ellumHMAC=e84666a38b6ed866c9d641cbecc78b80#10001 11 of 44 Review | Constants | Periodic Table Part D A student sets up two reactions. Reaction 1 uses 0.250 mol/L of reactant, and Reaction 2 uses 0.410 mol/L of reactant. How many times faster is Reaction 2 compared to Reaction 1? Express your answer as a multiple of the rate for Reaction 1 to three significant figures. Note that Reaction 1 is already written for you, so just enter the number. ▾ View Available Hint(s) ▼ Hint 1. Setting up the ratio Since 0.410/0.250 = 1.64, the second reaction contains 1.64 times the amount of reactant compared to Reaction 1. The reaction rate is proportional to the square of the amount of reactant, so the rate will increase by a factor of the square of 1.64. VO 5 ? rate of Reaction 2 X rate of Reaction 1 Mal | ΑΣΦ 8 an 3:28 PM
ellumHMAC=e84666a38b6ed866c9d641cbecc78b80#10001 11 of 44 Review | Constants | Periodic Table Part D A student sets up two reactions. Reaction 1 uses 0.250 mol/L of reactant, and Reaction 2 uses 0.410 mol/L of reactant. How many times faster is Reaction 2 compared to Reaction 1? Express your answer as a multiple of the rate for Reaction 1 to three significant figures. Note that Reaction 1 is already written for you, so just enter the number. ▾ View Available Hint(s) ▼ Hint 1. Setting up the ratio Since 0.410/0.250 = 1.64, the second reaction contains 1.64 times the amount of reactant compared to Reaction 1. The reaction rate is proportional to the square of the amount of reactant, so the rate will increase by a factor of the square of 1.64. VO 5 ? rate of Reaction 2 X rate of Reaction 1 Mal | ΑΣΦ 8 an 3:28 PM
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter14: Chemical Equilibrium
Section: Chapter Questions
Problem 14.35QE
Related questions
Question
100%
Need help with this Chemistry Problem
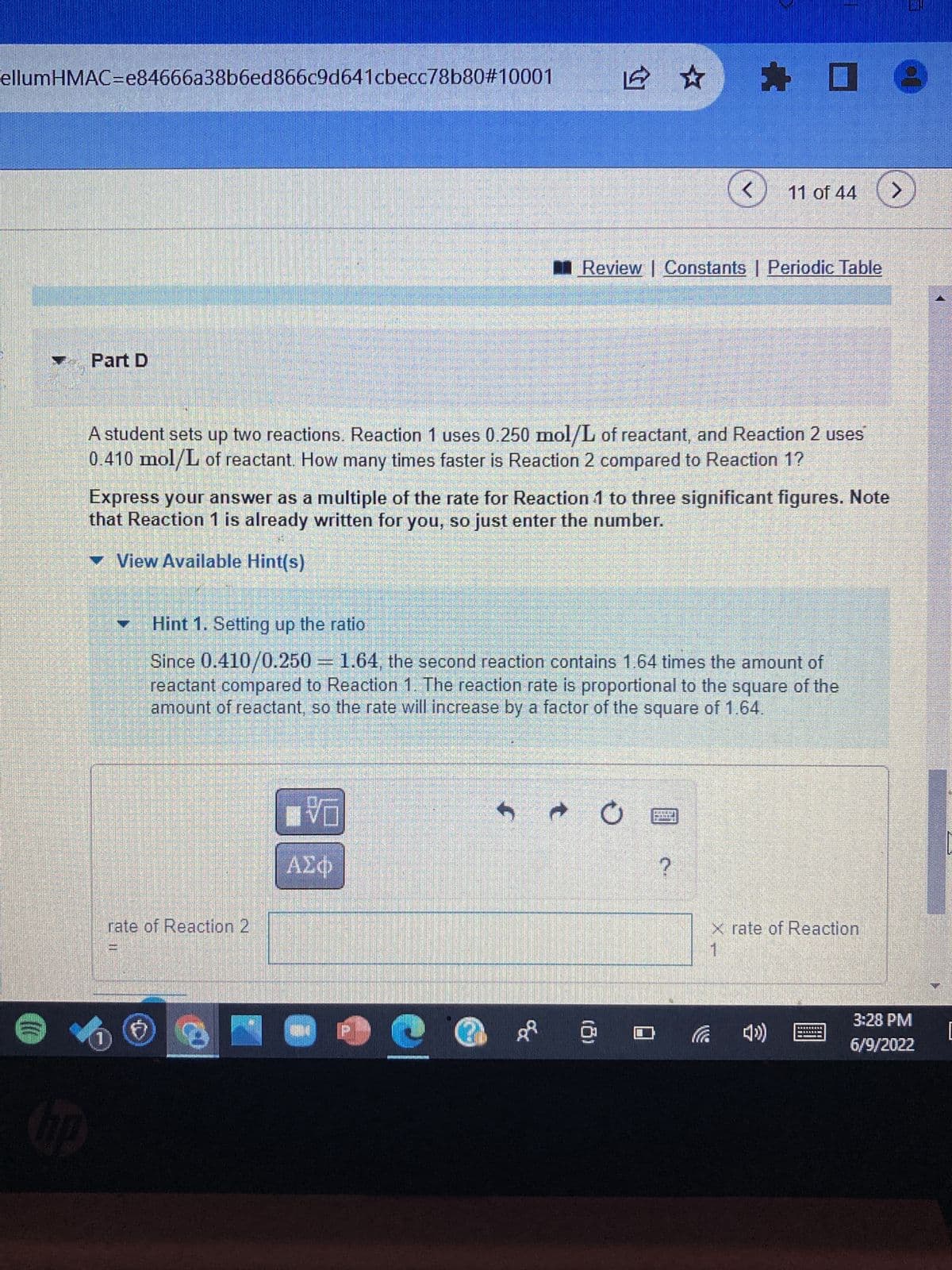
Transcribed Image Text:ellumHMAC=e84666a38b6ed866c9d641cbecc78b80#10001
1 ☆
E
< 11 of 44
Review | Constants | Periodic Table
Part D
A student sets up two reactions. Reaction 1 uses 0.250 mol/L of reactant, and Reaction 2 uses
0.410 mol/L of reactant. How many times faster is Reaction 2 compared to Reaction 1?
Express your answer as a multiple of the rate for Reaction 1 to three significant figures. Note
that Reaction 1 is already written for you, so just enter the number.
▾ View Available Hint(s)
T
Hint 1. Setting up the ratio
Since 0.410/0.250 = 1.64, the second reaction contains 1.64 times the amount of
reactant compared to Reaction 1. The reaction rate is proportional to the square of the
amount of reactant, so the rate will increase by a factor of the square of 1.64.
■マロ
5è O
M流iLin
BLO
ΑΣΦ
rate of Reaction 2
x rate of Reaction
- 100 ÅR
10
☐
09
?
3:28 PM
6/9/2022
[
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning