Q: Draw and explain the acetylation reaction of methyl salicylate. Give an explanation of the reaction…
A: acetylation can be done by reaction of methyl salicylate with derivative of acetic acid.
Q: Explain briefly why sulfur mustard is soluble in the lipid bilayer membranes that surround human…
A: Sulfur mustard has atomic charge polarization. Also, it has smaller molecular volume which makes it…
Q: Explain in detail the structure of Lactic Acid.
A:
Q: Draw a structure for cycloheptanecarboxylic acid
A: The structure of the chemical compounds gives the 3-D structure of the bonds present in the molecule
Q: Write a general reaction showing the hydrolysis of anester using an acid catalyst.
A: Hydrolysis: When the water molecule is added to any reaction in the presence of acid or base is…
Q: draw the structure of; terephthalic acid
A: Terephthalic acid is an organic compound having chemical formula C6H4(CO2H)2. It is the common name…
Q: Compare and contrast dehydration and hydrolysis reactions
A: Both dehydration synthesis and hydrolysis reactions play important role in the synthesis of…
Q: Draw the structure of 4-hydroxypentanoic acid
A: 4-hydroxypentanoic acid is a carboxylic acid-containing carbon compound and substituent -OH at the…
Q: what is the name of this structure?
A:
Q: What chemical test will you use to differentiate acetone from dibenzalacetone?
A: In this question, we will used Iodoform chemical test. This test is used to differentiate between…
Q: Explain Reduction of Aldehydes and Ketones ?
A: The addition of hydrogen is called a reduction reaction. Hydrogen may add in the form of a hydride…
Q: Draw the general structure of a purine.
A: Purine is one of the heterocyclic aromatic compounds which basically constituted from aromatic…
Q: draw the structure of dichloroacetic acid
A: Dichloroacetic acid having chemical formula C2H2Cl2O2 is a derivative ( analogous) of acetic acid.…
Q: triethylammonium chloride structural formula
A: In structure of triethylammonium chloride , there are three ethyl groups attached to Nitrogen atom…
Q: What is an cyclic anion?
A: Abstraction of a proton from a cyclic compound lead to the formation of a cyclic anion.
Q: Describe preparation of dibenzalacetone
A:
Q: The compound above is NOT a. a carcinogen b. soluble in benzene c. an aromatic compound d. soluble…
A: The given compound, anthracene with molecular formula C14H10, is an aromatic compound and most of…
Q: What do you mean by decarboxylation?
A: Given,Decarboxylation reaction
Q: Vanillin
A: CONDENSED STRUCTURAL FORMULA :- It is a more compact way of drawing the structural formula of a…
Q: What is/are true regarding the following compound? * СООН IUPAC name is 4-Carboxy-2- pentylbutanoate…
A:
Q: What is the difference between alkyl dimethyl benzyl ammonium chloride and hydrogen peroxide used in…
A: A question based on disinfectant, which is to be accomplished.
Q: What is the name of this structure?
A: To write the name of given compound we have to follow the given steps . (1) Identify the longest…
Q: Explain the ilyl ether ?
A: Silyl ethers are functional groups with Si covalently bonded to an alkoxy group.
Q: What is the condensed structural formula for the products obtain from the hydrolysis of ethyl…
A: To find: The condensed structural formula for the products obtained from the hydrolysis of ethyl…
Q: What is the oxidation number of carbonyl carbon in methanal and methanoic acid?
A: The oxidation number of carbonyl carbon in methanal can be calculated as follows:
Q: What is the major advantage of base-catalyzed hydrolysis of esters over acid-catalyzed hydrolysis?
A:
Q: What is the structure of benzoic formic anhydride
A:
Q: draw the structure of benzoic acid.
A: Compounds containing benzene ring in it are termed as aromatic compounds.
Q: Draw a structural formula for the keto form of enol
A: The two forms i.e., enol and keto forms are constitutional isomers of each other. A compound…
Q: What is the equation for the dissociation of a carboxylic acid in water?
A:
Q: Which is true of the reactivity of aldehydes c
A: C=O group is called carbonyl group , In organic chemistry with this carbonyl group two functional…
Q: Why conversion of ethanol into ethanoic acid is an oxidation reaction?
A: Oxidation Reaction is a reaction of carbon compounds with oxygen. Basically, oxygen atom is added in…
Q: Discuss the biochemical effects of acute vs. chronic ethanol intoxication
A: Biochemical effects of acute ethanol intoxication: 1.) Plasma [Mg2+] will increase. 2.) Plasma…
Q: What was formed in the autooxidation of benzaldehyde?
A: Benzaldehyde on autoxidation form benzoic acid.
Q: An alcohol undergoes dehydration in the presence of a catalyst to form and .
A: alcohol upon reaction with protic acids tends to lose a molecule of water to form alkenes these…
Q: Benzo(a)pyrene is a well known carcinogen. Based on its chemical properties, explain why…
A: Solution - Benzo(a)pyrene - Benzo[a]pyrene is a polycyclic aromatic hydrocarbon and the result of…
Q: what is the effect of Terephthalic acid in environment?
A: Terephthalic acid is a sand-like white powder. Terephthalic acid is made by oxidation of xylene.
Q: Explain the acidity of carboxylic acids ?
A: Acids are substances which give H+ ion in solution. Lewis acids are electron acceptors. Carboxylic…
Q: Arrange in increasing polarity: Propanol Propylamine Propanone Propanoic acid
A: The polar molecules are those that have ac permanent dipole moment and, the polarity in the molecule…
Q: Provide the chemical properties of acetone
A:
Q: Which of the following functional groups can react with a fatty acid to form an ester bond? A) amine…
A: Answer is B) alcohol
Q: What is the structure of butyl acetate?
A: Introduction: Molecular structure shows the arrangement of atoms in a space. The organic structures…
Q: What are the relative solubilities of benzoic acid and urea in water/and in denatured alcohol?
A:
Q: Define about Reduction of Acid Chlorides and Esters ?
A: Reduction of carbonyl compounds : The reduction of carbonyl compounds like aldehydes, ketones, acid…
Explain the General Features of Dehydration in Acid ?
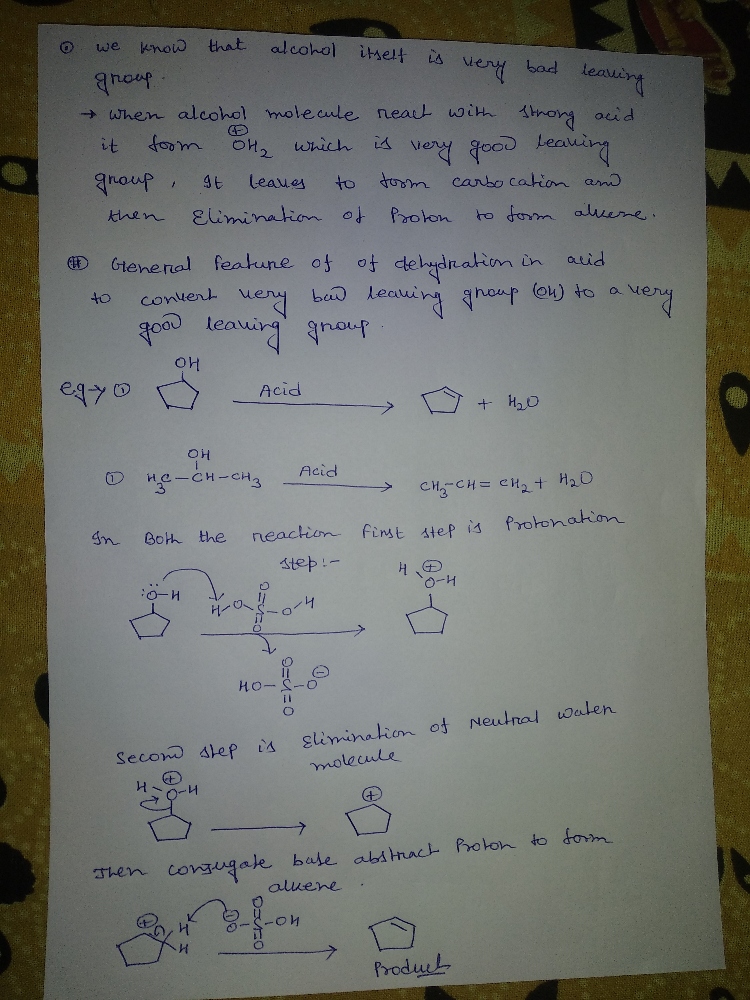
Step by step
Solved in 2 steps with 1 images
