Elementary sulfur is often found after a volcanic eruption. The sulfur dioxide gases react with the hydrogen sulfide gases released from the eruption. 8SO2(g) + 16H2S(g) → 3S8(s) + 16H20(I) Substance AH;(kJ/mol) AG;(kJ/mol) SO-(g) H;S(g) S8(s) H2O(1) -296.4 -300.4 -20.15 -33.0 -285.8 -237.2 a. Using the thermodynamic data provided at 25.0°C determine the entropy of reaction (J/Kmol). b. Will this reaction be spontaneous at all temperatures? Explain your answer using the values calculated.
Elementary sulfur is often found after a volcanic eruption. The sulfur dioxide gases react with the hydrogen sulfide gases released from the eruption. 8SO2(g) + 16H2S(g) → 3S8(s) + 16H20(I) Substance AH;(kJ/mol) AG;(kJ/mol) SO-(g) H;S(g) S8(s) H2O(1) -296.4 -300.4 -20.15 -33.0 -285.8 -237.2 a. Using the thermodynamic data provided at 25.0°C determine the entropy of reaction (J/Kmol). b. Will this reaction be spontaneous at all temperatures? Explain your answer using the values calculated.
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter21: Rates Of Chemical Reactions, Ii. A Clock Reaction
Section: Chapter Questions
Problem 2ASA
Related questions
Question
please help me by solving accurate and all sub-parts exact please do it fast I'll rate it up pls accurate thanks
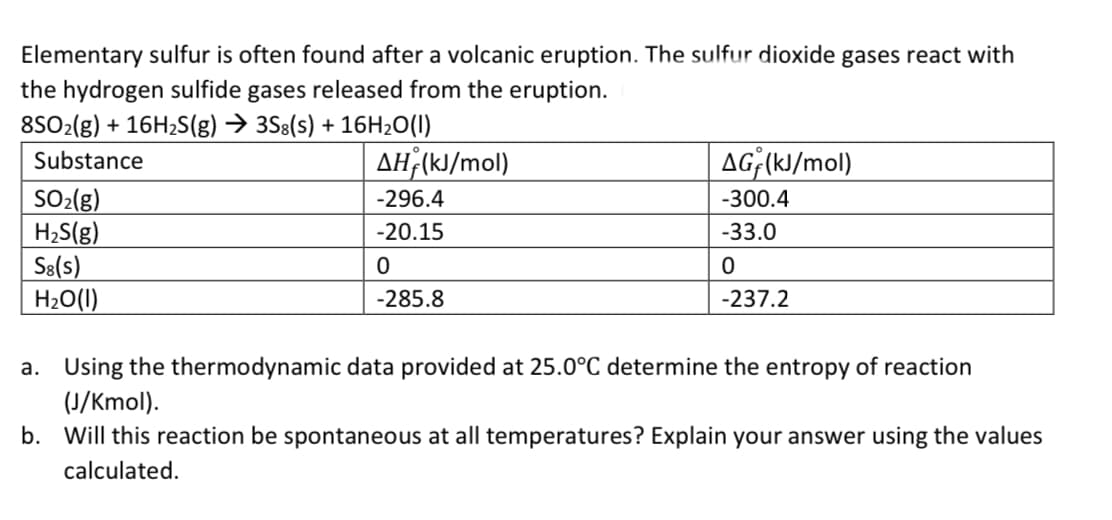
Transcribed Image Text:Elementary sulfur is often found after a volcanic eruption. The sulfur dioxide gases react with
the hydrogen sulfide gases released from the eruption.
8SO2(g) + 16H2S(g) → 3S8(s) + 16H20(I)
Substance
AH;(kJ/mol)
AG;(kJ/mol)
SO-(g)
H;S(g)
S8(s)
H2O(1)
-296.4
-300.4
-20.15
-33.0
-285.8
-237.2
a. Using the thermodynamic data provided at 25.0°C determine the entropy of reaction
(J/Kmol).
b. Will this reaction be spontaneous at all temperatures? Explain your answer using the values
calculated.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning