Draw the structure(s) of the major organic product(s) of the following reaction. + 1. in benzene 2. aqueous HCI • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. Do not include the amine from which the enamine was derived. • • If no reaction occurs, draw the organic starting material. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate multiple products using the + sign from the drop-down menu. ? n ChemDoodle Ⓡ < >
Draw the structure(s) of the major organic product(s) of the following reaction. + 1. in benzene 2. aqueous HCI • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. Do not include the amine from which the enamine was derived. • • If no reaction occurs, draw the organic starting material. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate multiple products using the + sign from the drop-down menu. ? n ChemDoodle Ⓡ < >
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter20: Acidity And Pka Of Phenols
Section: Chapter Questions
Problem 11E
Related questions
Question
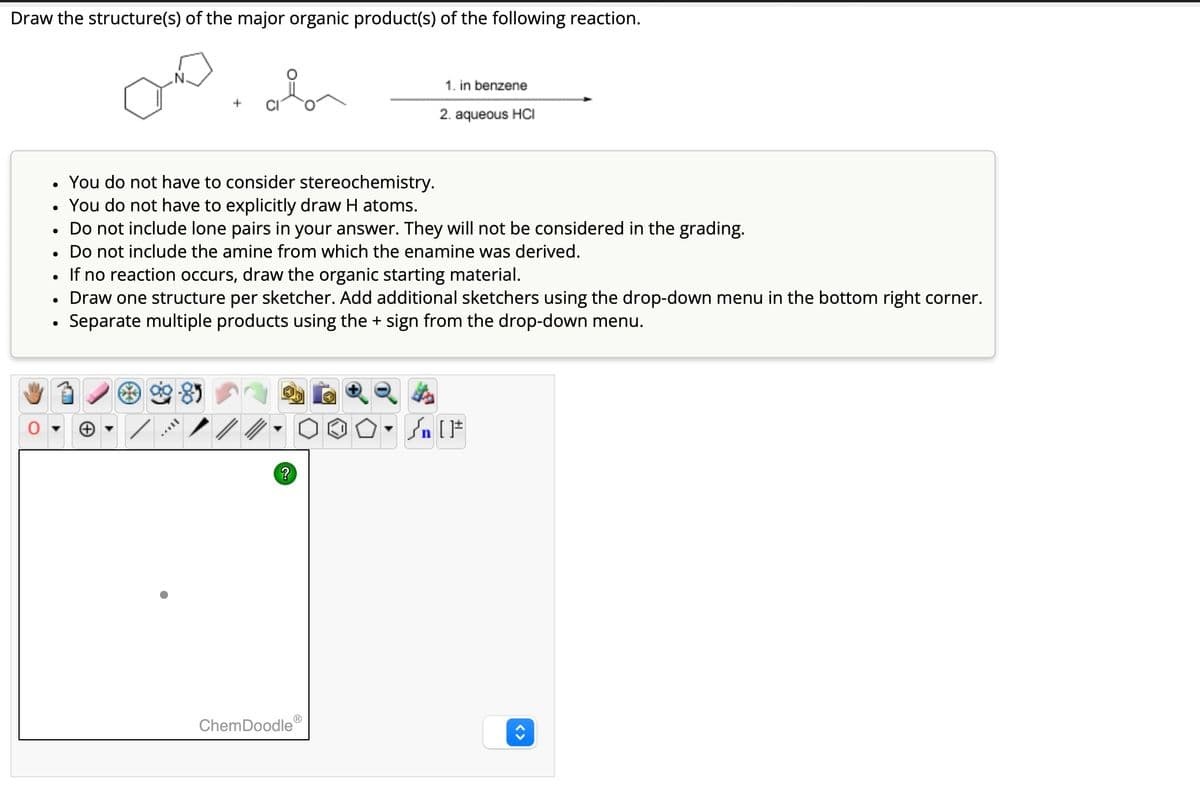
Transcribed Image Text:Draw the structure(s) of the major organic product(s) of the following reaction.
+
1. in benzene
2. aqueous HCI
•
You do not have to consider stereochemistry.
•
You do not have to explicitly draw H atoms.
•
Do not include lone pairs in your answer. They will not be considered in the grading.
Do not include the amine from which the enamine was derived.
•
• If no reaction occurs, draw the organic starting material.
•
Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
•
Separate multiple products using the + sign from the drop-down menu.
?
n
ChemDoodle
Ⓡ
< >
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning