Drinking a Soda 6 of 16 Constants I Periodic Table Before going in for an annual physical, a 70.0-kg person whose body temperature is 37.0°C consumes an entire 0.355-liter can of a soft drink (which is mostly water) at 12.0 °C. Part A What will be the person's body temperature Tinal after equilibrium is attained? Ignore any heating by the person's metabolism. The specific heat capacity of a human body is 3480 J/kg · K. • View Available Hint(s) ΥΠ ΑΣφ Tinal = °C Submit Part B Is the change in the person's body temperature great enough to be measured by a medical thermometer? (A high-quality medical thermometer can measure temperature changes as small as 0.1°C or less.) O yes O no
Drinking a Soda 6 of 16 Constants I Periodic Table Before going in for an annual physical, a 70.0-kg person whose body temperature is 37.0°C consumes an entire 0.355-liter can of a soft drink (which is mostly water) at 12.0 °C. Part A What will be the person's body temperature Tinal after equilibrium is attained? Ignore any heating by the person's metabolism. The specific heat capacity of a human body is 3480 J/kg · K. • View Available Hint(s) ΥΠ ΑΣφ Tinal = °C Submit Part B Is the change in the person's body temperature great enough to be measured by a medical thermometer? (A high-quality medical thermometer can measure temperature changes as small as 0.1°C or less.) O yes O no
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 63P: Heliox, a mixture of helium and oxygen, is sometimes given to hospital patients who have trouble...
Related questions
Question
Please help me with all the parts in this problem, and double check your answers as other tutors got it wrong
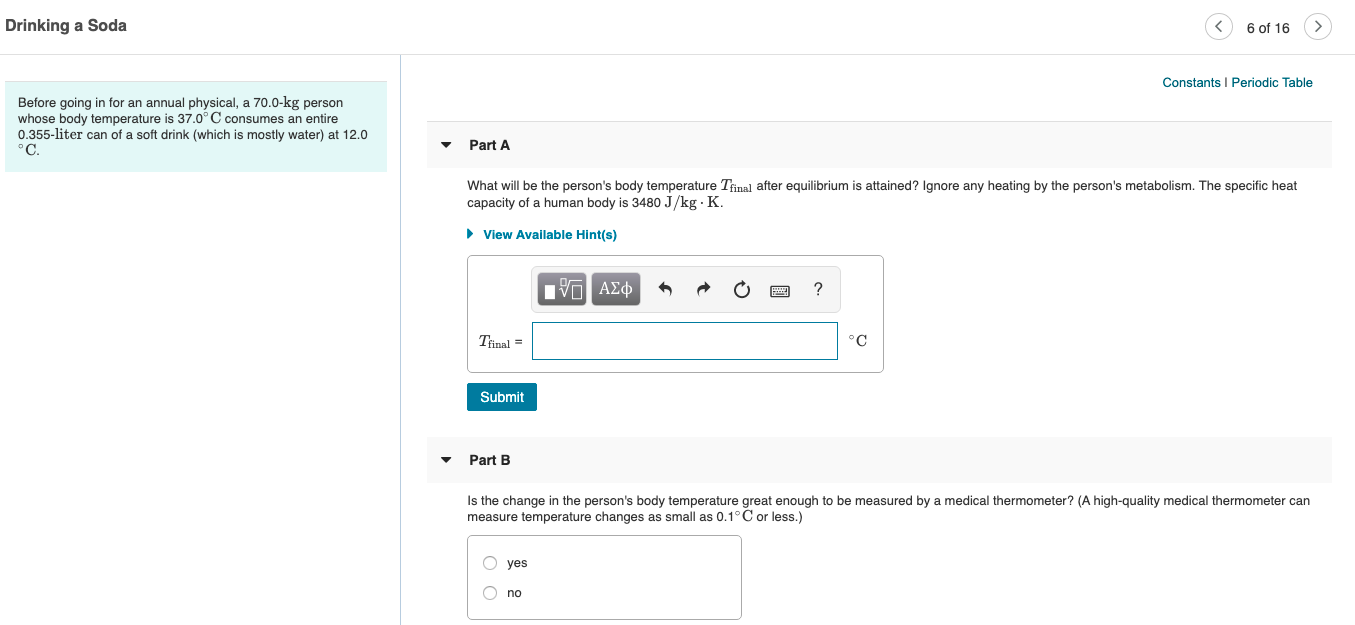
Transcribed Image Text:Drinking a Soda
6 of 16
Constants I Periodic Table
Before going in for an annual physical, a 70.0-kg person
whose body temperature is 37.0°C consumes an entire
0.355-liter can of a soft drink (which is mostly water) at 12.0
°C.
Part A
What will be the person's body temperature Tinal after equilibrium is attained? Ignore any heating by the person's metabolism. The specific heat
capacity of a human body is 3480 J/kg · K.
• View Available Hint(s)
ΥΠ ΑΣφ
Tinal =
°C
Submit
Part B
Is the change in the person's body temperature great enough to be measured by a medical thermometer? (A high-quality medical thermometer can
measure temperature changes as small as 0.1°C or less.)
O yes
O no
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Physics for Scientists and Engineers, Technology …
Physics
ISBN:
9781305116399
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning