Q: A 9.00 L tank at 5.93 °C is filled with 7.54 g of boron trifluoride gas and 17.1 g of dinitrogen…
A: Partial pressure = total pressure x mole fractions of the gas. Using this formula , calculate the…
Q: Write a balanced nuclear equation for the following: The nuclide nitrogen-13 undergoes positron…
A: Positron emission of nitrogen-13 is produce carbon-13.
Q: question. A helium-filled weather balloon has a volume of 861 L at 19.9°C and 751 mmHg. It is…
A: According to ideal gas equation, (P1V1 ) /T1 = (P2V2)/T2
Q: What is wrong with each name 2-bromobenzene. 3,3-dichlorotoluene. 1,4-dimethylnitrobenzene
A: Toluene is benzene attached with one -CH3 group. In nitrobenzene -NO2 group is attached to the…
Q: Write a balanced nuclear equation for the following: The nuclide carbon-14 undergoes beta emission.
A:
Q: ) H;(g) + CO;(g) 27 H,0(g) + CO(g)
A:
Q: Calculate the pH of a solution that is 0.0736 M KC2H3O2 at 25°C.
A:
Q: The following data were obtained for the reaction of NO with O2. Concentrations are in molecules/cm3…
A: To determine rate law , we would first write a general rate law expression. Then we plug the values…
Q: OH (S) (S) (R) (R) OH OH OH OH (S) (R) (S) (R) OH OH
A:
Q: Which of the following solids would be expected to display the highest melting point? A) NaCl B)…
A: Here we have to find the solid that will expected to display the highest melting point. By…
Q: It refers to how a circumstance can occur without the help of any external factors? a. Enthalpy O b.…
A:
Q: Decane (C10H22) is the hydrocarbon combusted in diesel fuel engines, the equation is shown below.…
A: Given-> 2C10H22 + 31O2---> 20CO2 +22H2O Weight of CO2 = 30.0 gm
Q: 22-11 Complete the following reaction sequence from the synthesis of otteliones A and B (Org. Lett.…
A: These are the examples of ketal deprotection, esterification and conversion of vic-diol to alkene.
Q: The Ksp for BaCO3 is 5.1×10−9.5.1×10−9. How many grams of BaCO3 will dissolve in 1000. mL of…
A: Answer: 1000mL is equal to 1 Liter, therefore first of all we will find out the solubility in mol/L…
Q: 1. If 22.5 L nitrogen at 748 mm Hg are compressed to 725 mm Hg at constant temperature. What is the…
A:
Q: Chemistry write General formula, suffix/prefix , example Structure, example Name, distinguishing…
A:
Q: The pH of 0.20 M HN03 is O a. 1.58 Ob. 0.70 C. 0.20 Od. 0.63
A: Using formula in these questions: PH = -log[H+] Where as [H+]-------> concentration of H+ Using…
Q: It refers to a condition of a chemical reaction by which a forward reaction occurs simultaneously…
A:
Q: Nitrogen molecules (N2) are moving all around you right now at room temperature (~300 K). Take the…
A:
Q: The pH of 0.20 M HNO3 is O a. 0.63 O b. 1.58 O C. 0.70 O d. 0.20
A: pH = - log [H+ ] Strong acids undergo complete ionisation in solution. Thus, [H+] =…
Q: 1. Synthesize the following alkene through the Wittig reaction of a carbonyl compound and a Wittig…
A: 1
Q: 6) Identify the reagents needed for steps a-d in the following scheme: OH a b HO OH OCH3
A: Identify the reagents for the given reaction
Q: Solid potassium sulfate is slowly added to 150 mL of a 0.388 M calcium acetate solution until the…
A:
Q: c) 4 HCI(g) + O2(g) D2 2 CI,(g) + 2 H,0(g) (6)*0
A:
Q: Show the saponification of a liquid soap.
A: The explanation is given below-
Q: Cyclohexanone is oxidized to adipic acid with the following equation. Twenty gram (20g) of…
A: Actual yield = 15 g Theoretical yield = 25 g
Q: Hydrogen was collected over water at 21.4°C on a day when the atmospheric pressure was 741. Torr.…
A: Answer: This question is based on ideal gas equation which is shown below: PV=nRTPVRT=nn=PVRT…
Q: For any reaction at equilibrium, which of the following is TRUE? AS=0 O b. AG=0 AH<0 O d. AS<0
A: The equilibrium indicated by ∆G = 0 is known to be the equilibrium of spontaneity. This implies that…
Q: Let’s say you have a 75% sugar solution… You want: 100mls of 35% solution. How much stock, how…
A: Given :- Concentration of stock solution = 75% Concentration of dilute solution = 35% Volume of…
Q: The Ksp for an ionic solid, AB2, is 2.821*108. The molar mass of this ionic solid is 99.59 g/mol.…
A:
Q: Write a balanced nuclear equation for the following: The nuclide nitrogen-13 decays to carbon-13 by…
A: We have to predict the balance nuclear equation.
Q: 3. (6)What is the pH of a buffer that is 0.12M lactic acid (HC3H5O3) and 0.1OM sodium lactate? Ka=…
A:
Q: Hydrogen was collected over water at 21.4°C on a day when the atmospheric pressure was 741. Torr.…
A:
Q: aw. No electronic device allowed. Write the dissociation reactions for the solution made by adding…
A:
Q: Me но CF3 Me D A в ÇF3 Me Me. Me „CF3 Me F;C Me Me Me Ме- "Me Me Me Me `Me Me O:
A: This is an example of addition reaction of an alkene. A carbocation intermediate is formed by…
Q: chatelier's principle S₈(g) + 120₂(g) ↔ 8 SO₃(g) + 808kcals what will happen when... (A) Sulfur…
A: Given- S8(g)+12O2(g)<->8SO3(g)+808 kcals
Q: (Q19) What is the pH of a solution that is 0.768 in hydrosulfuric acid (H2S)? Ka1 = 8.9 x 10-8 and…
A:
Q: Which of the following measures the molecular randomness of a reaction? a. Enthalpy O b. Entropy O…
A: Here some physical parameters are given.
Q: Which element is most likely to form three covalent bonds? Group of answer choices S N O C Si
A: Element which form three covalent bond must have atleast three valence electron.
Q: CALCULATE THE Concentration iron(III) thiocyanate ion [FeSCN2+] (M )
A: Insufficient information but I know the all data so I tried to solve it. If you have different data…
Q: Balance each redox reaction in acid solution using the half reaction method. II. 5. H.О2 + Crz0, 2-…
A: To balance given redox reactions , we would first determine oxidation and reduction half reactions.…
Q: Draw the resonance structures of methoxy benzere. what is the kyhrid? resonance
A:
Q: A gas occupies a volume of 323 mL at a pressure of 505 torr. To what pressure must the gas be…
A:
Q: Which of the following statements is true? Question 19 options: A) When two opposing…
A: -> At equilibrium Rate of forward reaction = Rate of backward reaction Hence, option (1) is…
Q: When the nuclide boron-8 undergoes positron emission: The name of the product nuclide is The symbol…
A: Given, When boron-8 nuclide undergoes positron decay the formed nuclide is:
Q: 2. (4) Which of the following would not make a good buffer system? a) HCI/Cl- b) NH3/NH, с) Н-СОНСО;…
A:
Q: what intermolecular forces are present in quaternary ammonium salts? are there any chiral centers…
A:
Q: Give clear handwritten solution) give reaction mechanism and major product for the all three…
A: The Given question is various types Of bromination Reaction.
Q: AsO,3 + I2 → AsO,3 + I (basic) Cr + CIO, → CrO2 + CIO;- (basic)
A: Balanced the given redox reaction under basic condition---
Q: Substitutions on aromatic rings are generally carried out by electrophilic substitution, but for the…
A: A question based on aromatic compounds that is to be accomplished.
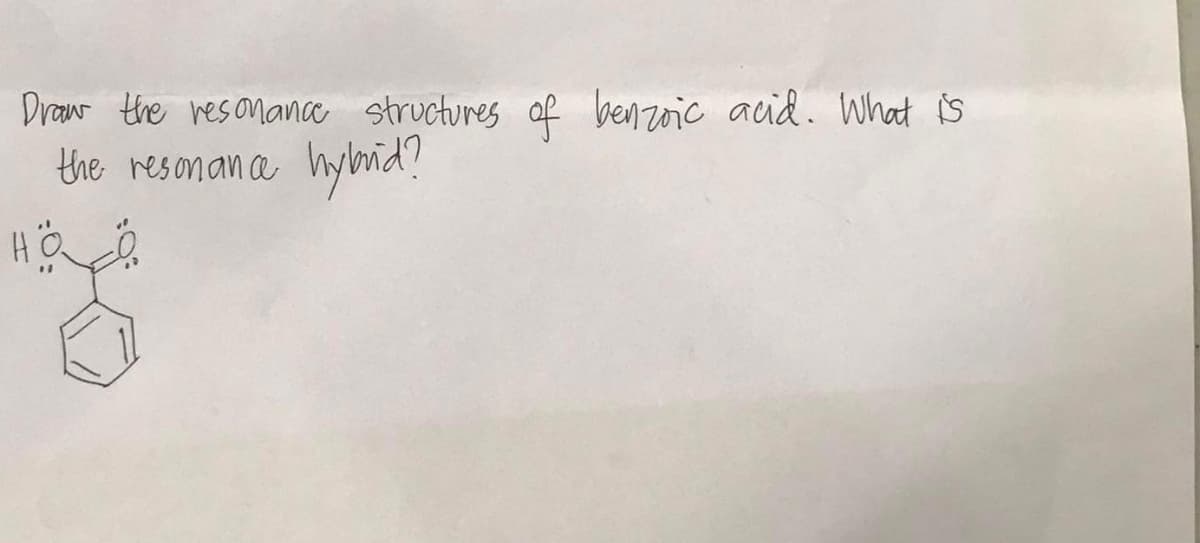
Step by step
Solved in 2 steps with 2 images

- Hydrocarbon A possesses a significant dipole, even though it iscomposed of only C—C and C—H bonds. Explain why the dipole arisesand use resonance structures to illustrate the direction of the dipole.Which ring is more electron rich?draw the resonance for the following structures?draw the resonance structure of benzaldehyde
- please determine resonance structure of acetic acid and fluroacetic acid!Draw one aromatic resonance form of the structure shown below.Draw thee other resonance structures for the following carbocation. Use electronpushing arrows in order to derive one resonance structure from another. Indicate theformal charge on each atom in your structures.
- Draw the Resonance structures of m-nitrophenol with explanationResveratrol is an antioxidant found in the skin of red grapes. Itsanticancer, anti-inflammatory, and various cardiovascular effects areunder active investigation. (a) Draw all resonance structures for theradical that results from homolysis of the OH bond shown in red. (b)Explain why homolysis of this OH bond is preferred to homolysis ofeither OH bond in the other benzene ring.Draw significant resonance structures for the following compound:

