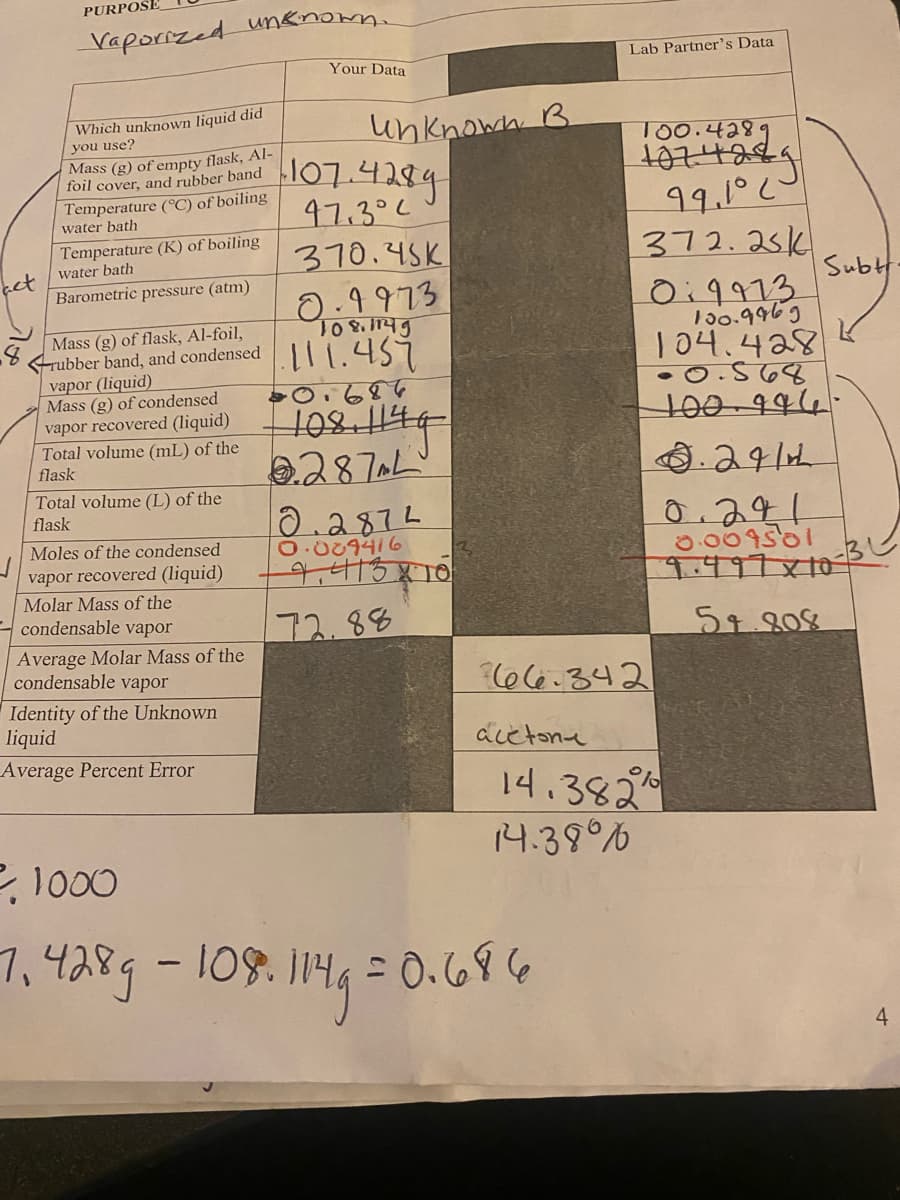
et Vaporized Which unknown liquid did you use? Mass (g) of empty flask, Al- foil cover, and rubber band Temperature (°C) of boiling water bath Temperature (K) of boiling water bath Barometric pressure (atm) Mass (g) of flask, Al-foil, rubber band, and condensed vapor (liquid) Mass (g) of condensed vapor recovered (liquid) Total volume (mL) of the flask Total volume (L) of the flask Moles of the condensed vapor recovered (liquid) Molar Mass of the condensable vapor Average Molar Mass of the condensable vapor Identity of the Unknown iquid verage Percent Error Your Data unknown B 107.4289 97.302 370.45K 0.4973 108.1149 111.459 0.686 108,114g 2.287AL 0.2872 0.089416 9.413810 72.88 Lab Partner's Data acetona 100.4289 1074289 99.1°20 372.251 66.342 Sub. K 019973 100.9969 104.428 -0.568 100-996- .241 0.241 0.009501 9.497x10-31 54.808 14.382% 143907
et Vaporized Which unknown liquid did you use? Mass (g) of empty flask, Al- foil cover, and rubber band Temperature (°C) of boiling water bath Temperature (K) of boiling water bath Barometric pressure (atm) Mass (g) of flask, Al-foil, rubber band, and condensed vapor (liquid) Mass (g) of condensed vapor recovered (liquid) Total volume (mL) of the flask Total volume (L) of the flask Moles of the condensed vapor recovered (liquid) Molar Mass of the condensable vapor Average Molar Mass of the condensable vapor Identity of the Unknown iquid verage Percent Error Your Data unknown B 107.4289 97.302 370.45K 0.4973 108.1149 111.459 0.686 108,114g 2.287AL 0.2872 0.089416 9.413810 72.88 Lab Partner's Data acetona 100.4289 1074289 99.1°20 372.251 66.342 Sub. K 019973 100.9969 104.428 -0.568 100-996- .241 0.241 0.009501 9.497x10-31 54.808 14.382% 143907
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter11: Solutions
Section: Chapter Questions
Problem 50P
Related questions
Question
Molar mass of the condensable vapor
Transcribed Image Text:act
PURPOSE
Vaporized unknown.
Which unknown liquid did
you use?
Mass (g) of empty flask, Al-
foil cover, and rubber band
Temperature (°C) of boiling
water bath
Temperature (K) of boiling
water bath
Barometric pressure (atm)
Mass (g) of flask, Al-foil,
rubber band, and condensed
vapor (liquid)
Mass (g) of condensed
vapor recovered (liquid)
Total volume (mL) of the
flask
Total volume (L) of the
flask
Moles of the condensed
vapor recovered (liquid)
Molar Mass of the
condensable vapor
Average Molar Mass of the
condensable vapor
Identity of the Unknown
liquid
Average Percent Error
Your Data
Unknown B
107.4289
97.302
370.45K
0.9973
108,1145
111.459
-0.684
108,1149
287L
0.2872
0.089416
- 9,413810
172.88
acetona
Lab Partner's Data
66-342
00.428
99.1°20
372.251
09913
130.9969
104.428
-0.568
100-996-
.241
0.241
0.009501
9.497×10-3
54.808
2, 1000
1
7, 428g - 108.114g = 0.686
14.382%
14.38°
Subt
4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning