Q: 3. Will the equilibrium re-established after one of your groupmates added an increment in the…
A: The position of equilibrium changes when we change the temperature of the reaction.
Q: b. If there are 0.400 moles of N204 and 0.500 moles of NO2 in a 1.00 L flask at equilibrium at 20°C,…
A:
Q: 35L of butene (CAH9) undergoes combustion at STP in the presence of excess oxygen. Write the…
A: Given, 3.5 L of Butane undergoes combustion of excess oxygen.
Q: ने ड.न० तोा oरी कबबक डि अकलबकदल it25.00 me vimegुa ड अकनबनटल to नमह एदपांपवोलत्e Paोकर्व पयंने…
A:
Q: 16 17 18 19 20 21 22 23 24 = 25 26 27 15 The rate at which a certain drug is eliminated by the body…
A: In case of first order reaction : Half life time = 74 minutes. Initial concentration = 0.16 μg/ml.…
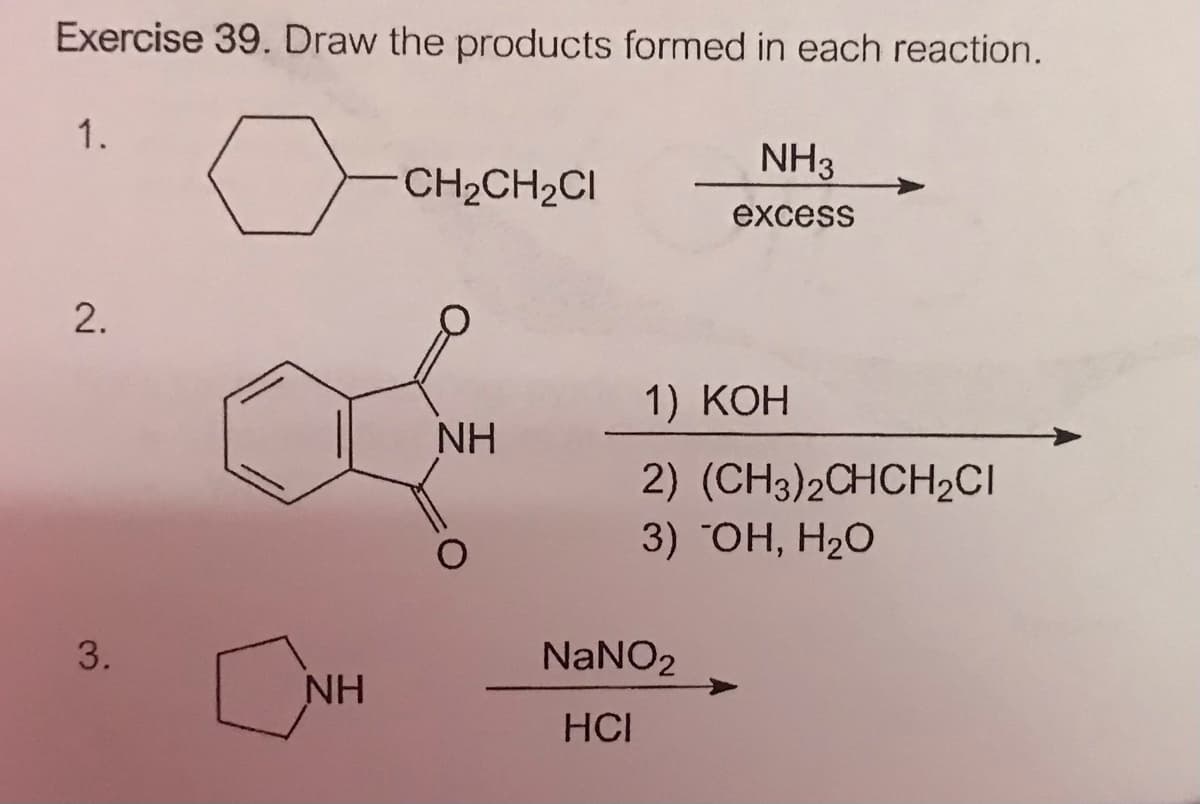
Q: Propose a synthesis for the following transformation. OEt H' HO,
A:
Q: A chemist titrates 50.0 mL of a 0.5634M acetic acid (HCH, 3 acetic acid is 4.76.
A: at equivalence point Millimoles of acid = milli moles of base Salt of Weakacid with strong base…
Q: Convert Molarity to Normality or vice versa as appropriate. a. 1.50 N Sr(OH)2 b. 2.0 М НзРОа 6.00 N…
A: Here some solutions are given with their concentration either in Molarity Or in Normality. We have…
Q: QUESTION 15 Which of the following is diamagnetic? O 13AI 15P 24Cr 48C. 83BI QUESTION 16 Which ion…
A:
Q: Consider the following equilibrium reaction: 2NO2(g) = 2NO(g) +O2(g) In a container held at 375. K,…
A:
Q: 21-25. Determine the boiling point of a solution of 10 grams of a substance of molecular weight 94…
A:
Q: For each solute, click the button under the better solvent. solute Which is the better solvent?…
A: Solubility of different solutes in different solvents
Q: CH3 CH3 OH HO, CH3 H3C CH3 H3C `CH3 H3C (-)-menthol (--menthone (+)-neomenthol Hi, Could you please…
A: A stereogenic center is defined as the atom in the organic structure where changing the orientation…
Q: Acetylenic fatty acids are found in many tropical plants. Compound H, C13H2502, is one such…
A: Note : Compound H absorbs 5 equivalents of hydrogen. Therefore compound is have two triple bonds and…
Q: 8. H2N-NH- CH3 RÖOOH 10. 1. NaBH4 2. H20 9.
A: In this question we have to tell the product of the reaction.
Q: A. Calculate the molar solubility of CaF, in a 0.1450 M cesium fluoride, CsF solution. Solubility =…
A:
Q: Find the AH for the reaction below, given the following reactions and subsequent AH values: H,SO ()…
A: ∆H of a reaction depends on Stoichiometry of reaction If a thermochemical equation is reversed…
Q: Consider the given octahedral compounds: 1. Which of the compound/s do NOT have a Cz axis of…
A:
Q: 7.67 Ammonia may be used as the anodic reactant in a fuel cell. The reactions occurring at the…
A: Given reactions are : NH3 (g) + 3OH- (aq) -----> 1/2 N2 (g) + 3H2O (l) + 3e- O2 (g) + 2H2O (l)…
Q: CH3 NaNH, ? CI liquid NH3 A) CH3 CH3 B) CH3 CH,NH2 CI -NH2 -NH2 NH2 C) CH3 D) CH3 CH3 CH3 -NH2 +…
A:
Q: Identify the followving amino acid and specify its configuration: D or LandR or S. ball & stick V -+…
A: The amino acid is Histidine. Its configuration is D (more electronegative nitrogen on right side).…
Q: SATURATED HYDROCARBON Worksheet No. 1 I. Give the name and condensed formula of the following: H HH…
A: Condensed formula is that which is written by considering only atom not bonds between them thus all…
Q: 1. Tungsten metal, W, is used to make incandescent bulb filaments. The metal is produced by a…
A: First write the balanced reaction between WO3 and H2. Using that reaction find mass of W or tungsten…
Q: A 1.75 mol sample of an ideal gas is allowed to expand adiabatically and reversibly to twice its…
A:
Q: For a particular reaction at 2504 °C. AG=-1294.49 kJ, and A.S = 545.75 J/K. Calculate AG for this…
A:
Q: Calculate the actual cell potential if you have a 1 M solution of MgSO4, but only a 0.5 M solution…
A:
Q: Specify the formal charges (both sign and magnitude) on the atoms labeled a-c. a H,C-CH2- 1) a b :o:…
A: In this question, we have to write the formal charges on the atoms labelled as a-c.
Q: A chemist fills a reaction vessel with 8.77 atm methanol (CH,OH) gas, 3.65 atm oxygen (0,) gas, 2.21…
A:
Q: POLYMER 1 molecular mass (g) weight |(g/mol) 50000 9.5 25000 8 3000 13.25 10000
A:
Q: What volume (in mL) of 0.350 M HClO4 is required to prepare 1.5 L of 0.150 M HClO4?
A:
Q: Create a timeline that shows the history of the development of periodic table of elements
A: Periodic table is a table of the chemical elements arranged in order of atomic number, in rows and…
Q: cid is used to titrate an unknown base, and the pH of solution is recorded during the titration.…
A: Solution: In acid-base titration, the main advantage is we can calculate unknown concentration if…
Q: he freezing point depression?
A:
Q: How many minutes will it take to
A:
Q: A В 1. Н* 2. МеОН C HO
A: Alkene reacts with alcohol in presence of an acid catalyst by an electrophilic addition reaction…
Q: e1/K B. -3.232 J/K C. +2.308 J/K D.) -2 n of the following is true for isothermal expansion of gas…
A: 7) mole(n) = 1 mol T1 = 298 K T2 = 333 K □S =2.303 nCv.log(T2/T1) = 2.303 × 1 × 2.5 ×8.314…
Q: A. Identification. Name the organic structural formula of the following carbon chains below. CH3…
A: In this question we have to tell the name of the compounds.
Q: Why is it necessary to have different methods for appl
A:
Q: When the Pb2+ concentration is 1.02 M, the observed cell potential at 298K for an electrochemical…
A:
Q: there are 0.400 moles of N2O4 and 0.500 moles of No2 in a 1.00 L flask at equilibrium at 20°C, what…
A: Given- Moles of N2O4=0.400 mol Moles of NO2=0.500mol Volume=1.00L Temperature=20°C Keq=?…
Q: Calculate the pH at 25 °C of a 0.26M solution of sodium benzoate (NaC,H¸CO,). Note that benzoic acid…
A: Given, Temperature (T) = 25°C Concentration of sodium benzoate (NaC6H5CO2) = 0.26 M pKa of benzoic…
Q: Which of the following combinations of quantum numbers is permissible? A)n = 1, l = 2, ml = 0, ms =…
A: For a given principal quantum number n , l values : 0 to (n-1) ml values : -l to +l ms values :…
Q: Question 8: Calculate the number of protons, neutrons and electrons in the following atoms. Element…
A:
Q: two are: ule punds t isomers isomers FH,CH, H- -CH, CH- H- CH, CH, CH,CH,
A: Isomers having same molecular formula but different molecular structure and spatial arrangement…
Q: Identify the oxidizing agent, the reducing agent, the substance being oxidized, and the substance…
A: Oxidising agent is the substance which oxidises other species and reduces itself. Reducing agent is…
Q: Consider this reaction: H,CO, (ag) – H,0 (ag) +CO, (aq) At a certain temperature it obeys this rate…
A: The rate law of reaction states that the reaction is a First-order reaction. The rate of the first…
Q: Page fropase an efficient synthesisto.comvert the aformentioneolmimoryamine.into Lithe tertiany mne…
A:
Q: How long will it take to plate out 3.0 g Ni from aqueous Ni2+ with a current of 113.1 A? HOW DO WE…
A: Given:: Mass of Ni=3.0 g molar mass of Ni = 58.6934 g mol-1 Current i=113.1 A Time, t = ?
Q: 4. Steam activation is carried out using steam at temperatures of between - 800°C and 1000°C. At…
A: In this question, we have to write True or False regarding the given statements.
Q: Please provide a reasonable mechanism for the following transto the stereochemistry of all…
A:
Step by step
Solved in 4 steps with 2 images

- 1. James put the battery on the freezer. Will it prolong the batter life or not? Why? 2. What is photocatalyst and how can we prevent it? Answer 1 and 2Ochem prelab question I"ve attached part of the lab, but basically we are reducing m-nitroacetophenone to m-aminoacetophenone using granular tin. I've also attached one of my workups for the prelab, which require us to show the half reaction equation for reduction, the half reaction equation for oxidation and the overall balanced equation. Then determine the limiting reactant and theoretical yield Can you please check my work... especially my half reactions and see if those are correct (I'm not sure about the added e- and H+ Also, is the limiting reactant tin or should it be m-nitroacetophenoneHello! quick question, when a chem problem starts with "refer to the unbalanced equation below" , and theres a unbalanced equation below (lol), do i have to first balance the equation first and then solve for problem? or am i supposed to just solve assuming its a 1 for the coefficent of each reactant and product? thanks in advance!
- 18.A simple substitution or displacement reaction can be represented by the reaction. An example of substitution or simple displacement reaction is?Necessary answer. Unique option. A) CaCo3---------->CaO + CO2 B) HCl + NH3------>NH4Cl C) Cu + H2SO4 -----> CuSo4 + H2 D) S +¿ O2----------->SO2 E) None of the above.jorge was given an equation and told to predict the products. Her teacher marked her answer wrong. What is the error and what should her answer have been? Problem Jorge's Answer A1+Na2SO-->??? Na2+A1SO4Problem #10 on my review sheet
- Fun fact: ΔHrxn for all strong acid + strong base reactions is approximately-58 kJ mol-1. Explain this result based on what you know about strong acid andstrong base reactions. (write the net reaction for a few of these and compare them)also answer y product 2 is formed instead of product 3exercise:determine the product/major product in the following. answer only.

