Enzymes Calculate the AG between glucose and ATP catalyzed by hexokinase: glucose + ATP glucose-6-phosphate + ADP Is the reaction spontaneous?| Given data: Glucose-6-phosphate + H,O= glucose + P; AG' = -3138 cal/mole ATP+H,O =ADP+ P. AG'=-7700 cal/mole
Q: Consider the following reaction: CH,OPO CH,OPO CH-OH CH-OPO ATP ADP н но он phosphofructokinase-1…
A: In the third step of glycolysis, fructose-6-phosphate is converted to fructose-1,6-bisphosphate. An…
Q: Assuming that the human body has 4 X 10" cells and that ATP is being used at a rate of 10° ATP per…
A: ATP (adenosine triphosphate) is the biomolecule that is considered the energy currency of the cells.…
Q: One of the most common enzyme deficiencies in humans is a mutation in the gene coding for glucose…
A: It involves the oxidation of Glucose-6-Phosphate to 6-Phosphogluconolactone acid which in turn is…
Q: A critical reaction in the production of energy to do work or drive chemical reactions in biological…
A: Adenosine triphosphate is also called ATP. It is defined as the molecule that takes energy compounds…
Q: What is the standard free-energy change, ∆G°, under physiological conditions(E. coli grows in the…
A: Glycolysis is the metabolic pathway that converts glucose into pyruvate. The free energy released in…
Q: Suggest a name for the enzyme that catalyzes each of the following reactions. a. transfer of a…
A: Cellular respiration or cell respiration happens in the living cells. In the cellular respiration…
Q: Calculate for the following based on complete oxidation of 5 molecules of trisaccharide containing 2…
A: Complete Oxidation of carbohydrates takes place via 4 stages. They are ; 1. Glycolysis : here a…
Q: The complete combustion of palmitate and glucose yields 9781 kJ ∙ mol−1 and 2850 kJ ∙ mol−1 of free…
A: Cellular Respiration is the oxidative process through which energy is obtained from the food. The…
Q: Calculate the actual, physiological ΔG for the reaction at 37 °C, as it occurs in the cytosol of…
A: Creatine is an organic chemical substance present in the body that helps to store high-energy…
Q: How much free energy will be released during ATP hydrolysis of ATP in this condition? Please write…
A: The following equation relate standard-state free energy of a reaction with the free energy: ∆G…
Q: . If palmitic acid is subjected to complete combustion in a bomb calorimeter, one can calculate a…
A: The oxidation of the palmitic acid produces 8 acetyl Co-A + 7FADH2 + 7NADH in 7 cycles of the…
Q: Consider the reaction: Fructose-1,6-bisphosphate (F1,6BP) +→ Glyceraldehyde 3-phosphate (GAP) +…
A: All cells require energy in the form of ATP to carry out a variety of functions, such as active…
Q: Which of the following compounds would you expect to liberate the least free energy when hydrolyzed?…
A: All the given molecules contain high energy bonds. Hydrolysis of these bonds will release free…
Q: Which of these statements concerning this reaction is true? acetaldehyde + NADH ↔ ethanol + NAD+…
A: Cellular respiration is the process by which an organism produce energy in the form of ATP. This ATP…
Q: What are the usual ionic forms of ATP and ADP in typical cells? Does this information have any…
A: Cellular respiration is a catabolic pathway of the process of metabolism, where a series of chemical…
Q: Creatine phosphate has a standard state free energy of hydrolysis of -43.1 kJ/mol. The given…
A: Gibbs free energy (G) of a system is the usable energy present in the system. For a reaction, free…
Q: If the DGo for ATP hydrolysis into ADP + inorganic phosphate is -7.3 kcal/mole, and the DGo for…
A: The delta G (dG) or change in Gibbs free energy determines whether a chemical reaction is favorable…
Q: Consider the reaction: Malate + NAD +→ Oxaloacetate + NADH (AG° = +29.7 kJ/mol) A cell at 25°C has…
A: All cells require energy in the form of ATP to carry out a variety of functions, such as active…
Q: Twenty-three milligrams of glucose were eaten by the bacteria Sanacoccus pumasareus. Calculate t…
A: The answer is 2.8× 1021 molecules
Q: If 0.1 M glucose 1-phosphate is incubated with phosphoglucomutase, the glucose 1- phosphate is…
A: Answer to (i) Given, concentration of glucose-1-phosphate at equilibrium = 4.5 x 10-3 M And…
Q: You are given a solution in which a biochemical reaction is occurring, and you are unsure whether AG…
A: Chemical reactions are performed with the utilization of Gibbs (∆G) free energy. If the ∆G of a…
Q: Identical reactions are catalyzed by the enzyme hexokinase with a Km of 0.2 mM in le cells and by…
A: In a classic Michaelis-Menten graph, the y-axis represents reaction rate and the x-axis represents…
Q: For the following reactions, name both the enzyme that catalyzes the reaction and the required…
A: Enzymes are considered as catalytic protein which helps in the catalysis of reaction. The coenzyme…
Q: If the DGo for ATP hydrolysis into ADP + inorganic phosphate is -7.3 kcal/mole, and the DGo for…
A: Given: ADP + Inorganic phosphate →ATP ∆G10=-7.3 kcal/mol…
Q: What terms would best describe the above coupled reaction? (If the DGo for ATP hydrolysis into ADP +…
A: The Gibbs free energy change determines whether a biochemical reaction is favorable or not. It…
Q: Which of the following is the correct summary of phase II of glycolysis for each molecule of…
A: Glycolysis is the process by which glucose is broken down to form pyruvate. The process involves…
Q: Consider the coupled reactions: 1,3-BPG → 3-PG + Pi ΔG°'= -16.3 kcalmolkcalmol ATP → ADP + Pi…
A: Glycolysis is the first step of cellular respiration that involves production of pyruvate from the…
Q: The concentrations of pyruvate, NADH, H+, lactate, and NAD are 2, 1.5, 1.5, 1.2, 1.2 mm,…
A: The quantitative study of the energy change in the living cells and of the nature is known as…
Q: A total of 30.5 kJ mol-1 of free energy is needed to synthesise ATP from ADP and Pi when the…
A: Food is a substance that consists of various nutrients such as carbohydrates, fats, proteins,…
Q: The enzyme glutamine synthetase catalyzes the following reaction: glutamate + ATP + NH3 → glutamine…
A: Answer. Glutamine synthetase (GS) catalyzes the ATP-dependent condensation of ammonia and glutamate…
Q: Glucose 1-phosphate is converted into fructose 6-phosphate in two successive reactions. Reaction 1:…
A:
Q: The cleavage of ATP’s phosphoanhydride bond to form ADP yields approximately _____________ kJ/mol.
A: Phosphoanhydride bonds are the high energy bonds present in the three phosphate groups of the ATP…
Q: Walking consumes approximately 100 kcal/mi. In the hydrolysis of ATP (ATP → ADP + Pi), the reaction…
A: The energy currency of a living cell is called ATP. It is then hydrolyzed into ADP or AMP. The…
Q: AG" = -80.8 kJ/mol Glycolysis: Glucose + 2 NAD* + 2 ADP + 2 Pi 2 Pyruvate + 2 NADH + 2 ATP + 2 H2O;…
A: Gibbs energy, or free energy , is a quantity that is used to measure the maximum amount of work done…
Q: Consider the following list of phosphorylated compounds with their free energy changes of phosphate…
A: The Gibbs free energy change (∆G°) is the energy change that occurs in going from reactants towards…
Q: A critical reaction in the production of energy to do work or drive chemical reactions in biological…
A: ATP is an organic compound made of adenine, ribose sugar, and 3 phosphates. In living cells, ATP…
Q: For each of the following pair of compounds, choose the compound that would yield the most energy…
A: Food provides the chemical energy that animals need. Food is made up of organic molecules with…
Q: In the reaction: glycogen to glu-1-phosphate, what reaction is involved? Name the enzyme.
A: " Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: The synthesis of ATP by oxidative phosphorylation, using the energy released by movement of protons…
A: Introduction:- The electron transport chain is a collection of proteins and organic compounds…
Q: For the following reactions, name both the enzyme that catalyzes the reaction and the required…
A: The citric acid (CAC) cycle is the major metabolic pathway in the cell, which helps to form the…
Q: Write the equation for net citric acid cycle and calculate one cycle equivalent to how many ATP?
A: Aerobic cellular respiration takes place in the presence of oxygen. It has three major steps that…
Q: Convert the following reactions into "Metabolic Engineering" notation: a. C6H1206 + 2 H20 → 2 C2H60…
A: A set of metabolic reactions and processes involving the conversion of chemical energy into ADP is…
Q: The 2nd puzzle Taylor has left you is below. They are examining the reaction X-X-X-X + ATP = X-X-X +…
A: The biocatalyst, enzymes are proteins that involve increasing the speed of the reaction without…
Q: You have been the only one who has been able to this. It has three other parts as well, A) Which…
A: Gibbs free energy change, equilibrium constant and reaction quotients are values for a reaction…
Q: How many ATP molecules could maximally be generated from one molecule of glucose, if the complete…
A: Introduction ATP (adenosine triphosphate) is an energy-carrying molecule found in all living things'…
Q: As a result of an experiment following measurements were obtained from a cell: ATP concentration of…
A: Gibbs energy: ∆G = ∆G°' + 2.303 RTlogQ∆G = Actual free energy∆G°' = standard energyR = Ideal gas…
Q: Write a balanced equation for the complete metabolic oxidation of each of the following. Include O2,…
A: The β oxidation of fatty acid occurs in the mitochondria of cells.it occurs in three stages and they…
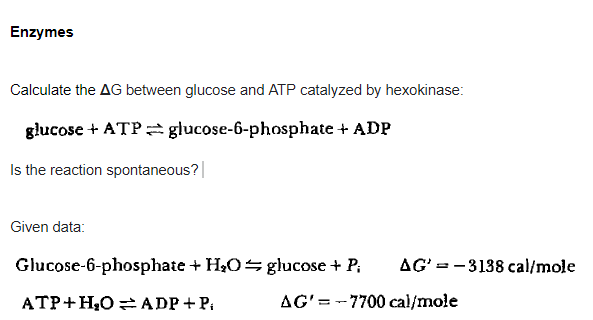
Step by step
Solved in 2 steps

- Consider the following chemical reaction: Glucose + ATP → Glucose-6-Phosphate + ADP + Pi Given the following information, calculate the actual free energy change (G’) of this reaction. G’ of Glucose + ATP → Glucose-6-Phosphate + ADP + Pi = -16.7 kJ/mol [Glucose] = 5.0 mM [ATP] = 1.85 mM [Glucose-6-Phosphate] = 0.083 mM [ADP] = 0.14 mM [Pi] = 1.0 mM Temperature = 37 C R (Gas Constant) = 8.314 J/mol•KWhat terms would best describe the above coupled reaction? (If the DGo for ATP hydrolysis into ADP + inorganic phosphate is -7.3 kcal/mole, and the DGo for maltose synthesis from glucose + glucose is +3.7 kcal/mole, calculate the standard free energy change for the combined reaction of ATP + glucose + glucose g ADP + maltose + inorganic phosphate.) it is non-spontaneous and endothermic (because the overall DGo is negative) it is spontaneous and exothermic (because the overall DGo is negative) it is non-spontaneous and endothermic (because the overall DGo is positive) it is spontaneous and exothermic (because the overall DGo is positive) it is non-spontaneous and exothermic (because the overall DGo is negative)Complete catabolism of one glucose molecule yields 38 ATP molecules. How many moles of ATP are produced by the complete catabolism of 10 moles of glucose?
- A total of 30.5 kJ mol-1 of free energy is needed to synthesise ATP from ADP and Pi when the reactants and products are at 1.0 M concentrations and the temperature is 25oC. Because the actual physiological concentrations of ATP, ADP, and Pi are not 1.0 M, and the temperature is 37oC, the free energy required to synthesise ATP under physiological conditions is actually ~ 46.2 kJ mol-1. A 68 kg adult requires an energy intake of 8,550 kJ of food per day (24 hours). Calculate the mass (in Kg) of ATP synthesised by a human adult in 24 hours, assuming that the percentage efficiency of converting inputted calories in ATP is 50%. What percentage of the body weight does this represent?How many ATP molecules could maximally be generated from one molecule of glucose, if the complete oxidation of 1 mole of glucose to CO2 and H2O yields 686 kcal of free energy and the useful chemical energy available in the high- energy phosphate bond of 1 mole of ATP is 12 kcal?The complete combustion of palmitate and glucose yields 9781 kJ ∙ mol−1 and 2850 kJ ∙ mol−1 of free energy, respectively. Compare these values to the free energy (as ATP) obtained though catabolism of palmitate and glucose under standard conditions. Which process is more efficient?
- In the third step of glycolysis, the given reactions are coupled. reaction 1: fructose-6-phosphate + Pi ⟶ fructose-1,6-bisphosphate + H2O (Δ? = −28 kJ/mol) reaction 2: ATP + H2O ⟶ ADP + Pi (Δ? = +13.8 kJ/mol) Calculate the overall ΔG (kJ/mol) for the coupled reaction.Using understanding of the thermodynamics laws, explain the energy transformations for the following processes. a. Glucose – Pyruvase + 2ATF + 2NADH b. Pyruvate – Acetyl-CoA + NaDH c. Citric acid – Oxoloacetate + 3NaDH + FADH2 + ATP d. H20 at PSII – ATP e. Photon at PSI – NADPH f. Co2 + RuBp – 3PG + ATP + NADPH – G3P (1/6 of which is converted to glucose)Assuming that the glycerol 3-phosphate shuttle is operative, a complete oxidation of five molecules of glucose (by a eukaryotic cell) into carbon dioxide and water would produce approximately _________ ATP molecules via oxidative phosphorylation only. (Consider NADH = 2.5 ATP and FADH2 = 1.5 ATP)
- Consider the coupled reactions: 1,3-BPG → 3-PG + Pi ΔG°'= -16.3 kcalmolkcalmol ATP → ADP + Pi ΔG°'= -7.3 kcalmolkcalmol Which of the following is TRUE about the total ΔG°’ for the coupled reactions? -9.0 kcal/mol; spontaneous +9.0 kcal/mol; nonspontaneous -23.6 kcal/mol; spontaneous +23.6 kcal/mol; nonspontaneousConsider the following equilibrium at 25ºC :Glucose-1-Phosphate Glucose-6-PhophateUsing the equilibrium concentrations of [Glucose-1-Phosphate] = 0.35 M and [Glucose-6-Phosphate] = 1.65 M, calculate BOTH K′eqand Gº′ for this reaction. Is this reaction exergonicor endergonic? R = 8.314 J/K·molHow many total (NOT NET) molecules of ATP are synthesized from ADP via glycolysis of a single molecule of glucose? Explain by detailing the reactions (steps) involved.

