Q: Consider a solution containing 5 moles of acetone and 5 moles chloroform. The vapor pressures of pur…
A: Pressure exerted by the vapour above the surface of liquid at equilibrium at a given temperature is…
Q: b. Document Sharing H2 ? Pd catalyst Draw the molecule on the canvas by choosing buttons from the…
A: Thank You.
Q: Provide the products of the following acid-base reactions: 요오 ii OH OH OMe e OH OH
A:
Q: If NaOH is combined with CH3OCH 3, which of the following reaction diagrams best represents the…
A: The question is asking us to determine the reaction diagram that best represents the reaction…
Q: Predict the product for the following reaction. To preview the image MeO. CHO heat + MeO MeO. ± OA…
A: Step 1: Step 2: It the the cyclo-addition reaction of pericyclic. Step 3: Step 4:
Q: Curve 3 best represents which species? Ne at 600 K Ne at 450 K Ar at 600 K
A: We need to find which curve is for which gas in the graph.
Q: An analytical chemist is titrating 156.3 mL of a 0.3700 M solution of ethylamine (C2H5NH2) with a…
A:
Q: Highlight each of the positions on the benzene ring that you expect will be most likely to react by…
A: The objective of the question is to identify the positions on a benzene ring that are most likely to…
Q: Add explaintion each step
A: Let's go through each step with explanations:Alanine (Ala, A): Alanine is a nonpolar amino acid,…
Q: Can you please include the species!
A: Step 1: Step 2: Step 3: Step 4:
Q: A 25.0 mL sample of 0.450 M (CH3)2NH is titrated with HBr. What is the pH of the solution after 30.0…
A: Ans
Q: Please show the synthesis for the following compound C, from any alkane (compound A). Compound C IR…
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: A certain liquid has a vapor pressure of 92.0 Torr at 23.0 °C and 211.0 Torr at 45.0 °C. Calculate…
A: Temperature is the only factor that affects vapor pressure and they are directly proportional to…
Q: For each of the substrates below, identify whether: (A) the rate of substitution doesn't depend on…
A: A question based on reaction mechanisms. For the given 4 reactants, the most appropriate option is…
Q: A chemistry graduate student is given 100. mL of a 1.70M hydrocyanic acid (HCN) solution.…
A: The volume of the solution = Concentration of HCN = of =
Q: None
A: Step 1: Basic discussion Step 2: Mechanism of the reaction Step 3:
Q: One of the following Diels-Alder reactions will occur more rapidly than the other. Draw the major,…
A: The Diels-Alder reaction is a cycloaddition reaction between a conjugated diene and a dienophile…
Q: Give proper explanation please. Hand written solution is not allowed.
A: Given: [HNO2]=0.150M;VHNO2=75.0mL;[NaOH]=1.00M;Ka=5.6x10−4Step 1: Write the acid-base…
Q: QUESTION 3 In order to induce precipitation of the salt, you need to add acid to the aqueous…
A: Answer:Salts are made during the reaction of acid and base where anion of acid combines with cation…
Q: 3) Predict the reactants used in the formation of the following compounds using Acid- Catalyzed…
A: Acid hydration of an alkene is a chemical reaction where an alkene reacts with an acid and water to…
Q: écules in the table below is stable, in the exact form in which it is drawn, at pH=11. If you decide…
A: The question's objective is to decide whether the given molecules are stable or not at the given pH…
Q: 1. For each of the following, show the predominant polymer structure that results. Include all end…
A: Step 1: Step 2: Step 3:Step 4:
Q: 65.0 mL of 0.575 M HNO, is titrated by 0.450 M KOH. Calculate the pH of the acid solution before any…
A:
Q: Question 1 (1 point) An electrochemical cell is set up with a Cu(s)|Cu(NO3)2(aq) electrode (E° red =…
A: The objective of the question is to identify the false statements about the operation of an…
Q: 4. Draw three possible hydrogen bonding interactions of isonicotinic acid (a) itself, (b) in water.
A: Given is organic compound. Hydrogen bonding is force of attraction between hydrogen covalently…
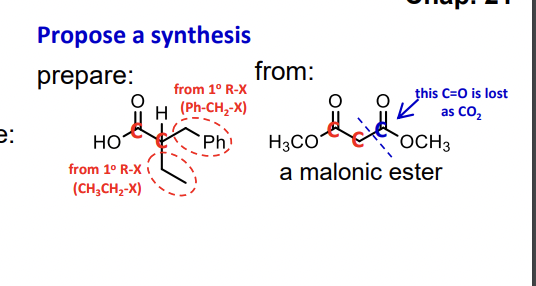
Q: 5. Provide a synthesis of the following molecule from the given starting materials. OMe + anything…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: 14. Consi 14. Consider the following pictures of gases at the same temperature. O = Ne and i. V. vi.…
A: The question is asking to rank the pictures of gases based on different parameters such as pressure,…
Q: None
A: Step 1:- Most soluble amine (i) because of shorter carbon chain length and hydrogen bond formation…
Q: 5 5. a. N-butynyl-N-butenylmethanamine C. b. N-butenyl-N-butynlmethanamine…
A: Step 1: identify the compound Step 2: write its iupac name Step 3: Step 4:
Q: None
A: Thank You.
Q: For the following reaction, the products are favored at equilibrium. Classify each of the reactants…
A: The objective of the question is to classify each of the reactants and products in the given…
Q: Zn(s) | Zn²⁺ (0.10 M) || Cu²⁺ (1.00 M) | Cu(s)? The standard cell potential for the reaction is…
A: Step 1:Step 2:
Q: 00000 0-3-3-NH + R' H₂O For the chemical reaction shown on the left, please select all the…
A: The statements that correctly describe the energy transformation occurring in the reaction are:…
Q: Current Attempt in Progress Identify the major and minor product(s) of the following reaction: Br x…
A: The objective of the question is to identify the major and minor products of the given reaction…
Q: Draw the major organic product for the reaction shown. H 1. NaBH4 2. H₂O Select Draw Templates More…
A:
Q: CH3CH2CH2CH2-Br Draw the major organic product(s) of the following reaction. 1 eq. NaNH2, NH3(1)…
A: The objective of the question is to predict the major organic product of the reaction involving…
Q: b) Draw the structure of each of the following complexes. Clearly show the stereocentre of central…
A: Structure of complex compound:A complex compound is a molecular structure with a central atom…
Q: A solution has [H+] = 4.2 x 103 M. The [OH] in this solution is O 4.2 x 10-11 M O 3.7 x 10-13 M O…
A: Answer:Product of hydrogen ion concentration and hydroxide ion concentration is always equal to the…
Q: A buffer solution contains 0.329 M NaH2PO4 and 0.324 M Na2HPO4. Determine the pH change when 0.080…
A: The chemical reactions involved are:1. H2PO4^- <-> H^+ + HPO4^2-2. HCl -> H^+ + Cl^-When…
Q: 11. Which of the following represents the integrated rate law for a first-order reaction? A. In =…
A: Thank You,
Q: Q5) What is the K,, for a silver salt, when the cell potential is 0.244 V versus a 0.5 M Ag'…
A:
Q: A 22.1 mL sample of 0.334 M methylamine, CH3NH2, is titrated with 0.276 M hydrochloric acid. At the…
A: The objective of this question is to calculate the pH at the midpoint of a titration between…
Q: i Provide the missing curved arrow notation. ס12 + од H C N O S F P C1 Br
A:
Q: I 3. All radioactive decay processes follow first-order kine - ics. What does this mean? What…
A: The statement that all radioactive decay processes follow first-order kinetics means that the rate…
Q: The hypothalamus produces hormones which are considered: Question 5 options:…
A: The objective of the question is to identify the correct description of the hormones produced by the…
Q: Question 28 Name using IUPAC nomenclature the starting material and the expected product below: Make…
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form…
A: Step 1: To solve this question, we need to draw the structural formulas for the diene and…
Q: 15.18 Determine the number and symmetry designations of the infrared-active C-O stretching modes in…
A: Infrared (IR) spectroscopy is useful for determining the stretching modes of chemical bonds,…
Q: Please step by step solutions
A: Ionic bonding is a type of chemical bond that forms between two atoms when one atom transfers…
Q: None
A: Ans part 1 Given Mass of UO2 = 2.26 kg= 2260 g Write the balanced equation UO2 (g) + 4 HF (g) →…
Step by step
Solved in 2 steps with 1 images

- The Darzens reaction invoIves a two-step, base-catalyzed condensation of ethyl chloroacetate with a ketone to yield an epoxy ester. The first step is a carbonyl condensation reaction, and the second step is an SN2 reaction. Write both steps, and show their mechanisms.The following reaction involves a hydrolysis followed by an intramolecular nucleophilic acyl substitution reaction. Write both steps, and show their mechanisms.When ethyl benzoate is heated in methanol containing a small amount of HCl, methyl benzoate is formed. Propose a mechanism for the reaction.
- Draw structures corresponding to the following names: (a) Phenyl benzoate (b) N-Ethyl-N-methylbutanamide (c) 2,4-Dimethylpentanoyl chloride (d) Methyl 1-methylcyclohexanecarboxylate (e) Ethyl 3-oxopentanoate (f) Methyl p-bromobenzenethioate (g) Formic propanoic anhydride (h) cis-2-Methylcyclopentanecarbonyl bromideIn the mixed Claisen reaction of cyclopentanone with ethyl formate, a much higher yield of the desired product is obtained by first mixing the two carbonyl components and then adding base, rather than by first mixing base with cyclopentanone and then adding ethyl formate. Explain.When 4-dimethylaminopyridine (DMAP) is added in catalytic amounts to acetic anhydride and an alcohol, it significantly increases the rate of ester formation. The process begins with a reaction between acetic anhydride and DMAP to form a highly reactive acetylpyridinium intermediate that is more reactive than acetic anhydride itself. Propose a mechanism for this process that includes the formation and reaction of the acetylpyridinium intermediate.
- Treatment of 2-cyclohexenone with HCN/KCN yields a saturated keto nitrile rather than an unsaturated cyanohydrin. Show the structure of the product, and propose a mechanism for the reaction.Show the products from reaction of p-bromoaniline with the following reagents: (a) CH3I (excess) (b) HCl (c) HNO2, H2SO4 (d) CH3COCl (e) CH3MgBr (f) CH3CH2Cl, AlCl3 (g) Product of (c) with CuCl, HCl (h) Product of (d) with CH3CH2Cl, AlCl3

