ercise 18.62 - Enhanced - with Feedback 12 Fine I Costants i Etios Part B ISSED THIS? Read Section 18.4 (Pages 803817) What is the volume of added base at the equivalence point for HF? o 30 0 mL samples, one 0 100 M HCl and the ner 0 100 M HF, were titrated with 0200 M KOH swer each of the following questions regarding ase two titrations. Express your answer in millititers. ml. Submit Request Answer revious
ercise 18.62 - Enhanced - with Feedback 12 Fine I Costants i Etios Part B ISSED THIS? Read Section 18.4 (Pages 803817) What is the volume of added base at the equivalence point for HF? o 30 0 mL samples, one 0 100 M HCl and the ner 0 100 M HF, were titrated with 0200 M KOH swer each of the following questions regarding ase two titrations. Express your answer in millititers. ml. Submit Request Answer revious
Chapter11: Solving Equilibrium Problems For Complex Systems
Section: Chapter Questions
Problem 11.22QAP
Related questions
Question
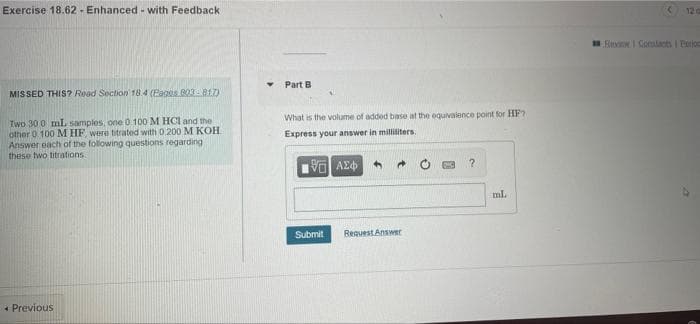
Transcribed Image Text:Exercise 18.62 - Enhanced - with Feedback
12 a
Eini I Cotacts I Eetios
Part B
MISSED THIS? Read Soction 18.4 (Pages 003-817)
What is the volume of added base at the equivalence point for HF?
Two 30 0 mL samples, one 0 100 M HCI and the
other 0 100 M HF, were titrated with D.200 M KOH
Answer each of the following questions regarding
these two titrations.
Express your answer in milliiters.
ml.
Submit
Request Answer
+ Previous
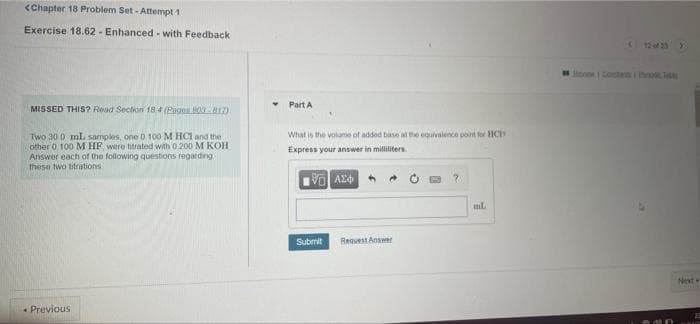
Transcribed Image Text:<Chapter 18 Problem Set - Attempt 1
Exercise 18.62 - Enhanced- with Feedback
(123
Sieome 1 Contan hod Tth
Part A
MISSED THIS? Read Section 18 4 (Pages 0017)
What is the volume of added base at the equivalence pont tor HC
Two 30 0 ml. samples, one D 100M HCl and the
other 0 100 MHF, were titratod with 0.200 M KOH
Answer each of the following questions regarding
these two titrations
Express your answer in milliliters
Submit
Request Answer
Next
- Previous
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

