You may want to reference (Page) section 5.6 while completing this problem. A gas mixture with a total pressure of 770 mmHg contains each of the following gases at the indicated partial pressures: 126 mmHg CO2, 234 mmHg Ar, and 193 mmHg O2. The mixture also contains helium gas. Part A What is the partial pressure of the helium gas? Express your answer in millimeters of mercury. PHe = 217 mmHg Submit Previous Answers Part B Correct Dalton's law of partial pressures states that the total pressure exerted by all of the gases in a mixture is the sum of the partial pressures exerted by the individual gases. The partial pressure of helium is determined by subtracting the partial pressures of CO2, Ar, and O2 from the total pressure. What mass of helium gas is present in a 10.2-L sample of this mixture at 278 K ? Express your answer in grams. - ΑΣΦ m = 5.14 ? g 09
You may want to reference (Page) section 5.6 while completing this problem. A gas mixture with a total pressure of 770 mmHg contains each of the following gases at the indicated partial pressures: 126 mmHg CO2, 234 mmHg Ar, and 193 mmHg O2. The mixture also contains helium gas. Part A What is the partial pressure of the helium gas? Express your answer in millimeters of mercury. PHe = 217 mmHg Submit Previous Answers Part B Correct Dalton's law of partial pressures states that the total pressure exerted by all of the gases in a mixture is the sum of the partial pressures exerted by the individual gases. The partial pressure of helium is determined by subtracting the partial pressures of CO2, Ar, and O2 from the total pressure. What mass of helium gas is present in a 10.2-L sample of this mixture at 278 K ? Express your answer in grams. - ΑΣΦ m = 5.14 ? g 09
Chapter5: Gases
Section: Chapter Questions
Problem 155CP
Related questions
Question
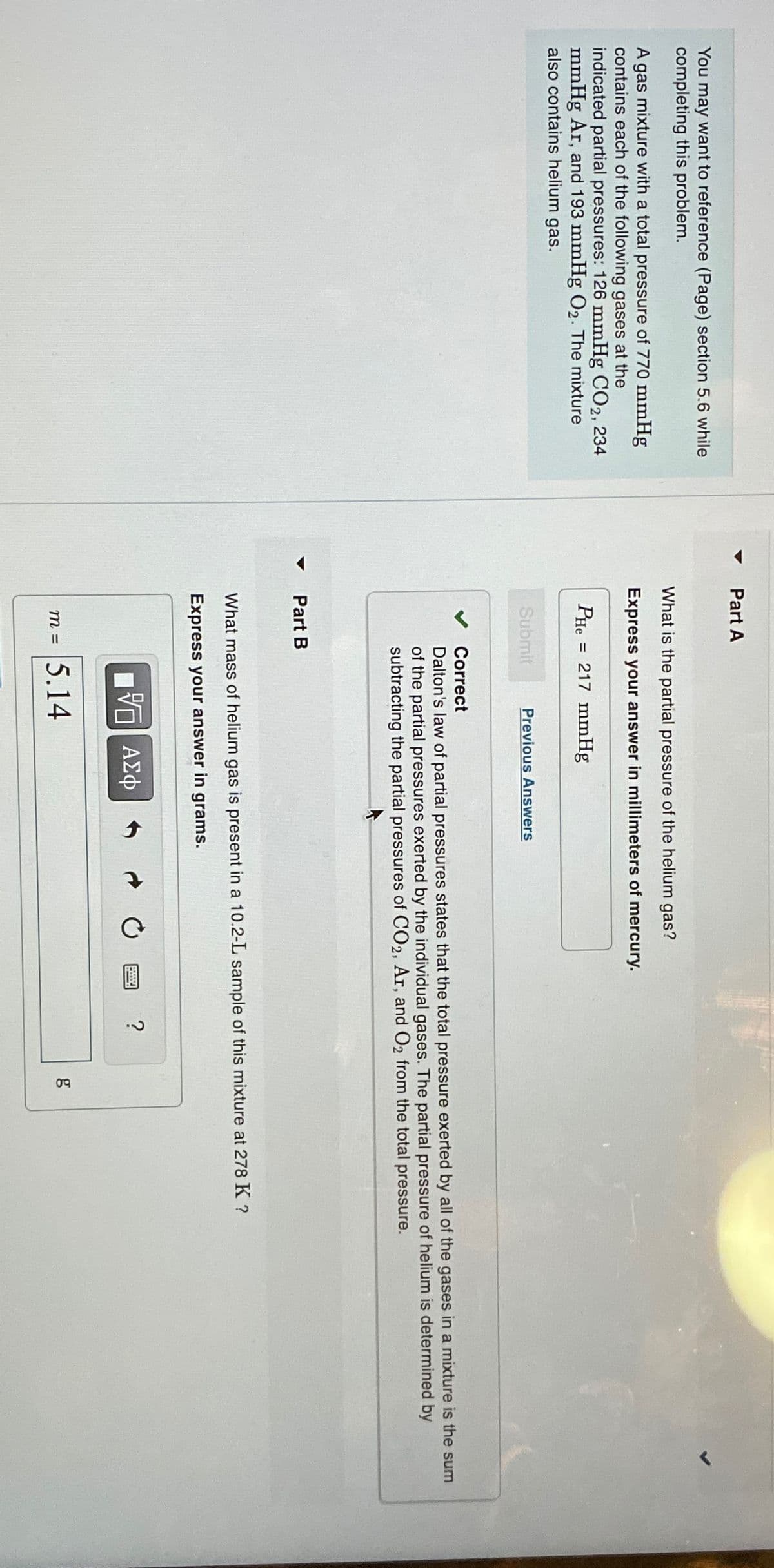
Transcribed Image Text:You may want to reference (Page) section 5.6 while
completing this problem.
A gas mixture with a total pressure of 770 mmHg
contains each of the following gases at the
indicated partial pressures: 126 mmHg CO2, 234
mmHg Ar, and 193 mmHg O2. The mixture
also contains helium gas.
Part A
What is the partial pressure of the helium gas?
Express your answer in millimeters of mercury.
PHe
=
217 mmHg
Submit
Previous Answers
Part B
Correct
Dalton's law of partial pressures states that the total pressure exerted by all of the gases in a mixture is the sum
of the partial pressures exerted by the individual gases. The partial pressure of helium is determined by
subtracting the partial pressures of CO2, Ar, and O2 from the total pressure.
What mass of helium gas is present in a 10.2-L sample of this mixture at 278 K ?
Express your answer in grams.
- ΑΣΦ
m = 5.14
?
g
09
AI-Generated Solution
Unlock instant AI solutions
Tap the button
to generate a solution
Recommended textbooks for you


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning