Q: Determine the number of atoms of C in 42.8 grams of C₆H₁₂O₆.
A: The objective of this question is to find out the number of carbon atoms in a given mass of glucose…
Q: Determine the mass, in grams, of 0.785 moles of Si (1 mol of Si has a mass of 28.09 g)
A: The number of molecules or atoms present in the one mole of the substance is equal to the Avogadro…
Q: Which of the following enzymes catalyzes a reaction with the pictured compound as an intermediate?…
A: Succinyl CoA synthetase facilitates a reversible process wherein Succinyl CoA reacts with inorganic…
Q: Provide the correct IUPAC name for the compound shown here.
A: Since,Rule of IUPAC-1) Longest chain as parent chain.2) Numbering start from those side where more…
Q: k Draw the major elimination and substitution products formed in this reaction. Use a dash or wedge…
A: The objective of the question is to find the major elimination and substituted products that formed…
Q: Give the reagents and conditions to provide the following transformations, usethe 1. 2. etc.…
A: The required reagents to carry out the reaction are clearly mentioned. Please find the attachment…
Q: Determine AS for the phase change of 1.37 moles of naphthalene from solid to liquid at 80.3 °C. (AH…
A:
Q: Draw the major elimination and substitution products formed in this reaction. Use a dash or wedge…
A: Substitution reaction is an organic reaction where an incoming nucleophile replaces the functional…
Q: Perform the following calculations, reporting the answers with the correct number of significant…
A: The question demands doing a mathematical calculation and stating the result in exponential format…
Q: You have 50.0mL of 0.514M NH3. Kb = 1.8x10-5 You add 15.0mL of a 0.05M HCl solution. What is the pH…
A: The pH of the final solution after adding HCl is approximately 5.13.Explanation:First, let's…
Q: Write the molecular formula of acetic acid. The color codes for the ball-and-stick model shown are:…
A: Here, in the structure, black color shows carbon, blue red shows oxygen, and white shows hydrogen…
Q: w the Hofmann product of the dehydration of this alcohol. ☐ ☐ : + P OH POCI 3 Click and drag to…
A: The objective of the question is to find the Hoffman product of the dehydration of the given…
Q: Consider the titration of a 30.0 mL sample of 0.126 M CO32- so HI. Determine each quantity. Kb = 2.1…
A: The objective of this question is to determine the concentration of a specific reactant in a…
Q: Which of the following compounds is THE MOST acidic? Select one: О а. NH3 ○ b. -NH2 ○ C. NH4+ ○ d. +…
A: Those compounds are called acids who can lose a proton.We also know that any acid when lose proton…
Q: A diprotic acid, H2A, has acid dissociation constants of Kal = 3.78 x 104 and Ka2 = 4.21 x 10-11.…
A: A diprotic acid can dissociate to produce two H+ ions.
Q: 20. How many different products are possible for the following reaction? Consider both regio- and…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Predict the products and stereochemistry for the following reduction reaction NaBH4 (a) EtOH H2…
A: Sodium borohydride is a reducing agent that reduces carbonyl compounds to corresponding…
Q: Identify the configuration of each chiral center in the following compound: m2 8 In the boxes below,…
A: Chiral centers, also known as stereocenters or chiral atoms, are a fundamental concept in…
Q: Calculate the pH of a 0.345 M aqueous solution of acetylsalicylic acid (aspirin, HC9H7O4) and the…
A: The answers were determined to be:pH=1.99[HC9H7O4]=0.335 M[C9H7O4-]=0.010 MExplanation:Step 1:…
Q: Consider the following data on some weak acids and weak bases: base acid K Kb name formula name…
A:
Q: A nucleophile donates a pair of electrons to form a new bond in a reaction. A nucleophile is also…
A: For a molecule to act as nucleophile, it must have either a lone pair or negative charge.
Q: Rank in order of Increasing Bronsted acidity the structures I, II and III.
A: According to Bronsted a acid is that which donate the proton (H+) in the solution.So in the given…
Q: Draw the major product for each of the following reactions: Incorrect. O: [H+] NH3 -H₂O CH3 ? Edit…
A: The given reaction is a reaction between a ketone and an ammonia in acidic medium. In this reaction,…
Q: 27 MÁ 27 Mr Dy + 8H² + 5 e¯ + Fe² = Mo² + 40+ Fe²" te
A: is given unbalanced reaction.Separating the reaction as: Oxidation half reaction: (loss of…
Q: what is the iupac name for this compound and where are the two chiral centers?
A:
Q: A 15.0 mL sample of 0.20 M of an unknown weak acid is titrated with 0.15 M NaOH at 25 oC. At 25 oC,…
A: Molarity of NaOH = 0.15 mol/LMolarity of weak acid = 0.20 mol/LpKa = 4.06
Q: raw the major products of the elimination reaction below. elimination would not occur at a…
A: The objective of the question is to predict the products formed in the following reaction given.
Q: Balance the reaction below in acid by selecting the correct coefficient for each component.Cr2O72− +…
A: Cr2O7²⁻ + 3HNO₂ + 5H⁺ → 2Cr³⁺ + 3NO₃⁻ + 4H₂OExplanation: Here's how we achieved the balanced…
Q: Draw a stable, uncharged resonance form for the structure shown. Be sure to include lone pairs of…
A: Resonance structure: These are the set of Lewis structures that describe the electronic bonding of…
Q: Draw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form…
A: The objective of the question is to predict the diene and dienophile for the given molecule.
Q: Show how you would make the TM from the given SM. HO HOC "COH
A: In the first step we have to reduce the keto group into alcohol group by the use of LiAlH4 /mild…
Q: Modify the given copy of the starting material to draw the major product(s). Use the single bond…
A: An electrocyclic reaction is a reaction that is either the ring opening of cycloalkane or the ring…
Q: What term best describes the relationship between the structures below? H, CI H₂N Enantiomers…
A: We have to choose the correct option.
Q: 8.-22. Match the following reaction sequences, all starting with benzene, to a product below. Steps…
A: All of the given reactions are electrophilic aromatic substitution reactions.
Q: Don't use hand raiting please
A: The objective of the question is to draw the Fischer projection of the reactant which on reducing…
Q: The illustration to the left represents a mixture of oxygen (red) and fluorine (green) molecules. If…
A:
Q: 1) Provide the reagent(s) and the reaction mechanism of the following reaction. H₂N
A: Benzene is converted into nitrobenzene.Nitro group is deactivating and meta-directing.Friedel Crafts…
Q: Show three or four different drawings that show three or four different relevant resonance…
A: Resonance structures are the different structures that represent the delocalization of electrons in…
Q: B. Which is the stronger Bronsted acid, A or B. Give a BRIEF explanation and support your assignment…
A: Any species that is capable of donating a proton is called Bronsted acid.A-H ----> A- +…
Q: Q24. The rate constants k₁ and k₂ for two different reactions are 1016 × e-2000/T and 1015 ×…
A:
Q: There was a titration between 0.15 M NaOH with a monobasic acid HCI (10.0 mL). Phenolphthalein was…
A: Volume of NaOH(V1) = 18.5 mLMolarity of NaOH(M1) = 0.15 MVolume of HCl(V2) = 10.0 mL
Q: We determined that the structure of BF3 is an exception to the octet rule (see above). 4. a)…
A: The formal charge on an atom in a molecule can be determined using the following equationThe valence…
Q: Calculate the total mass of the acids produced by the reaction of excess water with 20.83 g of…
A: Given that, phosphorus pentachloride reacts with water to form hydrochloric acid and phosphoric…
Q: Predict the equilibrium concentration of O2 in the reaction described below (for which Kc = 4.10 x…
A: The given reaction is:N2(g) + O2(g) 2NO(g)Kc = Volume of container = 1.0 LMoles of N2 = 0.20…
Q: Draw structural formulas for the a,ẞ-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: According to the following reaction, how many grams of phosphoric acid will be formed upon the…
A: Given reaction,Mass of water reacted= Molar mass of Mass of reacted=?Molar mass of
Q: HO. Na2Cr2O7 H2SO4, H₂O SOCI₂ OH H3O+ 1. LIAI(OMe)3H 2. H3O+ excess NH3
A: The reaction scheme is shown below.We have to provide the missing products and reagents.
Q: What term best describes the Tation below? COOH H3C-CH COOH соон HC-CH3 OH HO
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: 3. For the following transformations draw the product in the box below. Draw only one stereoisomer…
A: LDA is a strong base which also sterically bulky. Thus it accepts protons from less congested…
Q: What is the molar solubility of lead(II) fluoride, PbF2, whose Ksp is 3.2 × 10–8 in water?
A: Given that,The Ksp for The molar solubility of
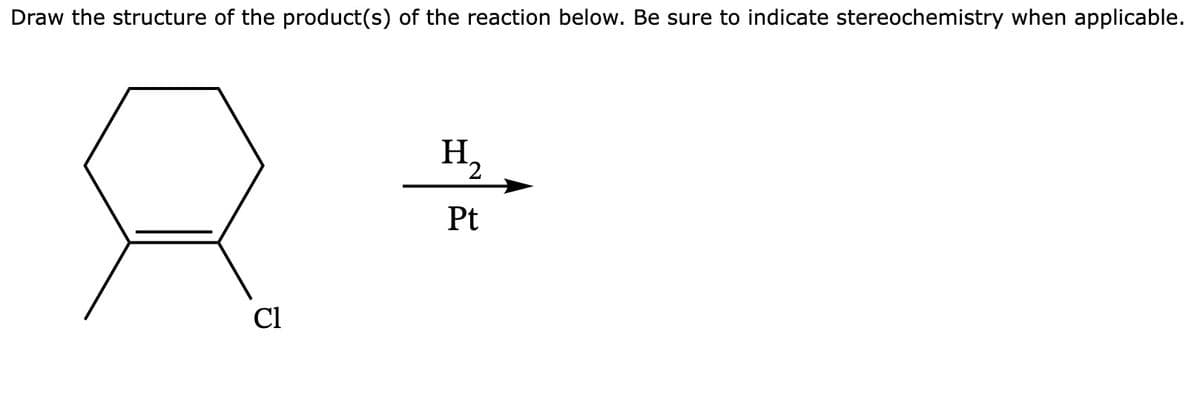
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- Complete the following reactions. Show all possible products and indicate stereochemistry where appropriate.Draw the product of an SN2 reaction shown below. Use wedge and dash bonds to indicate stereochemistry where appropriate. Indicate inorganic byproducts.Give the product/s for the following reaction and indicate what mechanism is involved in the formation of such product/s as SN1, SN2, E1, E2. CH3CH=CHCl + NaNH2
- Draw the product of an SN2 reaction shown below. Use wedge and dash bonds to indicate stereochemistry where appropriate. Ignore inorganic byproducts.Explain the following result. Although alkenes are generally more reactive than alkynes toward electrophiles, the reaction of Cl2 with but-2-yne can be stopped after one equivalent of Cl2 has been added.Complete these reactions, showing the stereochemistry of the product
- The reaction of tert-butyl cyclopentyl ether with trifluoroacetic acid is shown below. Explain what kind of reaction this is (SN1, SN2, E1, E2...) and show the mechanism for this transformation.Draw the products of the SN1 reaction below and use wedge-and-dash bonds to indicate the stereochemistry of any stereogenic centers.Draw the product of an SN2 reaction shown below. Use the dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers. Ignore inorganic byproducts.
- Give the following reaction A+3B --->2C.Can you assist me in identifying the error in my drawing? Is the stereochemistry correct? Any feedback will be greatly appreciated. Thank you. Question: Draw the structure of the product that is formed when the compound shown below is treated with cold, dilute KMnO4, HO-. Be sure to indicate stereochemistry if required.Please state the name of the reaction for the following and show complete arrowing pushing mechanisms to find Product 2.

