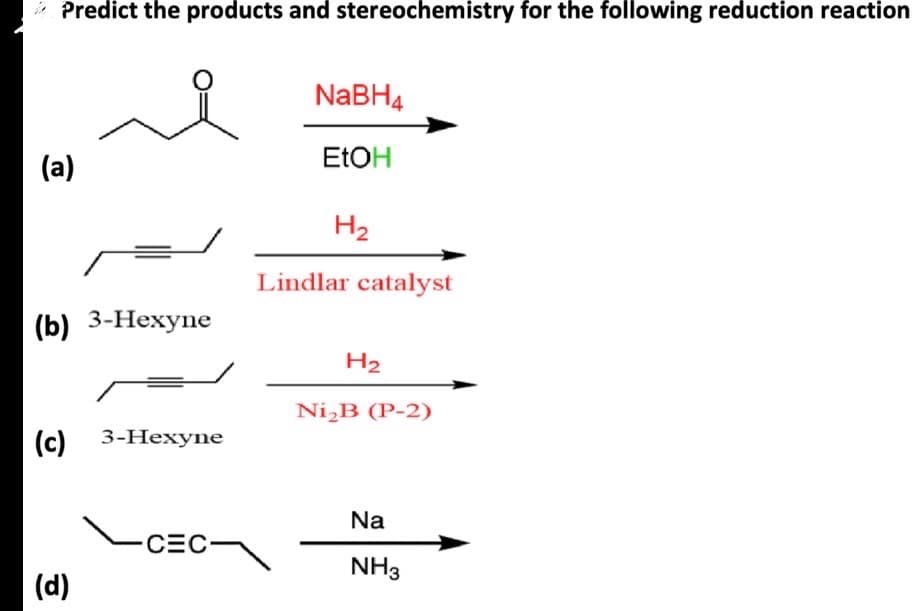
Predict the products and stereochemistry for the following reduction reaction NaBH4 (a) EtOH H2 Lindlar catalyst (b) 3-Hexyne H2 Ni₂B (P-2) (c) 3-Hexyne Na CEC- NH3 (d)
Q: how to rank F2, F2+, F2- from highest to lowest bond energy
A: We have to rank the species from highest to lowest bond energy.
Q: Rank in order of Increasing Bronsted acidity the structures I, II and III.
A: According to Bronsted a acid is that which donate the proton (H+) in the solution.So in the given…
Q: Which one of the following pairs of reagents would NOT give the product shown in the following…
A: The given reaction scheme and options are shown below.We have to select which one pair does not give…
Q: Draw the major product o
A: Given is organic reaction. The name of given reaction is hydrobromination reaction. The given…
Q: Draw the structure of the major E2 product.
A: Given is E2 reaction.E2 stands for biomolecular elimination reaction.It is single step reaction.In…
Q: A buffer is prepared by mixing 48.2 mL of 0.196 M NaOH with 144.3 mL of 0.231 M acetic acid. What…
A: The objective of this question is to calculate the pH of a buffer solution after the addition of a…
Q: Add formal charges to each resonance form of NCO¯. structure A Select Draw Templates More G : N C N…
A: Given ,Compound : Objective : Add formal charges on each atom in each resonance structure.
Q: Consider a 0.586 M aqueous solution of barium hydroxide, Ba(OH)2Ba(OH)2 (aq). How many grams of…
A: The objective of the question is to calculate the amount of Ba(OH)2 in grams, the number of…
Q: 2.3. Step by step retrosynthetic analys the starting material shown on the right. D desired…
A: The objective of this question is to draw the step-by-step retrosynthetic analysis of each of the…
Q: HO H2SO4
A: The reactant is an alcohol. It is an organic compound. The reagent is sulfuric acid . It is used to…
Q: Use the References to access important values if needed for this question. According to the…
A: 388.2 Explanation:The balanced chemical equation for the reaction is:H2(g)+C2H4(g)→C2H6(g) From…
Q: the oxidat number for sui each of the following (b) sulfite ion, SO3²-
A:
Q: Determine if the solution formed by each salt is pH acidic, basic, or neutral. KCIO4 Na2CO3 1.…
A: Answer:-This question is answered by using the simple concept of determination of nature of salt…
Q: A solution contains 6.29×103 M potassium sulfate and 1.10×102 M ammonium phosphate. Solid barium…
A: The question is based on the concept of solubility product principle.It states that when a weak…
Q: A scientist prepared a 0.034 M NH4Cl salt solution by dissolving the solid salt in water. Calculate…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Help pls #9
A: Below complete reaction is Given.Explanation:
Q: Q1: Device a synthesis of the following compound from cyclopentanone, benzene, and organic alcohols…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: (i) COOH (h) (1) 2 CH3CH₂Li (2) H3O+ H H3O+ ↑ C=PPh3 + 요. CH3 NILL H+, heat
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Aromatic Substitution Reactions 1. Predict the major product(s) of the following reactions: CI…
A: Note: “Since you have posted a question with multiple sub parts, we will provide the solution only…
Q: At 25°C, what is the hydronium ion concentration in a 4.8 x 10-2 M KOH solution? 4.8 x 10-2 M 2.1 x…
A: At 25°C, the product of concentrations of hydroxide and hydronium ion will be equal to the…
Q: You are working in a laboratory that makes frequent use of a scanning tunneling microscope (STM) to…
A:
Q: Which alkene below has the most stable carbon-carbon double bond?
A: The stability of alkene depends on the number of alkyl groups attached to the carbon atom of the…
Q: Provide the major product for the following reaction? 0 H₁ OH
A:
Q: Add any remaining curved arrow(s) to draw step 2 of the mechanism. Modify the given drawing of the…
A: The objective of the question is to predict the product formed in the following reaction given.
Q: Determine the overall orders of the reactions
A: Overall order of a reaction is the sum of the powers raised to all the concentration terms in the…
Q: Write a mechanism for the following reaction: HNO3 H2SO4 11+ Show all resonance contributors to any…
A: In halobenzenes, halogen substituents withdraw electrons from the ring via their -I effect and…
Q: You have an aliquot of 10 mL of 0.1097 mol/L phosphoric acid. The ka values for phosphoric acid are…
A: Volume of phosphoric acid = 10 mLMolarity of phosphoric acid sample = 0.1097 mol/LVolume of NaOH…
Q: A mixture of gaseous reactants is put into a cylinder, where a chemical reaction turns them into…
A: The objective of the question is to determine the nature of the reaction (exothermic or…
Q: Arrange the following compounds by their increasing order of reactivity with proper justification…
A: The reactivity of ketones in nucleophilic addition reactions can be encouraged with the aid of…
Q: The three mechanisms for aromatic substitution differ in the intermediate, the leaving group, and…
A: The objective of the question is to verify the truthfulness of the statement that the three…
Q: 1. For the following reaction, draw the structure of the major product and the mechanism by which it…
A: Alcohols when treated with phosphoric acid undergoes dehydration forming alkenes.Mechanism:Step-1:…
Q: A 260.0-mL buffer solution initially contains 2.5x102 M of HCHO2 and 2.5x10-2 M of NaCHO2 4 Part A…
A: Information about the question
Q: We have a weak aid with a Kg of 3.7. 10. The pH was found to be 3.66. What was the approximate…
A: equilibrium constant (Ka) = 3.7 10-7pH = 3.66 pH = -log[H+]
Q: To test for a cation, you add sodium bromide to an unknown solution and a pale-yellow precipitate…
A: The formation of a pale-yellow precipitate upon the addition of sodium bromide (NaBr) suggests the…
Q: Give detailed Solution with explanation needed of each options...don't give Handwritten answer...
A: reagent used is HgSO4 and H2SO4Explanation:hydration of alkynne prepares the two carbonyl compuds…
Q: 2. Predict the products for the following reduction reaction by LiAlH4 and NaBH4 and explain why the…
A: NaBH4 is a weak reducing agent, it reduces aldehyde and ketone to alcohol.LiAlH4 is a strong…
Q: Arrange these substances in order of increasing boiling point: H2 H₂O LICI H₂S OH₂<H₂O<LiCl < H₂S…
A: The objective of the question is to arrange the given substances H2, H2O, LiCl, H2S in order of…
Q: 5. What is the expected major product for the following reaction? 1. 03 2. Zn, CH3COOH
A: One of the important reactions of alkene is ozonolysis. In the ozonolysis reaction, the…
Q: You have 50.0mL of 0.135M HCl. You add 10.0mL of 0.25 M NaOH What is the pH of the solution now?
A: The objective of this question is to calculate the pH of a solution after the addition of a base…
Q: Calculate the concentration of the hydroxide ion in a solution that contains 5.5 × 10- 5 M of…
A: [H3O+] = 5.510-5 MKw = 1.010-14
Q: When a loan pre-approval is provided to a buyer, what does it mean? The borrower qualifies for a…
A: The objective of the question is to understand the meaning of loan pre-approval in the context of a…
Q: Give the structure of the major product(s) for each of the following reactions. If no final product…
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Use the References to access important values if needed for this question. According to the…
A: Given:carbon (graphite) (s) + oxygen (g) carbon dioxide (g)Moles of carbon dioxide, CO2 formed =…
Q: PCI3 atomic metallic solid ionic solid molecular solid atomic nonmetallic solid
A: The objective of the question is to classify the type of solid that PCl3 (Phosphorus trichloride)…
Q: Select the more electronegative element in this pair. Select the more electronegative element in…
A: The objective of this question is to identify the more electronegative element in each of the given…
Q: Determine whether the following compound is optically active or optically inactive: Ooptically…
A: Optical activity arises from the lack of a center of symmetry in a molecule, which means it cannot…
Q: Question 2. The cycloheptatrienyl cation was first prepared more than a century ago by reaction of…
A: The objective of the question is to draw the molecular orbital diagram for the cycloheptatrienyl…
Q: what's the number-average molecular weight for a linear polytetrafluoroethylene of total length…
A:
Q: To 0.340 L of 0.150 M NH3 is added 0.150 L of 0.120 M MgCl₂ ▾ Part A How many grams of (NH4)2SO4…
A: Firstly, calculate the moles of every compound used.n(NH3)= (0.150 mol/L )(0 .340 L) = 0.051 moles…
Q: 3. a. E,R is? b. E,S c. S,Z 4. What is the л-energy diagram of the compound in question 3? b. d. R,Z…
A: The objective of the question is to find the configuration along the double bonds of the given…
Step by step
Solved in 3 steps with 1 images

- Write the mechanism of the hydrolysis of cis-5, 6-epoxydecane by reaction with aqueous acid. What is the stereochemistry of the product, assuming normal backside SN2 attack?When 3-methyl-1-butene is reacted with 9-borabicyclo[3.3.1]nonane, the "1-ol" product is formed. What is the detailed reactin scheme for the transformation? Describe the purification procedure.Provide the reagents necessary to convert 3-methyl-2-butanol to 2-bromo-3-methylbutane. CHOOSE FROM THE FOLLOWING REAGRENTS BELOW conc. HBr NaBr, H2SO4 PBr3 HBr, peroxide Br2
- Predict the products of the following reaction. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below(ii) The elimination reaction between 2-bromobutane and NaOCH2CH3 gives two organic products. Draw a mechanism for the reaction which produces the major organic elimination product and provide a rationale as to why that is the major product.
- (10pts) Compound A, C10H16, was found to be optically active. On catalytic reduction over a palladium catalyst, 2 equivalents of hydrogen were absorbed, yielding compound B, CioH2o. On ozonolysis of A, two fragments were obtained. One fragment was identified as acetic acid (CHCOOH). The other fragment, compound C, was an optically active carboxylic acid, C8H14O2. Write reactions, and draw the correct structures for A-C, explain your answer in detail.I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Predict (by name) the major product for the following reaction when NBS/peroxide is added. The starting compound is byblo[4.4.0]dec-1-ene. Note the numbering and name the product.
- Predict the products of the following reactions. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.Predict the major products of the following reactions, including stereochemistry whereappropriate.(a) 2,2-dimethyloxirane + H+ >H218O (oxygen-labeled water)LDA/THF, 78°Clongrightarrow Predict the major product of the following a- halogenation/a - alkylation reactions under the given reaction conditions.

