Q: 2. For the hypothetical equilibrium reaction: M(g) + N(g) O(g) + P(g) ---> Predict whether the…
A: According to the Le Chatelier's principle- If an equilibrium is brought to change in reaction…
Q: Consider the following reaction where K. = 2.90x102 at 1150 K: 2 S03 (g)= 2 So2 (g) + 02 (g) A…
A: First we would Calculate value of reaction quotient ( Qc ) using given Concentrations of each…
Q: Which species functions as the reducing agent in the following reaction? 2…
A: If chemical species donate electron then called as reducing agent
Q: 2-aminophenol hydrochloride is pictured at right. As is, the acid is fully protonated, the H from…
A: 2-aminophenol hydrochloride, HO-C6H4-NH3+Cl- is a diprotic acid with pka values of 4.70 and 9.97.…
Q: gमक ठ९१०छ्लम्ख्च र्रल a+.त्मेंन वत्टवणणकদट amode ņ वोक zmes) | zmzt caq) ll Nizt car) Nics) Nirs) -…
A: In this question, we have to write the half reaction occuring at anode for the given galvanic cell…
Q: C,H O2 CO2 H;0 C;H12 co2 H,0 CH12 O2 CO2 H,0
A:
Q: When the Pb2+ concentration is 1.37 M, the observed cell potential at 298K for an electrochemical…
A:
Q: A system expands from a volume of 1.00 L to 2.00 L against a constant external pressure of 1.00 atm.…
A:
Q: The reaction of 2-methylpropene with HBr in ether is described below. Answer the following questions…
A: Given reaction is : Which product is the anti-Markovnikov product = ?
Q: the partial pressure of N2 is 2.70 x 105 Pa and the partial pressure of Ne is 5.00 x 105 Pa in a…
A:
Q: What is the product of the following reaction ? CH3(CH),CVAICI; CI CI C1
A: In this question we have to tell the product of the reaction.
Q: SOLUTE (NONELECTROLYTE) SOLVENT (WATER) Mass (g) MW (g/mol) Density (g/mL) 60 700 200 18 1.5 1.0
A: Answer: In this question we have to find out the value of different concentration terms with the…
Q: 3 2 1 O D 5. 1H +1H → On+. 4 4Be 4 O 2He 2Не 3 On 1H
A: In this question we have to tell the missing part in the reaction.
Q: What is the molarity of a solution of calcium chloride which contains 33500 mg of solute in a total…
A: Molarity is calculated using formula : Molarity = Moles of soluteVolume of solution (L) Note : As…
Q: Which solution will have the lowest pH? 0.25 M HClO 0.25 M NaClO 4 0.25 M HClO 4…
A:
Q: What is a correct name of the following compound ? NH O acetanilide O methylacetanilide…
A: The given compound is an amide (RCONH2) as it has a carbonyl group directly attached to N atom. The…
Q: What is the general layout/structures of the anode and cathode?
A: Given, the general layout/structures of the anode and cathode
Q: 38. /TEA CI H2N. еxcess NH2 excess
A:
Q: Iron by nature is paramagnetic metal. How the paramagnetic effect nature of Fe can be modified?
A: the solution is shown below.
Q: Which one of the followin shoul have the hiest boiling point? PH3 H2S HCL SiH4 H2O
A: Given substances are : a). PH3 b). H2S c). HCl d). SiH4 e). H2O Which one of the following…
Q: Given the following formation reactions of carbon dioxide, water, and ethanol, compute for the total…
A: Given-> C2H5OH(l) + 3O2(g) ---> 2CO2(g) + 3H2O(g)
Q: Glucose and galactose Energy of activation with lactase Energy Lactose and water Energy of…
A: Answer: In this question we have to mark the energy profile of the hydrolysis reaction of Lactose,…
Q: Question 4 What is the mass % of NaCl In a mixture that Is prepared by dissolving 2.40 g of NaCl In…
A: Mass of NaCl = 2.40 g Mass of H2O = 65.5 g Mass % of NaCl = ?
Q: The figure represents the spontaneous deposition of iodine in which iodine vapor, I2(g), becomes…
A:
Q: What is true regarding the reaction of acetyl chloride and methylamine ? Multi-select. Select all…
A: Acetyl Chloride reacts with methylamine to form N-methylacetamide Correct statement regarding this…
Q: 7. 2 SO3(g) 2 SO2(g) + O2(g) : Кр3 16.5; Pso3 = 0.45 atm; Pso2 = 1.52 atm; Po2 = 0.210 atm || 8. 2…
A:
Q: 1. O3 14. 2. (work-up) OH LOH C. A. B. 15. CI A. B. C. O, (CH),S O ... NaHCO, NatHCOg
A: In this question we have to tell the product of the reaction and also the reagent.
Q: What is the solubility of argon (in units of grams per liter) in water at 25 °C, when the Ar gas…
A:
Q: A student ran the following reaction in the laboratory at 1098 K: 2 S03 (g) 2 So2 (g) + 02 (g) When…
A: 1.) To calculate equilibrium constant , we first need to calculate equilibrium concentrations of all…
Q: The following statements/concepts describe thermochemical equations correctly EXCEPT ____________.…
A: The following statements/concepts describe thermochemical equations correctly EXCEPT ____________.
Q: Reactants A and B are mixed in a beaker by Ethan. He waits for it to react completely. He knows that…
A: GIVEN: Reactants A and B reacts completely to form 3 products. Product C is obtained first since it…
Q: R-CR R H 1 The following compound can be dassified as an acid NO to. Strong and a low pka value b.…
A:
Q: What. is the addit.on the pH of 25.0mLof resulting from e solution with pH 3,550 I1.25e? (10.9) of…
A:
Q: How to look into whethere there is a lack of electrons?
A: How to find out which atom has a deficiency of electrons.
Q: Provide the correct IUPAC name for the compound shown here. H3C H H CH-CH3 CH3
A:
Q: From benzene and thiophene, synthesize the compound below
A:
Q: 3. A0.1017-g sample of KBrO, (MM 167.0) was dissolved in dilute HCl and treated with an unmeasured…
A: Given reactions are : Mass of KBrO3 = 0.1017 g Molar mass of KBrO3 = 167.0 g/mole Volume of…
Q: In an experiment done, a sample of water was found to contain 400 g of calcium per m³ of sample.…
A: Ppm or ppb are concentration terms Here we are required to express the mass of calcium in (a) Parts…
Q: ᴡʀɪᴛᴇ ᴛʜᴇ ᴄᴏᴍᴘʟᴇᴛᴇ ʙᴀʟᴀɴᴄᴇᴅ ᴇQᴜᴀᴛɪᴏɴꜱ ᴏꜰ ᴛʜᴇ ꜰᴏʟʟᴏᴡɪɴɢ ᴄʜᴇᴍɪᴄᴀʟ ʀᴇᴀᴄᴛɪᴏɴꜱ. ꜱʜᴏᴡ ʏᴏᴜʀ ꜱᴏʟᴜᴛɪᴏɴ
A:
Q: I Review | Constants | Periodic Table What is the pH of a solution obtained by dissolving two The…
A:
Q: Consider the equation: 2A + 3B -->2C deltaG° (A) = 55.15 kJ/mol deltaG° (B) = -186.49 kJ/mal deltaG°…
A: Answer:- Given reaction 2A + 3B -->2C ∆G° (A) = 55.15…
Q: at a pH value of one, what will be the structures observed for both a and b?
A: here we are required to find structure of given amino acid at pH=1
Q: Which of the following aqueous solutions are good buffer systems? U0.36 M barium iodide + 0.29 M…
A: Buffer solution are those which resist any significant change in pH if any strong acid or base is…
Q: Consider the reaction data. A → products T (K) k (s-1) 225 0.372 625 0.611 What two points should be…
A: Here we are required to find the activation energy of the reaction
Q: A chemist performs a gravimetric analysis. The chemist combines 1.00L of 2.00MAgNO3(aq) with 1.00L…
A: The reactants given are AgNO3 and NaCl.
Q: What will be the pH of a buffer solution containing an acid with a pKa of 5.4 with an acid…
A: We have to predict the pH of buffer solution.
Q: Which calorie source is more concentrated: fat or carbohydrate? Explain your response.
A: Fat is more concentrated source of calories than carbohydrate. Even fat is more calorie…
Q: Consider the benefits and drawbacks of nuclear power before making a decision. Is there a compelling…
A: Benefits After the initial cost of construction, nuclear energy has the advantage of being one of…
Q: Bromine-88 is radioactive and has a half ife of 16.3 seconds. What percentage of a sample would be…
A:
Q: Common Name: MS IUPAC Name: Q Zoom Out CAS No.: Solvent: Non Polar BP: MP: 1.00 0.90 0.80 0.70 0.60…
A: We are given the mass spectroscopy data for an unknown compound and we have to find whether it…
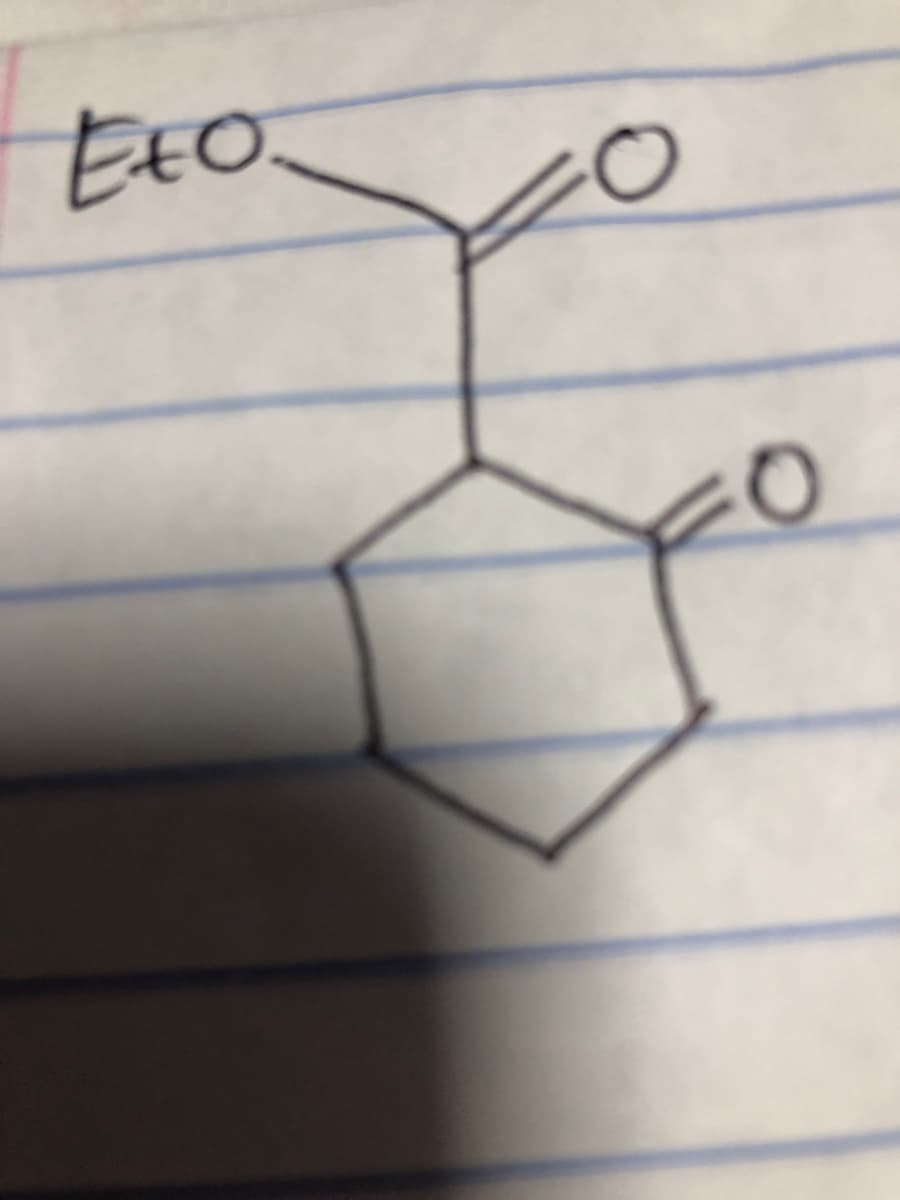
Step by step
Solved in 2 steps with 2 images


