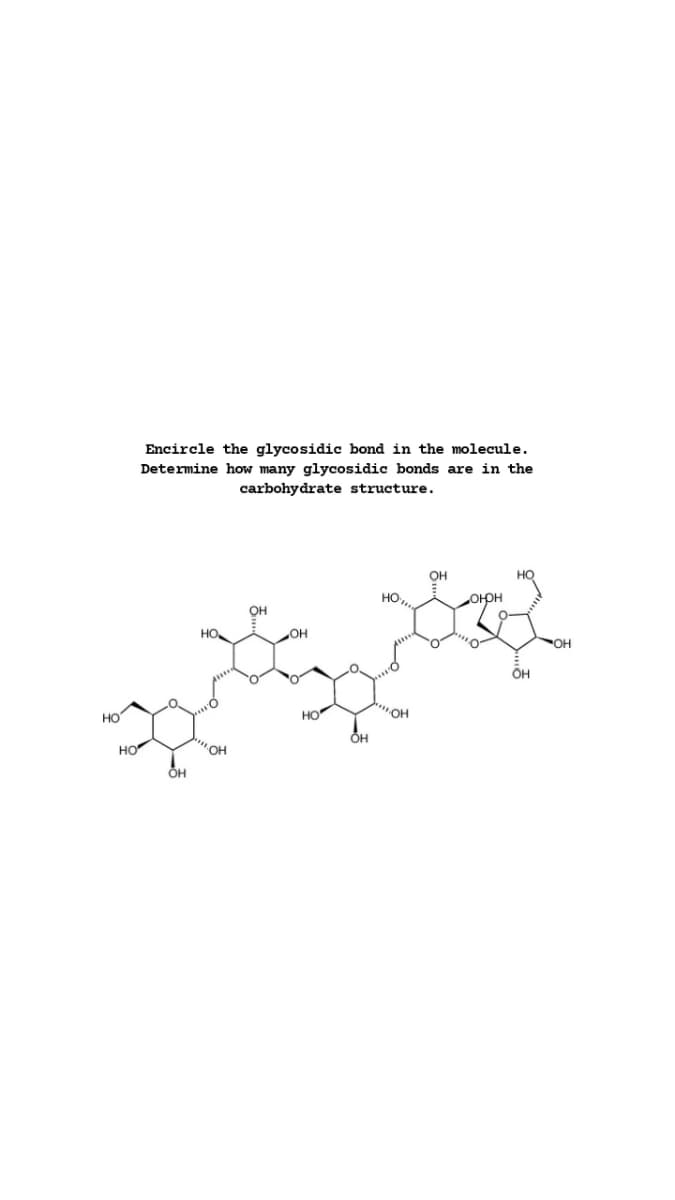
Encircle the glycosidic bond in the molecule. Determine how many glycosidic bonds are in the carbohydrate structure. он орн он HO он HO HO HO Он
Q: Each of the following enzymes are similar in that they are all regulation points for the…
A: Enzymes are chemical substances that function in several biochemical reactions and help in the…
Q: 1. A new drug was developed to inhibit RNA transcription of a new strain of bacteria infected in…
A: Enzymes are highly specialized proteins that have extraordinary catalytic power, greater than that…
Q: What is the reaction of fats with strong bases producing salts of the fatty acids known as metallic…
A: The triacylglycerols undergo stepwise enzymatic hydrolysis and liberate free fatty acids and…
Q: Which of the following molecules require(s) a transport/ shuttle system when transported across…
A: Glucose is utilized by glycolysis process, then it converts to Acetyl CoA which enters to TCA cycle…
Q: Which of the following best describes the mechanism of action of a cardiac glycoside? A)…
A: Cardiac glycosides are the compounds that are important for enhancing the force of heart to increase…
Q: Which compound is NOT a membrane lipid? a). Sphingolipid b). Glycerophospholipid c).…
A: A plasma membrane is composed of a lipid bilayer that allows the diffusion of non-polar molecules…
Q: Which amino acid sequence is likely to be found on the exterior of a soluble protein?
A: Proteins are polymers of amino acids linked by amide/peptide bonds. Amino acids are consist of amine…
Q: Do people need to feel socially and economically superior to others?
A: In the different nations, people have treated inequality, depending on their caste, economic levels…
Q: a. Anaerobic glycolysis can produce ATP at a much faster rate than aerobic oxidative…
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: first half of the Q cycle results in
A: The mechanism of Q-cycle is proposed by Peter Mitchell. Q-cycle terms as proton motive Q cycle. It…
Q: Ribose-5-phosphate is produced by oxidative decarboxylation of 6-phosphogluconate catalyzed by the…
A: Above questions is from PENTOSE PHOSPHATE PATHWAY.
Q: Describe and explain the function of uniprotkb-P39086
A: The P39086 is Glutamate receptor ionotropic (Ionotropic Glutamate Receptor) The neurotransmitter…
Q: 1 What is the dominant biochemical activity or molecular interaction that your protein is predicted…
A: Amyloid-beta precursor protein is single pass transmembrane protein with large extracellular domain…
Q: Which of the following CHÍNH, CH HO CHÁNH, CH₂OH I CH H₂N both a and b neither a nor b CHÍNH
A: Stereochemistry is also known as the subdiscipline of the chemistry. Stereochemistry involves the…
Q: State those enzymes involved in tricarboxylic acid and explain each.
A: The tricarboxylic acid cycle (TCA cycle) is a common metabolic pathway located in mitochondrial…
Q: 1. What are the different classes of lipids? State their function and identify their chemical…
A: A group of organic compounds includes lipids that are insoluble or poorly soluble in…
Q: what is the role of amino acids in purine metabolism?
A: Purine metabolism is described as a process in which different metabolic pathways are followed so as…
Q: The enzyme that catalyzes the conversion of H2CO3 to H20 + C02 is likely to be classified as
A: Enzymes is the substance or molecule which influenced the rate of reaction but not take part in the…
Q: Which process/es describe plant metabolism? O photosynthesis O Calvin cycle оо
A: The term metabolism describes all the chemical reactions involved in keeping the cells and organism…
Q: Which enzyme activity of the glycogen debranching enzyme is operating during the release of glucose…
A: The amylo-1, 6-glucosidase catalytic action of glycogen debranching enzyme permits it to hydrolyze…
Q: Alterations in our genome can lead to a variety of genetic diseases including cancers. These can be…
A: Genetic diseases occurs when some harmful variation occurs in the genetic makeup or genes of any…
Q: Using the Pearson square technique, determine the amount (lb) of skim milk (assume 0.0% milkfat)…
A: Skimmed milk contains no cream or less fat than regular milk. Typically, it contains just 0.1% fat…
Q: Identify and encircle the number of peptide bonds in the molecule. OH CH,SH…
A: Proteins are polymers of amino acids linked together by peptide linkages.
Q: 2.Why is it important for eukaryotic DNA to have multiple origins of replication, when a single ori…
A: DNA is a deoxyribonucleic acid. It is the main genetic component of eukaryotes and basic unit of…
Q: In terms of structure, what is the commonality between aldolase (glycolyssis) and transaldolase…
A: Aldolase is an enzyme that is involved in glycolysis and it converts fructose-1,6-bisphosphate into…
Q: How many molecules of Pyruvate can form from Glycerol metabolism as a by-product of fatty acid…
A: The major precursors of gluconeogenesis are lactate, glycerol, alanine, ketone bodies like…
Q: create a detailed flow chart or diagram that will illustrate all the important features of the…
A: Glycolysis is the metabolic pathway that converts glucose into pyruvic acid accompanied with the…
Q: 1. =0 Н—с —он Н—с —он Н—с —он CH2OH 2. нно н нонн онно I III TIT H-N-C-Ĉ-N-Ć-Ĉ-N-C-Ĉ-N--Ĉ-OH H-C-H…
A: Biological macromolecules can be classified into nucleic acids, proteins, lipids and carbohydrates.
Q: BSA (mg/ml) Absorbancy 540nm 0 0.158 1 0.210 2 0.260 3 0.305 4 0.360 5 0.410 6 0.455 7 0.510 8 0.530…
A: Biuret test is a chemical test used for detecting the presence of peptide bonds, in whose presence…
Q: 2. Use your knowledge of amino acids (and the R groups) and tertiary structures of proteins to…
A: The disease, which is referred to as sickle cell anaemia is caused because of mutation. In this…
Q: Consider 3 molecules of galactose: (write only the whole number; no decimal places) a. How many…
A: Galactose is an aldohexose sugar. The galactose unit is found in the milk sugar known as lactose.…
Q: What is the difference between substrate-level phosphorylation and oxidative phosphorylation? Please…
A: Phosphorylation : The transfer of the phosphate group from one compound to the other. Substrate…
Q: What is the meaning of DFR
A: DFR is Dihydroflavonol 4-Reductase (DFR), it is expressed by DFR gene.
Q: 1. What happens to the rate of liver glucose metabolism during moderate intensity exercise? 2. Would…
A: Carbohydrates are the main energy source for body, they broke down in to glucose as the sole ATP…
Q: Just answer the items with no answer yet, thank you!
A: Maltose is a disaccharide composed of α-D-Glucose molecules, which are linked together through α…
Q: Collagen is a fibrous protein which involves interactions between multiple helixes. Ohas a highly…
A: Collagen is a structural protein found naturally in humans and animals. It constitutes upto 30% of…
Q: b. High concentration of NADPH increases the rate of the pentose phosphate pathway by stimulating…
A: The pentose phosphate pathway is a metabolic pathway that occurs parallel to glycolysis in the…
Q: Why is fumarate hydrated? To enable it to interact with an enzyme O To form a double bond To reduce…
A: Introduction: The citric acid cycle is the final common pathway for the oxidation of various…
Q: Calculate AGo' for the reaction NADH + pyruvate → NAD+ + lactate You are given: Faradays constant F…
A: Electrons from NADH and a proton are used to reduce pyruvate into lactate in lactic acid…
Q: Which of the following monosaccharide phosphates is NOT produced in the pentose phosphate pathway?…
A: pentose phosphate pathway is a metabolic pathway of hexose oxidation where glucose-6-phosphate is…
Q: Why is the 3-Dimensional structure important for protein function?
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: CH2O에 off C1420H 3, 애 4 04 OH 4 of
A: The difference between alpha (α) and beta (β) glucose is the orientation of hydroxyl (-OH) group…
Q: A plant isolate was subjected to qualitative tests to determine the presence of macromolecules.…
A: Qualitative tests detect the presence of specific molecules in a sample solution. There are…
Q: Frank weighs 112 kg and John weighs 56 kg. Both are exposed to 5 mGy of gamma radiation. Do they…
A: The dosage of radiation absorbed per kilogramme. The level of radiation does not mean anything in…
Q: 7. Which of the following best characterizes the events that occur at an origin of DNA replication?…
A: Replication is the process of duplication of individual strands of a double stranded DNA. The…
Q: The enzyme that catalyzes the conversion of glucose to mannose is classified as a a. Isomerase b.…
A: Enzymes are classified as oxidoreductases, transferases, hydrolases, lyases, isomerases, and ligases…
Q: Enzymes exhibit saturation behavious because of a. limited amount of solvent b. lmited amount of…
A: Enzymes are protein molecules that increase the rate of reaction by decreasing the activation…
Q: The following is a part of the sequence on DNA template strand. 5' ATGGCCCGGTAAGTA 3' Write the…
A: A codon is a sequence of three consecutive nucleotides in a DNA or RNA that codes for a specific…
Q: Propose a mutation for Uniprotkb-P39086 and it’s effec
A: Mutations are the result of damage or alteration of a DNA gene such that the genetic message it…
Q: An unsaturated fatty acid produces more energy than the analogous saturated fatty acid. A) True B)…
A: Saturated fatty acids contain hydrocarbon chains that are only linked by single bonds. There are one…
Step by step
Solved in 2 steps with 1 images

- Circle and label the hemiacetal functional group and the acetal functional group in each of thefollowing carbohydrates:From the given statement below which is incorrect considering the biological roles of lipidsTRUE OR FALSE PROPERTIES OF FATTY ACIDS Contains polar hydrocarbon and non-polar carboxyl groups Carboxyl portion undergoes halogenation Forms ester bond on the hydrocarbon chain PROPERTIES OF POLYUNSATURATED FATS Forms single bonds and double bonds with hydrogen Undergo hydrogenation on bonds saturated with hydrogen Hardens when saturated with hydrogen
- Describe functions of carbohydrates. Describe the polymerization process of carbohydrates. Describe the degradation process of carbohydrates.During the dehydration reaction between two monosaccharides to create sucrose which is an example of a disaccharide, what chemical bond is formed between them? Van der Waals forces Ester bond Polar covalent bond Glycosidic bondDefinition of Lipids What is the Formation or breakdown
- The above monosaccharides (glucose and galactose) are held together in the disaccharide lactose by: disulfide bonds ether bonds glycosidic bonds ester bonds peptide bondslook at the molecule form for sucrose why does carbohydrates not follow the one to one carbon hydrogen oxygen ratio of monosaccharideWhich of the following membrane lipids have a direct glycosidic linkage between the head-group sugar and the backbone glycerol?a) Glycolipidsb) Phospholipidsc) Sphingolipidsd) Ether lipids
- Which one is NOT a property of glycerol? "backbone" in the simple lipid structure undergoes condensation with three molecules of fatty acids its OH groups reacts with the fatty functional groups of fatty acids it is a water-soluble ester 2. Why do fat molecules can pack closely together? "bends" polarity length LinearityAmylose is comprised of glucose monomers connected by what kind of glycosidic bond? a (1 -->4) a (1 --> 6) b (1 --> 4) b (1 --> 6) None of the aboveAdd these terms to your concept map should include these concepts and details: Structure (functional groups, shape); Monomer; Linkage; Properties; Functions; Key terms from the Partial List of Key Terms from the Minds Partial List of Key Terms essential nutrient carbohydrate monosaccharide isomer glycosidic linkage monomer polymer disaccharide oligosaccharide polysaccharide intermolecular force of attraction hydrogen bond macronutrient



