) gives a mixture of products, but the 1,2-disubstituted product often predominates. Draw the resonance hybrid for each reactant, and use the charge distribution of the hybrids to explain why the 1,2-disubstituted product is the major product.
- Diels—Alder reaction of a monosubstituted diene (such as CH2 = CH – CH = CHOCH3) with a monosubstituted dienophile (such as CH2 = CHCHO) gives a mixture of products, but the 1,2-disubstituted product often predominates. Draw the resonance hybrid for each reactant and use the charge distribution of the hybrids to explain why the 1,2-disubstituted product is the major product.Choose the correct option. 1. Which of the following reactions best describes the Diels-Alder reaction?A)electrocyclic reactionB)cycloaddition reactionC)sigmatropic reactionD)radical reactionE)nucleophilic substitution reaction 2.Which of the following statements best describes the theory of Conservation of Orbital Symmetry?A)Molecular orbital of the transition state must be similar to that of the reactant.B)Molecular orbital of the transition state must be similar to that of the product.C)Only s orbitals from reactants and products are utilized.D)Molecular orbitals of reactant and product must have similar symmetry.E)Molecular orbitals of reactant and product must have different symmetry. 3.Through what type of single pericyclic reaction did the reaction proceed? 5-methylcyclopenta-1,3-diene to 1-methylcyclopenta-1,3-dieneA)[1,2] sigmatropic hydrogen migrationB)[1,3] sigmatropic hydrogen migrationC)[1,4] sigmatropic hydrogen migrationD)[1,5] sigmatropic hydrogen migrationE)none of the…Show that the [4 + 2] Diels–Alder reaction is photochemically forbidden.P
- In which species (NO3- or CH3NO2) are the N-Obond(s) longer? In which species (NO3- or CH3NO2) are the N-Obond(s) weaker?Considering both 5-methylcyclopenta-1,3-diene (A) and 7methylcyclohepta-1,3,5-triene (B), which labeled H atom is most acidic? Which labeled H atom is least acidic? Explain your choices.(I) Give major products for 1,2-/1,4 additions (A) Diels-Alder (B-D) cycloadditions, [2+2] cycloaddition, and Retro-Dial-Alder (E) cycloaddition. Illustrate the regiochemistry and stereochemistry (meal, enantiomer, exo or Endo) for cycloaddition B-D.
- In 4+2 cycloaddition, ENDO stereochemistry is preferred because: endo stereochemistry allows the diene to adopt s-cis conformation. endo stereochemistry results in less steric hindrance. the transition state leading to endo stereochemistry is higher energy. it allows for additional π to π overlap between the diene and the dienophile.Can someone draw the arrows for this reaction so I can understand how the product was formed? I'm having trouble understanding that aspect of the Diels-Alder reaction.Considering both 5-methylcyclopenta-1,3-diene (A) and 7-methylcyclohepta-1,3,5-triene (B), which labeled H atom is most acidic?Which labeled H atom is least acidic? Explain your choices.



