Part III - B. A gas cylinder was measured to have different volumes at different temperature as shown in the Table 2. Table 2 VOLUME READING TRIAL TEMPERATURE TEMPERATURE (K="C+ 273.15) (°C) (mL) 1. Complete the data by converting the temperature into 1 25 2 2 30 57 Kelvin. 3 35 102 2. Plot the data. 4 40 152 Specific Week: 1 and 2 (LAS 1) Target Competency: Investigate the relationship between volume and pressure at constant temperature of a gas (S9MT-1lj-20) and Investigate the relationship between volume and temperature at a constant pressure (S9MT-Ilj-20) (This is a Government Property. Not For Sale.)
Part III - B. A gas cylinder was measured to have different volumes at different temperature as shown in the Table 2. Table 2 VOLUME READING TRIAL TEMPERATURE TEMPERATURE (K="C+ 273.15) (°C) (mL) 1. Complete the data by converting the temperature into 1 25 2 2 30 57 Kelvin. 3 35 102 2. Plot the data. 4 40 152 Specific Week: 1 and 2 (LAS 1) Target Competency: Investigate the relationship between volume and pressure at constant temperature of a gas (S9MT-1lj-20) and Investigate the relationship between volume and temperature at a constant pressure (S9MT-Ilj-20) (This is a Government Property. Not For Sale.)
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter1: Gases And The Zeroth Law Of Thermodynamics
Section: Chapter Questions
Problem 1.32E
Related questions
Question
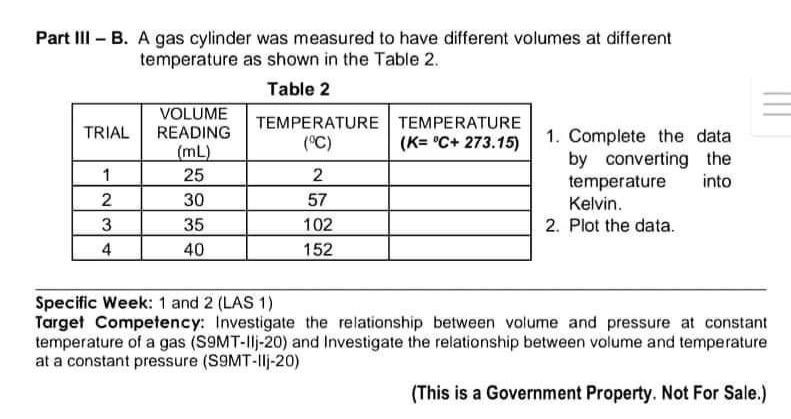
Transcribed Image Text:Part III - B. A gas cylinder was measured to have different volumes at different
temperature as shown in the Table 2.
Table 2
VOLUME
READING
TEMPERATURE
TRIAL
TEMPERATURE
(K="C+ 273.15)
(°C)
(mL)
1. Complete the data
by converting the
temperature into
1
25
2
2
30
57
Kelvin.
3
35
102
2. Plot the data.
4
40
152
Specific Week: 1 and 2 (LAS 1)
Target Competency: Investigate the relationship between volume and pressure at constant
temperature of a gas (S9MT-1lj-20) and Investigate the relationship between volume and temperature
at a constant pressure (S9MT-Ilj-20)
(This is a Government Property. Not For Sale.)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning