Q: 7.16 (a) What are the conjugate bases of the acids HF, [HSO,]¯, [Fe(OH,)J³+ and [NH,]+? (b) What are…
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first 3 sub-parts of…
Q: I = = = = + NH₂ Σ B Ph with the mechanism. H+
A:
Q: EXAMPLE 9-8 Calculate the hydronium ion concentration in a solution that is 2.0 X 10-4 M in aniline…
A: The Equilibrium Reaction of hydrolysis of C6H5NH3+ given in the question is - C6H5NH3+ + H2O…
Q: BrstcBry
A: Since we know that when Br2 react with alkene in presence of CBr4 it will give vicinal dibromides…
Q: A slightly soluble salt has the formula A, B, and a K of 6.7E-22. What is pB in a solution of this…
A: You have given - Ksp = 6.7 * 10-22
Q: Calculate AG° for the following reaction using AG°F values in your 2CH3OH(g) + 30O2(g) → 2CO2(g) +…
A: Given : We have to calculate the change in gibbs free energy for the reaction.
Q: when 268 des OF I FO₂ is diss 21 OF solution, it is found at Equilibrium Bat [OH-] = 2.13.-110-13…
A: Solution: In this we have to calculate acid ionization constant,
Q: Detemine the JOt|- pll, and pOHl of a solution with a H| of70x 10-" Ma 25 c. JOHF| = M pi = POH =
A:
Q: KOt-Bu Br OH HCI
A: The required reactions are
Q: A. How CO2 can be converted to value-added products mediated by ionic liquids such as CO2…
A:
Q: PalOAC), , K̟c02 ? + ? Nt-Buya 11:1
A:
Q: What is the AH° for the following reaction: H2O (1) + CH4 (g) --> CH;OH (I) + H2 (g) Given the AHf…
A: Consider the given reaction is as follows; H2O l + CH4 g → CH3OH l + H2 g The value of ∆H° for the…
Q: HA(aq) + OH(aq) - H,O(l) + A(aq) late the pKa value of the acid, and determine the number of moles…
A:
Q: If A=0.65 the molar absorptivity f(0.034) mol/lit. for a solution ,the = value of molar absorptivity…
A: The question is based on the concept of Lambert beer's law. It states that absorbance of a solution…
Q: If 35.09 of KCNIS added to 500mL of ater, what is the final Concentrahon of hydrocyanic acid? (Lšthe…
A:
Q: O'H Delete Reset H. C. be) (6)0 (s) 8. 7. 9. 1 2 3 4 5 CH,NH, Write the formula of the conjugate…
A: Applying concept of conjugate acid and conjugate base.
Q: Sample _0.IM NoCL pH (obtained by pH meter) = _4.38_ Reaction needed to predict pH: Equilibrium…
A: In an aqueous solution, the complete dissociation of a substance contains replaceable hydrogen to…
Q: ant adds lime (CaC mount of CaO) in
A: The decomposition reaction of CaCO3 is given as: CaCO3 ----> CaO + CO2 According to the balanced…
Q: 2+ hat will the concentration of Mg?t
A:
Q: 40 mg of sodium hydroxide is required to saponity Igm sample, this means that (MW of NaOH= 40…
A: Saponification value represents the number of milligrams of potassium hydroxide required to…
Q: 12. At 25 °C, What is the [OH] if [H*] = 0.1 M? Answer: Computation:
A: 12) Given, Temperature (T) = 25°C [H+] = 0.1 M [OH-] = ? The formula used here are, pH = -log[H+] pH…
Q: Calculate the ∆H of the following reaction. Show the complete and step by step solution. 1. LiOH(s)…
A:
Q: 2-14. Calculate the p-value for each of the indicated ions in the following: (a) Na*, Cl-, and OH-…
A: The p-value is calculated by the antilog of the concentration of the substance of interest. It can…
Q: B: What is the pH of 0.6 M NH.CI solution? Discuss your results. (K of NH= 1.8 x 105) NH. + H2O+ H3O…
A:
Q: I am getting the wrong amount of sig figs for each equation, even though I am using the smallest…
A: The mass of 1 mole of viruses is calculated as shown below,
Q: Kw = 10-14 What is the pH of a solution with hydroxide ion concentration equal to 10-4 M?
A: Given, Kw = 10-14 [OH-] = 10-4M Formula Used, pOH = -log [OH-] pH + pOH = 14
Q: 7-43. Supply the missing data in the table below. Molar Analytical Concentration, c (cr = cua + ca-)…
A: Given data:
Q: e. calculate A for the reaction 2 COte)+4 H2Ou 2 CHOHa) + 3 Ozte) f. calculate A CH3OHaj +2 Ozw ->…
A: To calculate ∆n of the reaction, we need to use the formula; ∆n = no. of moles of the product - no.…
Q: The exhaust of an tomobile co concentration in g/m3. Your Answer:
A:
Q: For a 0.0448 M aqueous solution of aniline, C6H5NH2 (Kb = 7.4E-10), determine the following. Kw =…
A: Given: The concentration of aniline, C6H5NH2 = 0.0448 M Kb for aniline = 7.4 × 10-10 Kw = 1.0×10-14…
Q: Find the concentration of Ni2+ in a solution of NiY2- at pH = 3.0. The formal concentration of NIY²-…
A: The equation for the complex formation is as follows. Ni2++Y4-→NiY2- The given values are as…
Q: 1- In the reaction of decomposition of H2O2; KI is a...
A: The decomposition of H2O2 in the presence of potassium iodide occurs as follows: 2H2O2 (aq) →KI (aq)…
Q: Q3: Calculate the concentration (M) for the solution of NaCI (M. wt. 58.51) that has w/ v% equal…
A: In the given question we have to calculate the concentration of the NaCl Solution. given that, w/v %…
Q: Pre-lab question #3-2: Consider the following equilibrium for nitrous acid, HNO2, a weak acid: HNO2…
A: When there is a balance between direct and reverse reaction, chemical equilibrium is achieved. The…
Q: the ph of a carbonate/bicarbonate mixture as 7.00 and the pka of the bicarbonate was measured as…
A: Given Information: pH of Carbonate (CO32-) to Bicarbonate (HCO3-) mixture = 7.00 pKa of Bicarbonate…
Q: Which response includes all of the following substances that have ∆Hf= 0, and no other substances?…
A: Concept- The enthalpy of formation is the standard reaction enthalpy for the formation of the…
Q: Which of the following is not used to be standardized against KHP? a. H2SO4 b. NaOH c. NH3 d.…
A: The following is not used to be standardized against KHP is given below.
Q: NO2 cormpound Value of melding pariny .paint Please write the name of the and write the take help…
A:
Q: Solution containing Ag*, Pb2+, and Cu2+ Add HNO3, so pH=1 Add H,S; Heat Agci %1D |X KoP Cus Y(aq)…
A: Solubility Product: It is the equilibrium constant for the solubilisation of a solid substance into…
Q: What is the enthalpy of solution of NaOH in kJ/mol? AHF[N2OH (s)] = -425.6 kJ/mol AH [NAOH (aq)] =…
A: Given, ∆Hf°[NaOH(s)]= -425.6 kJmol∆Hf°[NaOH(aq)]=-470.1kJmol
Q: Calculate the concentration of HS in an aqueous solution of 4.04x10 2 M hydrosulfuric acid, H,S…
A: Calculate concentration of HS- in the sample ?
Q: An aqueous solutio slowly added from that is 1.30×10-2 M а.) When the second i- the remaining conc…
A: The Solubility is defined as the maximum amount of solute that can be dissolved in a solvent at…
Q: meySi-I Cat. 2nlz Cet: DBu 5. e ther CH, C
A: We have to predict the product X for the following given reaction as follows in step 2:
Q: 9 what is the PH of a 2.5 M solutien ef CH, NH, CI ? CHgNHz pkb = 3.35) %3D
A:
Q: O Consider this acid-base reaction; CH3 CH₂ NH3 + H₂P CH3 = a) Draw the products. b) Draw the…
A: Note: Since you have posted a question with multiple sub-parts, we will solve the first three…
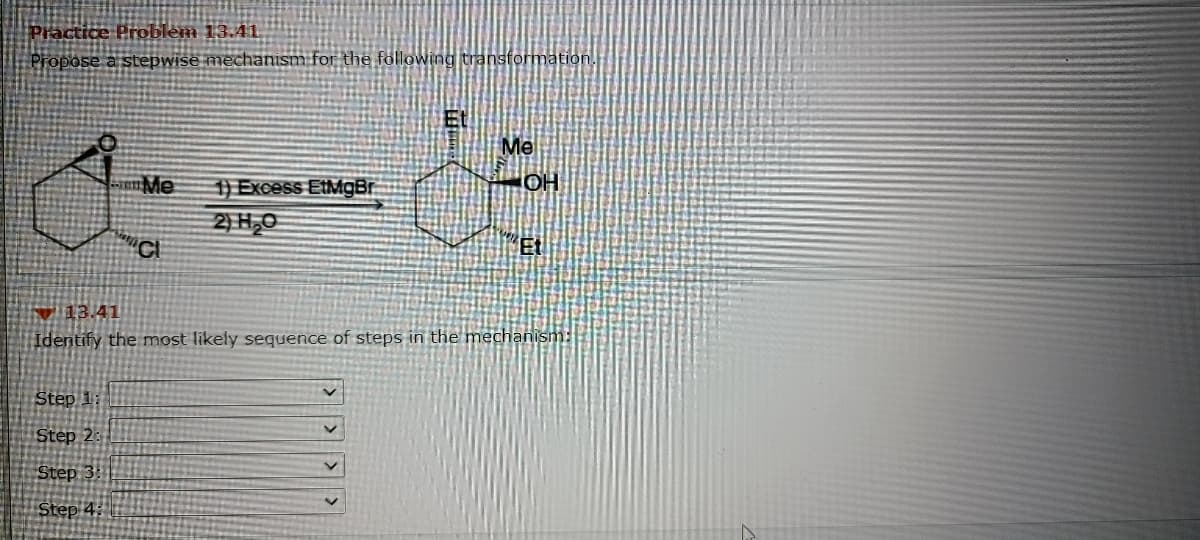
Q: Me. H Me a. b. С. H `CO,H Et Ci Ph, Me d. Me f. -CO,H е. Ph
A: The most common feature that leads to chirality in organic compounds is the presence of an…
Q: H-H (excess)
A: Addition of hydrogen in the presence of metal catalyst to alkene or alkyne is known as reduction…
Q: Assignment 1. Consider the acid-base reaction: H,CO,º → HCO; + H+ for which K = 10-6.35, Which way…
A:
Q: Calculate AG° for the reaction 3NO,(g) + H,O(I) --> 2HNO,(l) + NO(g). AG{(kJ/mol) H;O(1) HNO3(1)…
A: Please find the required solution in the attached image.
Q: Question attached
A: Given reactions, HSO4-(aq) + H3O+(aq) ↔H2SO4(aq) + H2O(l)NH3 + BCl3 → H3NBCl3
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Draw the structure of the desired dibromide product H. ) Propose a reasonable arrow-pushing mechanism for formation of the byproduct I.Use your mechanism to justify the stereochemistry in molecule I16. When cis-1,2-cyclopentanediol reacts with acetone in dry HCl, compound M is formed. What do you expect the hydrolysis of compound M to be when subjected (i) to base; and, (ii) acid a. resistant to base; resistant to acid b. resistant to base; readily hydrolyzed by acid c. readily hydrolyzed by base; resistant to acid d. readily hydrolyzed by base; readily hydorlyzed by acid 17. Following (16), would you expect that a similar compound be formed if the reactant was trans-1,2-cyclopentanediol? Why is this so? a. Yes; hydroxyl groups are on opposite side b. No; hydroxyl groups are on opposite site c. Yes; hydroxyl groups are on the same side d. No; hydroxyl groups are on the same sideWhile dehydrating 2-Methlycyclohexanol to 1-Methlycyclohexanol, and 3-Methlycyclohexanol 1)what is the common problem associate with E1 reaction? Discuss the intermediate. 2) Briefly compare and contrast E1 vs E2 Elimination reactions. INclude details such as speed of the reactions in relation to each other, rate-determining steps (E1), and stereochemical requirements (E2). Mention whether the steps of the reactions are reversible and discuss the conditions that can drive the equilibrium to favor product formation.
- 8. The preparation of the sex pheromone of the boll worm moth, (E)-9,11-dodecadien-1-yp acetate from compound A has been described. Suggest suitable reagents for each step in sequence.Benzene is one of the compounds used as octane enhancers in unleaded gasoline. It is manufactured by thecatalytic conversion of acetylene to benzene: 3C2 H2(g) ⇌ C6 H6(g). Which value of Kc would make this reactionmost useful commercially? Kc ≈ 0.01, Kc ≈ 1, or Kc ≈ 10. Explain your answerIn the following three compounds(1,2,3) arrange their relative reactivity towards the reagent CH3Cl / AlCl3. Justify your order?
- The following equation shows the bromination of compound 1. Propose a structure for product D and a curved arrow mechanism that accounts for its formation featuring the initiation, propagation and termination steps. can you also discuss the stereochemical outcome.Nitrating methyl benzoate. 1.Describe the mechanism of EAS clearly and in detail 2. What is the effect of subtituent of the outcome of EAS;include 3 classes of subtituentsWhen the following compound is heated, a product is formed that shows an infrared absorption band at 1715 cm-1. Draw the structure of the product
- γ-Butyrolactone (C4H6O2, GBL) is a biologically inactive compound that is converted to the biologically active recreational drug GHB (Section 19.5) by a lactonase enzyme in the body. Since γ-butyrolactone is more fat soluble than GHB, it is more readily absorbed by tissues and thus produces a faster onset of physiological symptoms. γ-Butyrolactone shows an absorption in its IR spectrum at 1770 cm−1 and the following 1H NMR spectral data: 2.28 (multiplet, 2 H), 2.48 (triplet, 2 H), and 4.35 (triplet, 2 H) ppm. What is the structure of γ-butyrolactone?1..Suggest mechanisms for the following reactions:Arrange the compounds in order of INCREASING reactivity towards bromination. I. Toluene, Nitrobenzene, Iodobenzene, Acetanilide II. Phenol, Acetophenone, Anisole, Chlorobenzene III. Aniline, Benzaldehyde, Benzonitrile, Benzenesulfonic acid

