Q: 2. Name each of the following compounds
A:
Q: dont provide handwriting solution ...
A: Step 1: Step 2: Ability to loose the proton is known as acidity .1. more the ability loose the…
Q: Draw the product of the reaction shown below at physiological pH (pH = 7.4). Ignore inorganic…
A:
Q: An unknown white powder have been seized by the police on the country border. The chemical analysis…
A: The objective of the given question is propose analytical strategy to establish whether the powder…
Q: Can you please include the species!
A: Step 1: Step 2: Step 3: Step 4:
Q: 1. What is the best method to obtain the product in the following reaction? Show all the steps and…
A:
Q: 9. Using the three balanced equations below, do 9.a through 9.d for each: a. b. C. d. How many grams…
A: Please Rate if this solution helps youThank You
Q: Name the following compounds. Do not use italics or boldface. a) N-CH2CH2CH3 CH3 N. b) CH3
A: Step 1:Name of molecules are a) 1-propylpyrrolidineb ) N,N-dimethylanilineNaming process of these…
Q: For of the following reaction, show the structural diagram and name of the reactant, product and the…
A: The objective of the question is to identify the reactants, products, and conditions necessary for…
Q: A 10.0 mL solution of 0.660 M NH3 is titrated with a 0.220 M HCl solution. Calculate the pH after…
A:
Q: Part 1 of 3 N, AS, P O<O<O Part 2 of 3 Sn, I, Rb O<O<O Part 3 of 3 Ca, O, P X
A: 1)The order of ionization energy for nitrogen (N), phosphorus (P), and arsenic (As) is N > P >…
Q: dont provide handwriting solution ....
A: The objective of the question is to find the concentration of OH- ions in a basic solution given the…
Q: 5 5. a. N-butynyl-N-butenylmethanamine C. b. N-butenyl-N-butynlmethanamine…
A: Step 1: identify the compound Step 2: write its iupac name Step 3: Step 4:
Q: None
A: In the molecule hemoglobin, the covalent interaction is represented by disulfide bridges . These…
Q: Based on the figure, which indicator would be appropriate to indicate the equivalence point of the…
A: Answer:Indicator is a chemical substance that changes its color at the equivalence point to indicate…
Q: QUESTION 38 What is the missing chemical, A, in the following reaction? HN HCI + A+ N
A: It is an organic reaction that consists of an amino alkylation of an acidic proton placed next to…
Q: Having trouble working through these questions
A: considering the solubility of Fe(OH)3 and the effect of NaOH on its solubility:a) Solubility…
Q: Identify ALL products of this reaction. Br Br NaO DMSO 00- DMSO = (solvent)
A: Here an alkyl halide begins halide react with sodium tert-butoxide in the presence of DMSO. We know…
Q: 17. Identify the major product of the following reaction. Br 008 Br Br2, FeBr3 Br ost for for || |||…
A:
Q: Naming and Drawing Organic Molecules Recognizing different skeletal structures How many different…
A:
Q: Complete the following solubilty table. (simply enter an S for soluble or an I for insoluble in each…
A: The cations and anions combine together to form a salt. The solubility of the salts (product) can be…
Q: None
A: Reaction 1: In this reaction, OH group have lone pair of electrons then they attack on the carbonyl…
Q: Draw a structural formula for the product of the reaction shown. CH3 H3CC CN L H H CH3 • Use the…
A: The given reaction is Diels alder reaction. The Diels alder reaction is defined as, Diene in…
Q: 8. Predict the product and draw a complete curved arrow mechanism for following reaction. Include…
A: Addition reaction-: The reaction in which a simple molecule is added to unsaturated compound mostly…
Q: 2. Rank the bolded-H in the following series of compounds from most (1) to least (5) acidic. You do…
A: The acidic strength of a substance depends on the stability of the conjugate base formed. The higher…
Q: what is the mechanism?
A: Step 1: Step 2: Step 3: Step 4:
Q: What is the systematic name of the product P of this chemical reaction? + KOH P+ H2O OH X G
A: Step 1:Given , CH₃COOH + KOH → [P] + H₂O The complete chemical reaction between acetic acid…
Q: Write the equillibrium expression for C(s) + 2N2O(g) CO2(g) + 2N2(g)
A: The objective of the question is to write the equilibrium expression for the given chemical…
Q: Naming and Drawing Organic Molecules Recognizing different skeletal structures How many different…
A:
Q: None
A: Step 1:For the three compounds, the nitrogen is connected to one carbon thus, it is a primary…
Q: 5. Draw the product and predict the position of the equilibrium for the following sigmatropic shifts…
A:
Q: You may want to reference (Page) section 5.6 while completing this problem. A gas mixture with a…
A: The objective of the question is to find the partial pressure of helium gas in a mixture and then to…
Q: What type of reaction is this? In this diagram, what is the potential energy of C? In this diagram,…
A: The graph of PE vs Reaction pathway is given
Q: Which of the following pairs of substances can NOT be used to make a buffer solution? a. HF & KOH…
A: Buffer systems are used in chemical systems to maintain the pH of the system constant. They are…
Q: -1 38. Aniline, C6H5NH2, when reacted with picric acid gives a derivative with an absorptivity of…
A: Given: ϵ=134cm−1⋅g−1⋅L;C=1.00x10−4M;b=1.00cm;MC6H5NH2=93.13g/molStep 1: Write the Beer's Law…
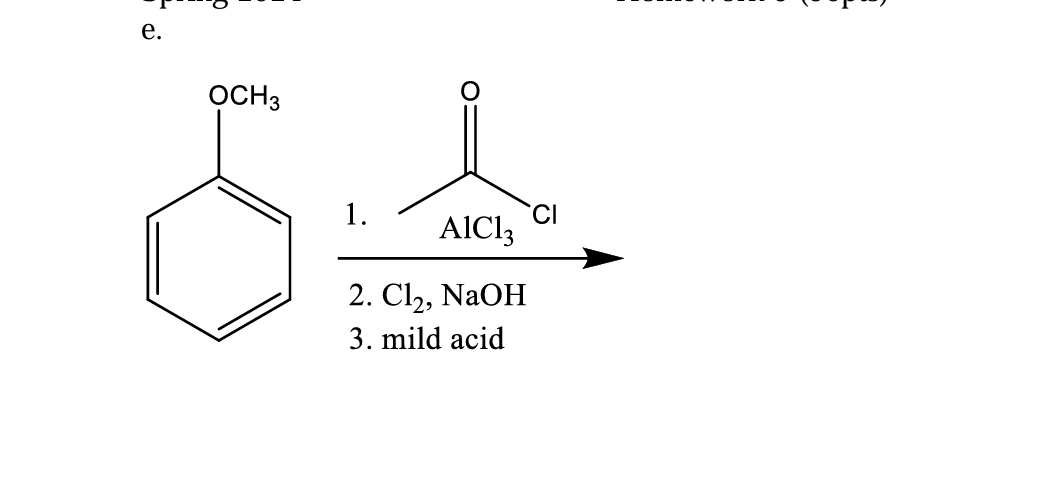
Q: H3CO -LL F CH3Cl AICI3
A: The aromatic electrophilic substitution reaction is a reaction in which an electrophile replaces the…
Q: écules in the table below is stable, in the exact form in which it is drawn, at pH=11. If you decide…
A: The question's objective is to decide whether the given molecules are stable or not at the given pH…
Q: Devise a synthesis for this product by using no larger than two or three carbons, or benzene as your…
A: The objective of this question is to to plan the synthetic route, considering the reactivity of…
Q: (b) Show how to synthesize the molecule at right, using the molecule at left as a carbon source,…
A: The addition of an enolate ion to an alpha-beta unsaturated carbonyl compound is called a conjugate…
Q: None
A: The Diels-Alder reaction involves cycloaddition of a conjugated diene (a molecule containing two…
Q: 6. Provide a synthesis for the molecule shown below. 1-Butanol is (again) your only allowed carbon…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: What ratio of NaCN to HCN is needed to prepare a pH 9.90 buffer? (Ka of HCN is 4.9 × 10⁻¹⁰)
A: The objective of this question is to find the ratio of sodium cyanide (NaCN) to hydrogen cyanide…
Q: Don't use hand raiting please
A: Given: [CH3OH]=20.3M;d=0.858g/mol;MCH3OH=32.05g/mol;m=???mol/kgStep 1: Analyze the given…
Q: The International Space Station is equipped with a Vika Oxygen Generator which uses the following…
A: The given reaction is:Volume (V) = Pressure (P) = 0.21 atmTemperature (T) = 24.5oCGas constant (R) =…
Q: Question 11 Type only one correct ACRONYM or only one short abbreviation for each chemical structure…
A: Since you are given multiple questions, I will be solving first 3 questions according to Bartleby…
Q: energy heat + H3CO A B reaction coordinate
A: The objective of the question is find out the mechanism of both the product formation and mention…
Q: Propose an efficient synthesis for the following compound using the malonic ester synthesis: EtO OEt…
A:
Q: What is the orbital diagram for Fe2+ that represents the ground state and the orbital diagram for…
A: An orbital diagram depicts the distribution of electrons within the orbitals of an atom or molecule.…
Q: Predict the major product for the following reaction. EtO. OEt Br- x Br 1) NaOEt 2) H₂O, heat
A:
Q: Indicate the units of the reaction rate for the kinetics of order 0, 1, 2 and 3. Are they the same?
A: The objective of the question is to determine the units of the reaction rate for the kinetics of…
Step by step
Solved in 2 steps with 1 images

- Titration of a 25.00 mL sample of acid rain required 12.4 mL of 0.009879 M NaOH to reach the end point. If we assume that the acidity of the rain is due to the presence of sulfuric acid, what was the concentration (in mol/l and %) of sulfuric acid in this sample of rain?Titration of a 25.00 mL sample of acid rain required 13.23 mL of 0.009567 M KOH to reach the end point. If we assume that the acidity of the rain is due to the presence of sulfuric acid, what was the concentration (in mol/l and %) of sulfuric acid in this sample of rain?What volume of 0.171 M KOH,0.171 M KOH, ?,V, can be neutralized with 58.5 mL58.5 mL of 1.21 M HNO3? V=
- In an impure sample of table salt that weighed 0.8421 g when dissolved in water and treated with excess AgN03 formed 2.044 g of AgCl. Listing the percentage of NaCl is in the impure sample. What is the list of three sources of error in this experiment?Sample: Saline 0.900% (m/v), in sodium chloride (NaCl). Data: M.A. (g/mol): Na = 22.9898; K = 39.0983; Cr=51.9961; Ag = 107.8682; Cl = 35.453; N=14.0067; O = 15.9994. Material available in the laboratory's warehouse: Reagents: distilled water; standardized solution of silver nitrate (AgNO₃), at a concentration of 0.09980 mols/L; Potassium chromate solution (K₂CrO₄) 1%; ammoniacal ferric alum solution [Fe(NH₄)(SO₄)₂] and; nitrobenzene (or cooking oil, alternatively); 0.1000 mols/L potassium thiocyanate (KSCN) standard solution. Glassware: beakers of all sizes available on the market; 50, 100, 150, 200 and 250 ml Erlenmeyers; 10.00 and 25.00 mL volumetric pipettes; 15.00 mL burette; 30.00, 50.00 and 100.00 mL volumetric flasks Calculate the molarity of saline, in terms of the NaCl concentration (concentration in mols/L). (a) 0.900 mols/L (b) 5.84 mols/L (c) 0.154 mols/L (d) 0.0154 mols/LWhat volume of 0.441 M KOH,0.441 M KOH, ?,V, can be neutralized with 50.0 mL50.0 mL of 1.64 M HNO3? ?= mL
- To prepare 50 mL solutions of 0.01 M KMnO4 in water (calculation to prepare chemicals in spectrophotometer)What volume in mL of 0.1354 M NaOH is required to nitrate 15.00 ML of 0.1002 M acid (HAc)?What volume of water should be added to 100.0 mL of a 0.500 N Cr2(SO4)3·18H2O solution to obtain a final concentration of 0.075 M? Show complete solution *Also find the number of replaceable ions (n)
- Titration of a 20.0-mL sample of acid rain required 1.7 mL of 0.0811MNaOH to reach the end point. If we assume that the acidity of the rain is due to the presence of sulfuric acid, what was the concentration of sulfuric acid in this sample of rain?Use the following atomic masses (in g/mol): Mg = 24.31; H = 1; S = 32.06; O = 16; Na = 23; Cl = 35. 45; Ca = 40.08; C = 12.01; N = 14.01; Mg = 24.31 A Kjeldahl procedure was used to analyze 356 μL of a solution containing 48.36 mg protein/mL. The liberated NH3 was collected in 5 mL of 0.0336 M HCl and the remaining acid required 6.34 mL of 0.010 M NaOH for complete titration. How many % nitrogen is in the protein sample?To what volume (in mL) must 27.4 mL of 17.2 M H2SO4 be diluted to produce 1.43 M H2SO4?









