Q: For a compound below tell how many types of nonequivalent protons there are. C1 04 06 05 07
A: The non-equivalent proton is one that has a different environment and each proton produces a…
Q: This question concerns part 1 of this lab. You need to make a buffer solution by mixing both a weak…
A: Given,molarity of sodium acetate = 0.100 Mvolume of sodium acetate = 5.00 mLpH of the solution =…
Q: If 30 g of acetone was combusted with a stoichiometric equivalence of oxygen, how many grams of…
A: The objective of the question is to find out the amount of oxygen required to combust 30 g of…
Q: An a(1 - 4) glycosidic bond is found in which of the following? O all of these снон H Сон H H OH он…
A:
Q: Consider the titration of 100.0 mL of 0.200 M acetic acid (K = 1.8 x 10) by 0.100 M KOH. Calculate…
A: Given that 100 ml acetic acid of 0.200 M.Ka of acid = 1.8 × 10^-5by 0.100 M KOH.
Q: Calculate the pH of a solution of 0.0005 M sodium actate (NaAc) at 25 °C.
A: Step 1:To calculate the pH of a solution of sodium acetate (NaAc), we need to understand that sodium…
Q: Name the following compounds. Do not use italics or boldface. a) N-CH2CH2CH3 CH3 N. b) CH3
A: Step 1:Name of molecules are a) 1-propylpyrrolidineb ) N,N-dimethylanilineNaming process of these…
Q: Help solve 6.
A: Given: [Ca2+]=2.0x10−3M;massF−=1.0g;V=1.0x103L;MF−=19.00g/mol;Ksp=3.45x10−11The Ksp value is…
Q: For the following compound: OH Draw a mechanism for the tautomerization process under ACIDIC…
A: Tautomerism is a type of structural isomerism in which the there occurs proton migration from -OH to…
Q: Aldosterone secretion is regulated by: Question 7 options: A)…
A: The question is asking about the factors that regulate the secretion of Aldosterone, a hormone that…
Q: A chemist dissolves 293. mg of pure potassium hydroxide in enough water to make up 160. mL of…
A: Given that,Mass of pure potassium hydroxide,Molar mass of Volume of the solution,pH of the solution…
Q: The ESI mass spectrum of the protein transferrin gave a regular series of peaks, with the most…
A: Step 1: reason for regular series of peaks observed in the ESI mass spectrum Step 2:calculating the…
Q: 14 Homework Unanswered Due Apr 11th, 11:59 PM Select the correct retrosynthesis to obtain the…
A: To answer this query, permit's observe the stairs of retrosynthesis. 1. Identify the target…
Q: 3. Draw the structure of glycine at each of the following pH a. 5.97 I b. 13.0 c. 2.0 d. 11.0
A: Step 1: Step 2: Step 3: Step 4:
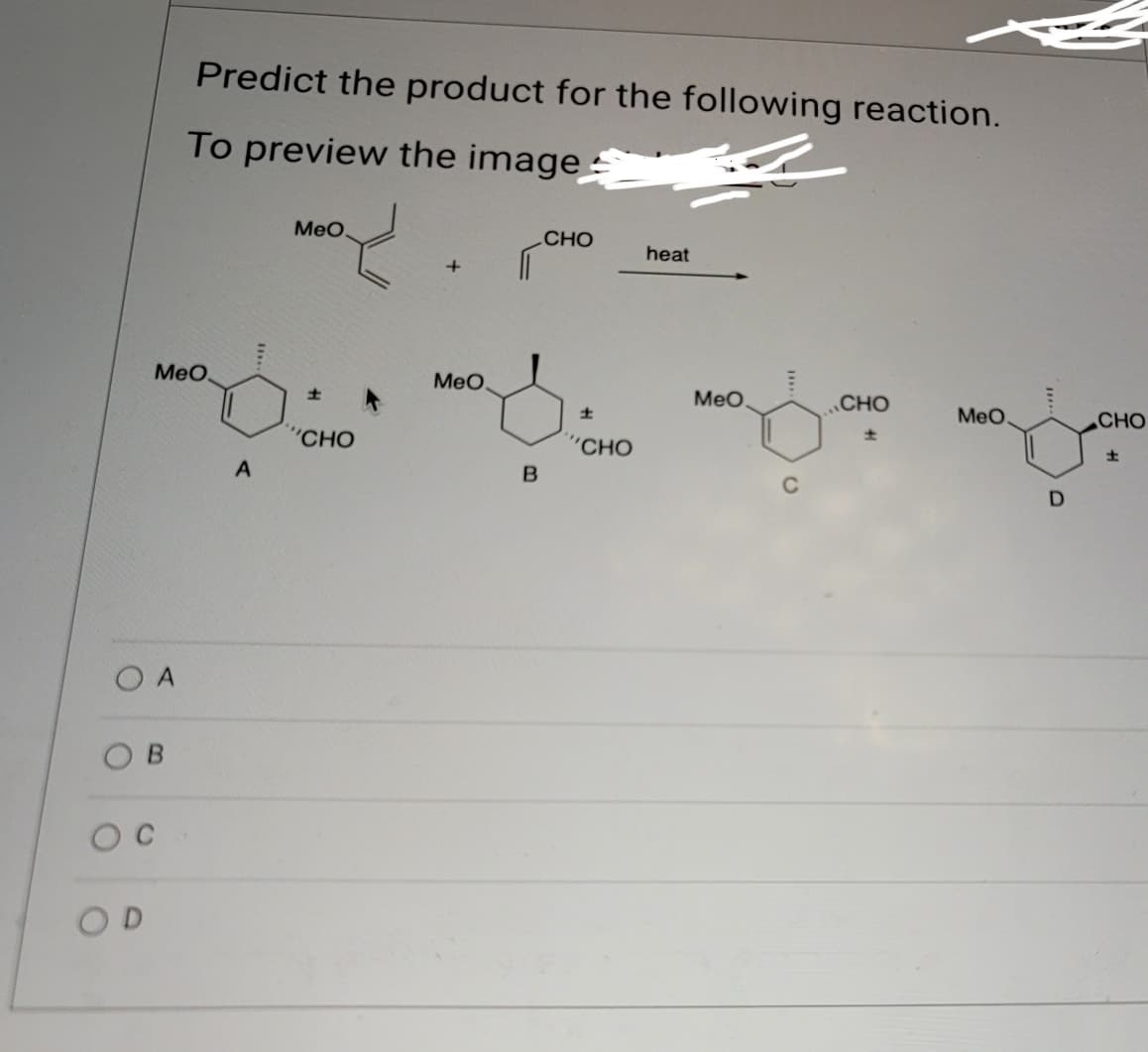
Q: Question 13 Identify the product (or multiple products) for each of the following reactions: A B C D…
A:
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: Incorrect Your answer is wrong-you missed some atoms. Highlight in red the higher priority…
A:
Q: Naming and Drawing Organic Molecules = Numbering the main chain of branched alkanes For each organic…
A: Step 1: Step 2: select the longest chain and give the number Step 3: Step 4:
Q: The pKa of benzoic acid is 4.2 Using the Henderson Hasselback equation calculate the ratio of…
A: The objective of the question is to calculate the ratio of ionized to unionized benzoic acid at a pH…
Q: (19) Explain the following polymerization mechanism by showing propagation reactions with the…
A: The objective of the question is to explain the polymerization mechanism involving a catalyst and a…
Q: The adrenal medulla secretes which of the following in the greatest quantity? Question 16…
A: The objective of the question is to identify which substance is secreted in the greatest quantity by…
Q: (please type answer fast).
A: The objective of this question is to calculate the pH of the solution after a certain volume of…
Q: None
A: Isomerism is the phenomenon where two or more molecules have the same molecular formula but differ…
Q: need help show charges and hydrogens on the oxygen or hydrogen atoms where appropriate .
A: The objective of the question is to identify the species found in the mixture.
Q: 1. Calculate Ksp for lead (II) chloride, if a 50.0 mL saturated solution of lead (II) chloride…
A: Note: As per the guidelines, the solution of the first question has been made. For the expert…
Q: Predict the major products of this organic reaction. Be sure you use dash and wedge bonds to show…
A: Step 1: When alcohol reacts with TsCl (thionyl chloride) and pyridine, it causes a substitution…
Q: how to synthesize this compound?
A:
Q: If a container of fuel is thought to release a total of 11,000 kJ of energy into a room with a…
A: The objective of the question is to calculate the average temperature increase in the room when a…
Q: A combinatorial library was built by reacting the carbonyl compounds in group A with the Grignard…
A: 1. When a halide reacts with Mg metal, it forms Grignard reagent. The given halides will form the…
Q: A о в C D ОЕ OF Question 23 Identify the product (or multiple products) for each of the following…
A: Step 1:Step 2: Step 3: Step 4:
Q: Draw major E2 ELIMINATION product. Hint: Review Chapter 10.3, the elimination can only occur if the…
A:
Q: b) Br2 H₂O c) d) H₂O Catalytic H₂SO4 1) BH₂.THF 2) H₂O₂/NaOH
A: The first two reactions are electrophilic addition reactions of carbon-carbon multiple bonds.The…
Q: Pls help me solve the following question and explain it making sure its correct!!
A:
Q: None
A: Step 1:- Most soluble amine (i) because of shorter carbon chain length and hydrogen bond formation…
Q: Draw the products of this halogenation reaction. Use a dash or wedge bond to indicate the…
A: Organic reactions can be defined as the reactions in which organic reactants react with each other…
Q: Question 7 Please predict the products for each of the following reactions: 1. 03 2. H₂O NaNH2 10 1.…
A: Alkyne reacts with HgSO4, H2SO4 and H2O to produce ketone.This reaction is known as hydration…
Q: Only typed solution
A: The objective of this question is to calculate the pH of a buffer solution prepared by mixing NH3…
Q: The actual vapor pressure is 260 torr. Is this an ideal solution? If not, what kind of deviation…
A: The given solution contains acetone and chloroform. Number of moles of acetone = 5 molNumber of…
Q: The mole fraction of neon in dry air near sea level is 0.00001818, where the molar mass of neon is…
A: Step 1: Use the Raoult's Law to solve the problem. Refer to the solution below.
Q: 14 Homework Unanswered Due Apr 11th, 11:59 PM Select the correct retrosynthesis to obtain the…
A:
Q: List the following compounds in order of increasing level of oxidation: OH II III Give detailed…
A: The objective of this question is to write the given compounds in increasing levels of…
Q: Please step by step solutions
A: Ionic bonding is a type of chemical bond that forms between two atoms when one atom transfers…
Q: Please show the synthesis for the following compound C, from any alkane (compound A). Compound C IR…
A: The question is based on the concept of organic spectroscopy. We need to analyse the spectral data…
Q: Question 10 A photon is quantum of electromagnetic radiation. A True B False
A:
Q: Please correct answer and don't use hend raiting
A: Dipole moment is the net overall polarity generated between two atoms when they are connected to…
Q: Question 9 Please predict the products for each of the following reactions: 1.03 2. H₂O NaNH, 1.…
A: Step 1: Step 2: Step 3: Step 4:
Q: Question 1 Please predict the products for each of the following reactions: 1. 03 2. H₂O NaNH, H2 Pd…
A: Step 1:Step 2: Step 3: Step 4:
Q: Zn(s) | Zn²⁺ (0.10 M) || Cu²⁺ (1.00 M) | Cu(s)? The standard cell potential for the reaction is…
A: Step 1:Step 2:
Q: What is the partial pressure (in atm) of gasoline if 1 gallon of gasoline completely evaporates into…
A: The objective of this question is to calculate the partial pressure of gasoline in a room after 1…
Q: 2. Propose reasonable products for the following reactions: OH Br H* OH t-BuOK t-BuOH CH2 NaOH of-06…
A: Step 1: Step 2: Step 3: Step 4:
Step by step
Solved in 2 steps with 1 images

- In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:Qa 18. Bromination of 5a-cholestan-3-one occurs at C-2 or C-4 to give 2 regioisomers. Two unsaturated ketones with lambdamax = 230 nm and lambdamax = 241 nm are yielded during dehydrobromination. Use Woodward-Fieser rules to distinguish between the two.Show the complete mechanism of the acidification of disodium adipate and calculate pKeq for both protonation steps. Note: pKa1 = 4.0, pKa2 = 4.4 for adipic acid. A mechanism must show all nbe’s and show movement of e’s with curved arrows.
- 5. Predict the product and show the mechanism of the following reaction:(Write out complete equations showing the structure of all reactants and products.) 2-methoxyheptane + Excess conc HBr/heat ----->Predict the product, draw the mechanism, and plot the reaction coordinate diagram for theE1 reaction.Predict the reaction of methyl 3-oxocyclohexane-1-carboxylate with NaBH4/ h20 show the mechanism
- E. J. Coreys 1964 total synthesis of -caryophyllene (essence of cloves) solves a number of problems of construction of unusual-sized rings. The first step uses an efficient photochemical [2 + 2] reaction. The desired stereochemistry and regiochemistry had been predicted based on model reactions. (a) [2 + 2] Reactions are quite common in photochemical reactions. Would this reaction be predicted to occur in the ground state? The next steps follow. Basic alumina is a chromatography support that will often act as a base catalyst. (b) What is the mechanism of the first step? (c) What is the mechanism of the second step? (d) Look at later steps in the synthesis. Does the stereochemistry of the added carbomethoxy group matter? The next steps are shown here. (e) What is the structure of compound (A)? (f) Give a mechanism for the formation of the cyclized product. (g) Give a mechanism for the first step. Hint: Attack on the lactone carbonyl may be the first step. (h) Give a structure for product (B). The following two steps are next. (i) Show the reactions of (B). (j) Write a mechanism for the ring-opening reaction. Hint: Note the presence of an acidic proton and a good leaving group in the molecule. The synthesis was completed by the following steps. (k) What is (C)? (l) What reagents would you use for these transformations?Isoleucine, another of the twenty amino acids found in proteins, is metabolized by a pathway that includes the following step. Propose a mechanism.Please show me the mechanism for O3+2-methyl-2-butene, stoping at the ozonide?
- the bromination of trans-stilbene produces a vicinal dihalide, 1,2-dibromide-1,2-diphenylethane. how do you know what a product's configuration (RR/SS, RS/SR, or pure RR or SS) is based on the mechanism of reaction?Consider the series of the trans effect: CO, CN-, C2H4 > PR3, H-, CH3- > C6H5- > NO2-, SCN-, I- > Br- >Cl- > py > NH3 > H20 What would be the major product of the following reaction? Select one:Predict the products of the following reaction. Co(s) + xsF2(g)---->







