Enhanced - with Feedback I Review | Constam | temperature, in degrees Celsius, °C, for each of the dn do not change: • Part A reference (Pages 270 - 271) Section 8.4 while oblem. A sample of helium gas with a pressure of 270 Torr at 0°C is heated to give a pressure of 2500 Torr Express your answer to two significant figures and include the appropriate units. T = Value Units Submit Request Answer • Part B A sample of air at 35 °C and 800. mmHg is cooled to give a pressure of 690. mmHg. Express your answer as integer and include the appropriate units. ? T = Value Units
Enhanced - with Feedback I Review | Constam | temperature, in degrees Celsius, °C, for each of the dn do not change: • Part A reference (Pages 270 - 271) Section 8.4 while oblem. A sample of helium gas with a pressure of 270 Torr at 0°C is heated to give a pressure of 2500 Torr Express your answer to two significant figures and include the appropriate units. T = Value Units Submit Request Answer • Part B A sample of air at 35 °C and 800. mmHg is cooled to give a pressure of 690. mmHg. Express your answer as integer and include the appropriate units. ? T = Value Units
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter9: Energy And Chemistry
Section: Chapter Questions
Problem 9.102PAE: 9.102 A runner generates 418 kJ of energy per kilometer from the cellular oxidation of food. The...
Related questions
Question
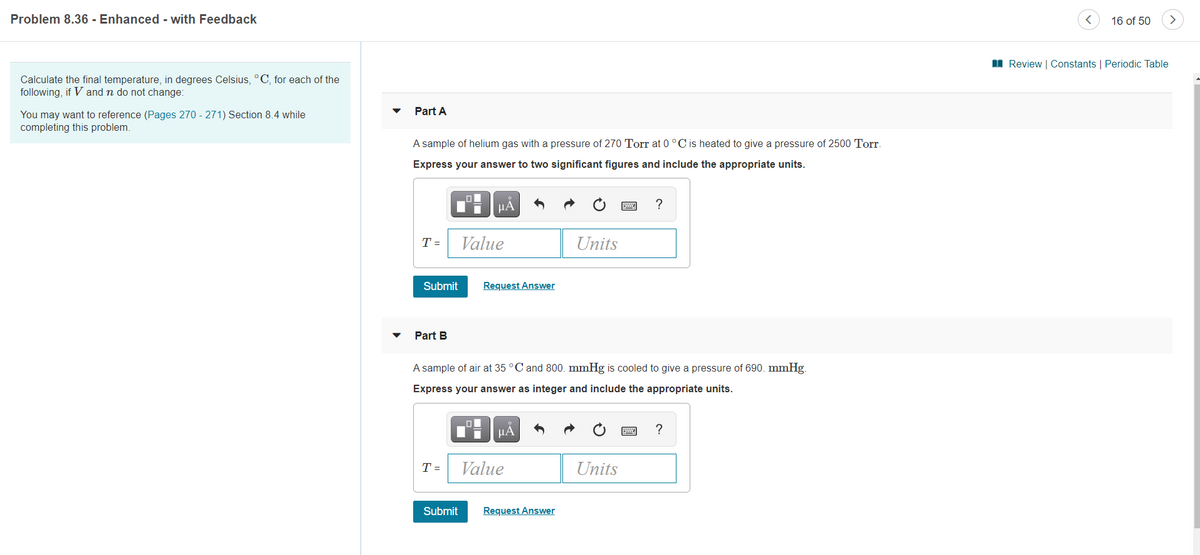
Transcribed Image Text:Problem 8.36 - Enhanced - with Feedback
16 of 50
>
I Review | Constants | Periodic Table
Calculate the final temperature, in degrees Celsius, °C, for each of the
following, if V and n do not change:
Part A
You may want to reference (Pages 270 - 271) Section 8.4 while
completing this problem.
A sample of helium gas with a pressure of 270 Torr at 0°C is heated to give a pressure of 2500 Torr.
Express your answer to two significant figures and include the appropriate units.
HA
?
T =
Value
Units
Submit
Request Answer
Part B
A sample of air at 35 °C and 800. mmHg is cooled to give a pressure of 690. mmHg.
Express your answer as integer and include the appropriate units.
μΑ
?
T =
Value
Units
Submit
Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning