Q: Et Me Me Excess EtMgBr 2) H,0 Et
A: Please find the attachment.
Q: Benzoic acid can be prepared as follows. Calculate the percentage yield NAOH HCI CN -COO Na -COOH…
A:
Q: 28 R'OH Br
A:
Q: Br Br ÇI
A:
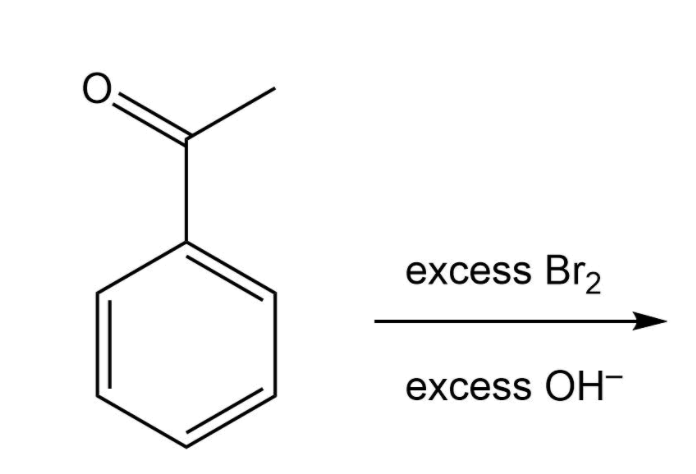
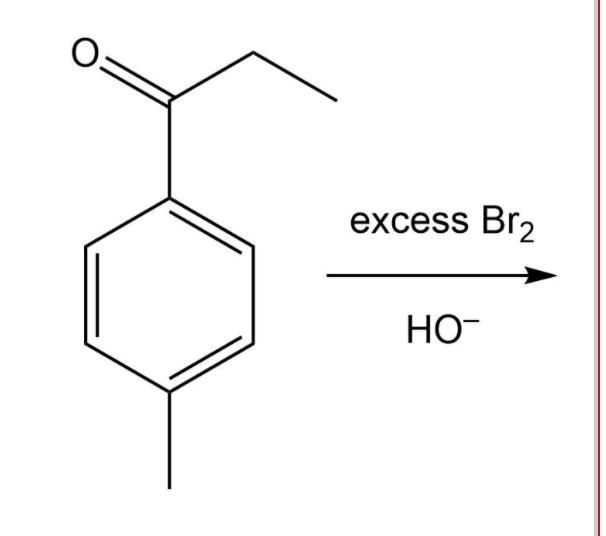
Q: Br + Br 2 ? (e)
A: We have give the IUPAC name of the product formed.
Q: Determine the unknown cation present in each test tube using the data tabulated below: Choices: A.…
A: We need to determine the cation present in each mentioned test tube, with respect to the tests given…
Q: hat is (are) the product(s) of the following reaction? 1. KMNO 2. H.О, Но- OH OH HO HO, HO, II IV…
A: Alkaline addition of KMnO4 in cyclohexene produce cis diol. This is called as syn addition
Q: Add Stoichmetric factors: ______Cr+ _______H20 yields ______Cr3O4+ +________H2
A:
Q: one of the following is primary standard for iodine O a. potassium hydrogen phthalate b. sodium…
A: Standardization is the process of determining the exact concentration of the solution. Primary…
Q: 2. Complete the following reactions: B. 2 Cu I THE H20, HCI
A: We have to complete the given reaction scheme. The required reaction scheme is shown below.
Q: Br*
A: Given,
Q: 1. hv + Br2
A: The given compound is an alkane. The alkane gives a free radical bromination reaction with bromine…
Q: Br Br
A: Here the first reaction involves the conversion of an alkyne to an alkane. The conversion of an…
Q: What is Kc for the reaction: Na2CO3 (s) + CO2 (g) + H20 (g) =2 NaHCO3 (s)? [NAHCO3]^2 / [Na2CO3] x…
A:
Q: CN CN base NC CN
A: A question based on oxidation reaction, which is to be accomplished.
Q: If the concentration of fluoride anion and aluminum cation was increased to 5 M, by how much would…
A:
Q: 4NH3(g) + 302(g) → 2N2(g) 6H20(g) с. Shift (PNH3) (Роз) (PN2) (Рнзо) K Addition of 02 Addition of N2…
A: Equilibrium constant depends on temperature and is independent of the actual quantities of reactants…
Q: How do I start balancing the following equation? K + ZnCl2 > KCl +Zn
A: Balanced chemical equation of a reaction is written according to law of conservation of mass.…
Q: a) Br b) но Br 'S
A:
Q: Draw the product of each reaction.
A: Reactions of amine with alkyl halide lead to alkylation of amine. In this alkylation are ends when…
Q: 1. ВН/ THF 1. CHзMgBr, A 2. Н2О2, ОН", H20 2. Н2О or HзО* Give all 2 products (2 pts each)
A: Alkene or alkyne reacts with borane, this reaction is called hydroboration reaction.…
Q: What is AH° for the reaction below? TICI4(9) + 2H20(1) --> TIO2(s) + 4HCI(g) Species TICI4(g) H20(1)…
A: Target equation : TiCl4(g) + 2H2O(l) = TiO2(s) + 4HCl(g) (1) Given that : Ti(s) + 2Cl2(g) =…
Q: When the following equation is balanced, what the coefficient of Cr(OH)3? Cr2(SO4)3 + – RbOH →_…
A: Given Reaction Cr2(SO4)3 + RbOH → Cr(OH)3 + Rb2SO4 Coefficient of Cr (OH)3 in…
Q: Br Br Br 二○
A:
Q: Br но
A: Molecule which have chiral centres will be chiral molecule But If any molecule have plane of…
Q: Br() "0. 2 Br(aq) + Reactants Products LIOH(s) "0. Litaq) + OH(aq) Reactants Products H20 FeF,(s)…
A: Since you have posted question with multiple subparts as per guidelines we can answer only three per…
Q: different reaction with the starting 1. Li (a) 2. Cul A 3. Br 1. Mg 2. H20 B (b) HO, PBr3 C SOCI2…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: Br NH2
A: Boiling point depends on the intermolecular forces of attraction present between the molecules .…
Q: Choose the correct ionic equation for the following reaction: Na2CO3 + Ca(OH)2 CaCO3 + 2NaOH Select…
A: Molecular Equation :- Na2CO3 (aq) + Ca(OH)2 (aq) ------> CaCO3 (s) + 2 NaOH (aq)
Q: HOW TO BALANCE : As2O3(s) + NO3-(aq) → H3AsO4(aq) + N2O3(aq)
A: Since in As2O3 we have As in 3+ state while in H3AsO4 As is in 5+ state Hence As is being oxidised…
Q: dissociate into 3 mole ions
A: Insoluble compounds doesn't dissociate into ions Strong acid fully dissociates into ions
Q: „Br но CI
A: Questions are based on the concepts of Organic reactions. a reactant is converted into another…
Q: Choose the correct ionic equation for the following reaction: NazCO3 + Ca(OH)2 CaCO3 + 2NaOH Select…
A: Given: Reaction of sodium carbonate with calcium hydroxide To find: correct ionic equation Solution:…
Q: How to balance H2NCONH2+H2O=NH3+CO2
A: A balanced equation contain same number elements on both side of the equation. Given equation is,…
Q: c) HCIO
A: In this question asked which is called hypochlorous acid, and four options are given.
Q: 3. Ba(OH)26) + heat -
A:
Q: Draw the product of each reaction.
A:
Q: *** O I + Z 1. Na° 2. dilute H30 Но OCH3 120
A:
Q: LIAIH, 12.
A: The given reaction is opening of epoxide of ring using LiAlH4 as a reagent.
Q: What type of reactions are the following reaction? 2 KCl(aq) + Pb(NO3)2 → KNO3(aq) + PbCl2(s)
A:
Q: Br Br но в
A: NBS is N-bromosuccinimide that is used for allylic bromination of alkene. This reaction follows free…
Q: ammonium carbonate + chromium (III) acetate Mol: Ionic: Net:
A:
Q: Br Br ОН
A: In the given reaction sequence two substituents on the benzene ring are attached to the para…
Q: SO3² is an amphoteric substance True False
A: Amphoteric substances are those who act as acid as well as base. According to bronsted lowry, An…
Q: Identify the oxidizing agent in the following reaction 2Na + 2H2O 2ѪOH + H2 A. Na B. H2O C. NaOH D.…
A: The addition of hydrogen or removal of oxygen is known as reduction and the addition of oxygen and…
Q: 8. Refer to Matching. OH 9. Refer to Matching. Br .coo-
A: The synthesis of the above given molecules are as follows,
Q: 7) 8) KHSO4(s) -> AgNO3(aq) + K₂SO4(s) + Na₂CrO4(aq) SO3(g) + — Ag₂CrO4(s) H₂O + NaNO3(aq)
A: The given reactions that need to balanced are - KHSO4s → K2SO4s + SO3g + H2O…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- What is the balanced equation for the combustion of C8H7NO2SBrCl in aC,H,N,S elemental analyzer?Which of the following is true for EDTA solution? A. It always needs to be adjusted (it needs to be standardized). B. Primer adjusted (standardized) against MgSO4 solution C. Primer is adjusted (standardized) against NaCl solution D. Needs to be set immediately after preparationBromine H2O + Cl2 H20 ----- __________. A. violet and reddish brown layered solution B. No reaction
- The zinc contained in a 0.7555-g sample of foot powder was titrated with 21.27 mL 0.01645 M EDTA. Find the percent Zn (MM=65.41) in the sample.Find the conc of free Mg2+ in 0.050 M Na2[Mg(EDTA)](i.e. MgY2-) at pH 9.00.what is the purpose of hydroxylamine hydrochloride in the spectrophotometric determination of iron in FAS
- The thiourea in a 1.455 g sample of organic material was extracted into a dilute sulfuric acid solution and titrated with 37.31 mL of 0.009372 M Hg2+ via reaction: 4(NH2)2CS + Hg2+ →[(NH2)2CS]4 Hg2+A solution of DNA (in TE buffer) gave an A260 - 0.245. The absorbance of the solution at 320 nm is 0.045. What is the concentration of DNA in the solution? a. 10ug/ml b. 20ug/ml c. 5 ug/ml d. 30ug/mlA 0.2185 g sample of NaCl was assayed using Volhard method using 50 mL of 0.998N silver nitrate and 11.9 mL of 0.1350N ammonium thiocyanate. Calculate the NaCl in the sample. MW NaCl = 58.45

