Q: Which of the following factors that affect rate of reaction applies only to heterogeneous systems?…
A: The factor which affect rate of reaction of heterogeneous systems : • heterogeneous system is the…
Q: How many grams of silver metal will be deposited from a solution that contains Ag+ ions if a current…
A: Amount of current passed through the solution containing Ag+ ions = 0.695 A Time of this current…
Q: What comes to mind when you think of the terms “system” and “surroundings”? Provide 2-3 examples of…
A: In the study of chemical reactions and processes, the terms "system" and "surroundings" play a…
Q: 200000 Time--> Abundance 30000- 25000 20000 15000 10000 5000 m/z--> 3.00 4.00 0TTT 40 53 81 5.00 109…
A: The question is based on the concept of mass spectroscopy . we need to assign characteristic…
Q: 1. Indicate the product which will be formed in the following reaction. OH T CI EtOH
A:
Q: 2. Devise a synthetic strategy to complete the following transformation. b. НО j→ m CN
A: The given reactions are shown below We have to carry out the above transformation.
Q: major organic product? XO Et "OEt 0 A NaOH/H₂0 H30°, 4.
A: Esters undergo basic hydrolysis to give carboxylic acids. If a stable carboanion can be formed by…
Q: 7 A 1 M solution of AgNO3 is placed in a beaker with a strip of Ag metal. A 1 M solution of Cu(NO3)2…
A: (a). Prediction of which electrode serves as the anode and which as the cathode :- The electrode…
Q: The table is missing the 6th Carbon
A: To analyse the structure and construct a table depicting chemical shift values of each carbon .…
Q: The reaction of sodium with chlorine to produce table salt, sodium chloride, has a ΔH = -411 kJ/mol.…
A: Reaction of sodium and chlorine to produce sodium chloride, with ΔH = -411 kJ/mol. Inital…
Q: Draw the product(s) 1. NaNH2 2. sec-butyl iodide 3. Na, liquid NH3
A:
Q: A flask at room temperature contains equal numbers of di-nitrogen molecules and krypton atoms. (a)…
A: The question is based on the concept of gaseous state . we need to identify the gas having higher…
Q: Naming ionic compounds with common polyatomic ions Fill in the name and empirical formula of each…
A: Several cation and anions are given in this question. Hence it is possible to write ionic compounds…
Q: 7) Propose a synthesis. H D
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: A carbonyl compound has a molecular ion with a m/z of 86. The mass spectra of this compound also has…
A: A carbonyl compound is a class of organic compounds that contain a carbonyl group, which is a…
Q: Select the product that would form from the following propagation step: ind xx
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: Which of these answers is not a correct match between a measurement parameter & its standard unit?…
A:
Q: The pH of a bicarbonate-carbonic acid buffer is 5.63. Calculate the ratio of the concentration of…
A: pH = 5.63 NOTE: please note as pKa value is not provided , I have used standard value. if your…
Q: According to valence bond theory, there are several different numbers of bonds that phosphorus could…
A: Valence bond theory takes into account the number of valence electrons or electrons present in the…
Q: Consider the following equilibrium: 2NH, (g) N₂(g) + 3H₂(g) AG-34. kJ Now suppose a reaction vessel…
A: Given reaction is the formation reaction of Ammonia and we have to find out the direction of…
Q: Calculate the [H3O+] of an aqueous solution that is 1.5 × 10 Min OH. Enter your answer in scientific…
A: Given, [OH-]= 1.5 *10-5M
Q: NO₂ Br j b k OH 1 010 C=N 910 m
A: A question based on nomenclature. Several structures are given and the ester structure among them is…
Q: Complete the balanced dissociation equation for the compound below in aqueous solution. If the…
A: When an ionic compound /polar compound or a salt is dissolved in water it dissociate into it's…
Q: Lead(II) nitrate and ammonium iodide react to form lead(II) iodide and ammonium nitrate according to…
A:
Q: n = 6559 CH4 → moles 40.84 0.0821 R= T= 25% +273 = 298 K X 16. Methane, CH4, is being considered for…
A:
Q: What is the hybridization of the central atom in each of the following: Drag the appropriate items…
A:
Q: Consider the reaction CO(g) + 3H₂(g)- →CH4(g) + H₂O(g) Using the standard thermodynamic data in the…
A:
Q: When isobutylene undergoes radical-initiated polymerization, which of the following would be formed…
A:
Q: If the recorded mass of a metal block is 78.180 grams, how many significant figures are in that…
A: Significant figures are the digits in a number that indicates reliability of a measurement
Q: Br 1 Mg ↑
A: The question is based on the concept of organic synthesis. we need to synthesize the product using…
Q: For the reaction 2CH4 (g) + 3 Cl2 (g) → 2 CHCl3 (l) + 3 H2 (g), ΔH° = -118.6 kJ. ΔH°f = -134.1…
A: a balance chemical equation, 2CH4(g) + 3Cl2(g) -----> 2CHCl3(l) + 3H2(g) ∆Ho = -118.6 kJ &…
Q: a. H y" nuc: fast b. nuc: slower
A: The carbonyl group in carbonyl compounds in polar in nature due to the difference in…
Q: Analyze the IR for the molecular formula C8H8O3 (vanillin). List only the relevant absorbancies (it…
A: Question is based on the concept of IR spectroscopy. we need to analyse the spectra.
Q: Please explain, give correct answer and typed answer only no AI generated answer please
A: For the Fischer esterification reaction, The volume of the isobutanol used is Valc = 0.75 mL. The…
Q: Identify the reagents necessary for the following hydrolysis. OMe NH3 ROH, pyridine O [H+], MeOH O…
A: In hydrolysis, a water molecule splits into a hydrogen ion (H+) and a hydroxide ion (OH-), and these…
Q: Answer the questions in the table below about the shape of the chlorine pentafluoride (CIF)…
A: ClF5 forms one sigma bond with each fluorine surrounding it . In excited state it can form 5 bonds…
Q: Analyze the CNMR for the molecular formula C8H8O3 (vanillin). Be very specific in explaining why the…
A: To predict that the given 13C NMR spectrum is for the compound, Vanillin, C8H8O3. The structure of…
Q: dentify whether each species functions as a Brønsted-Lowry acid or a Brønsted-Lowry base in this net…
A:
Q: For the reaction 2Co³+ (aq) + 2Cl(aq)-2Co²+ (aq) + Cl₂(g). E = 0.483 V what is the cell potential at…
A:
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: This question belong to Redox reactions that is Balancing of Redox reaction. We balanced the Redox…
Q: If you have a complex ion in equation solution in the solution is yellow, what is the approximate…
A: Using a color spectrum wheel, it is possible to determine what color a solution will appear based on…
Q: The nucleophilic reaction between phenyl a-disulfone and hydrazine occurs according to the following…
A:
Q: A 3.5 mol sample of an ideal gas is compressed from 20.8 L to 12.5 L. What is its change in entropy…
A:
Q: A volume of 105 mL of H₂O is initially at room temperature (22.00 °C). A chilled steel rod at 2.00…
A:
Q: Question 42 Why does the vapor pressure of water decrease when we add NaCl to it? A The ion-dipole…
A: The pressure exerted by vapours of a liquid above it liquid surface is called vapour pressure.
Q: 14. The compound CH3NH₂Br contains the active ion CH3NH3*. A solution from a bottle labeled 0.0493M…
A:
Q: Write the net cell equation for this electrochemical cell. Phases are optional. Do not include the…
A:
Q: Name the functional groups present in a fructose molecule and the link to water solubility
A: Fructose is a monosaccharide, a simple sugar, with the molecular formula C6H12O6.
Q: Acetanilide (C6H5NHCOCH3, M = 135.2) can be prepared from aniline (C6H5NH2, M = 93.1) and acetic…
A: The experimental yield of acetanilide = 10.0 g The given mass of aniline = 10.0 g The given mass of…
Q: The standard cell potential E°(cell) is +0.77 V for the redox reaction 2 Fe³+ (aq) + H₂(g) → 2…
A:
Explain the step by step process occruing here
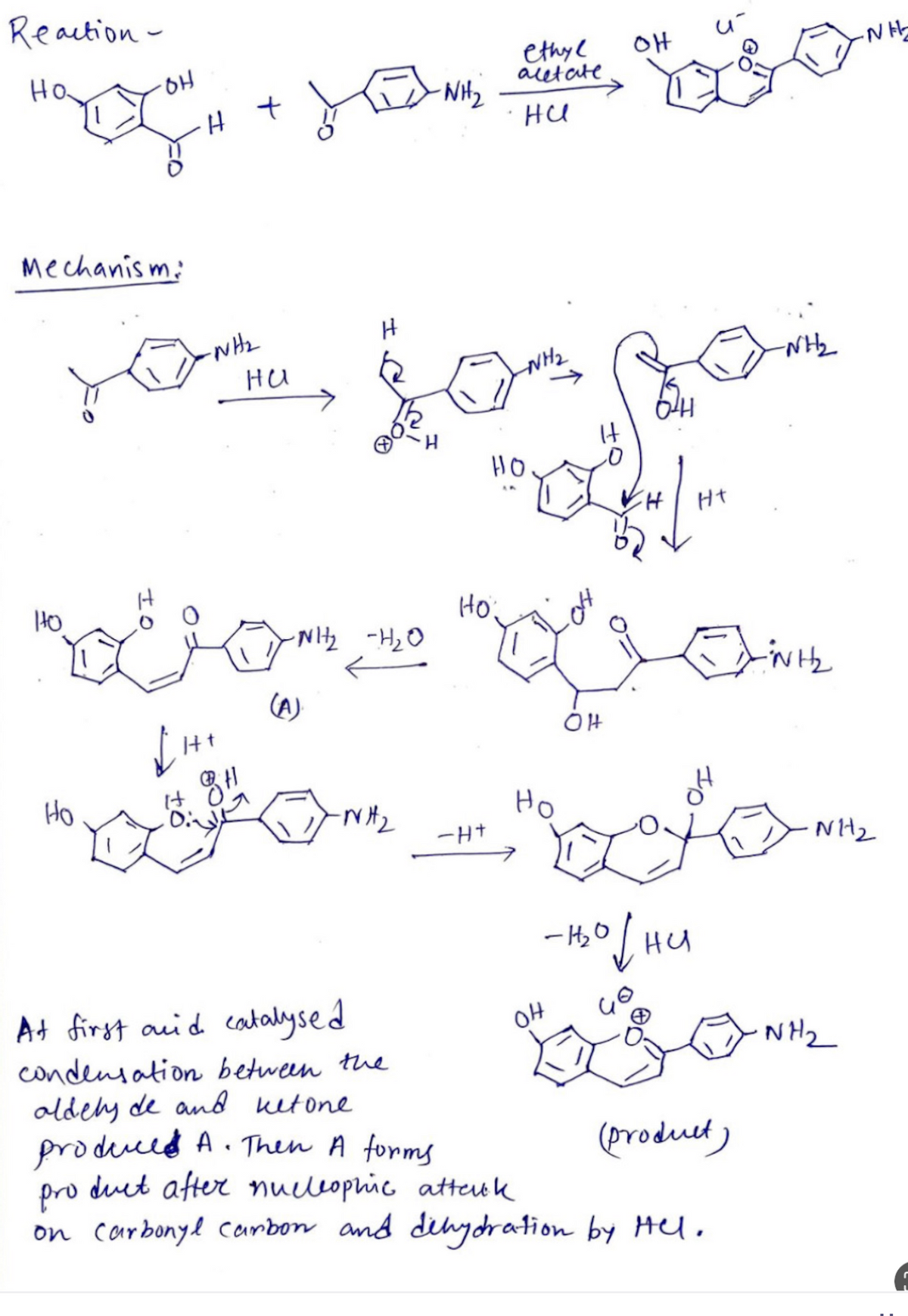
Step by step
Solved in 4 steps with 1 images

- The iodination of acetovanillone (1-(4-hydroxy-3-methoxyphenyl)ethan-1-one) occurs in the following reaction: C9H10O3 + NaI + NaOCl --> C9H10O3I + NaCl Draw the complete reaction mechanism of acetovanillone including curved arrows.Predict the product(s) and provide the complete mechanism for each reaction below.What steps are needed to prepare phenylacetylene, C6H5C = CH, from each compound: (a) C6H5CH2CHBr2; (b) C6H5CHBrCH3; (c) C6H5CH2CH2OH?
- The enolate derived from diethyl malonate reacts with a variety ofelectrophiles (not just alkyl halides) to form new carbon–carbon bonds.With this in mind, draw the products formed when Na+ −CH(CO2Et)2reacts with each electrophile, followed by treatment with H2O.Draw reaction mechanisms with all reactants, arrows, intermediates, and products. Your mechanism must account for all the products if more than one product is formed. 4-methycyclohexanol with phosphoric acid H3PO4 to for 1-methycyclohexene, 3- methylcyclohexene and 4-methycyclohexeneThe Favorskiireaction involves treatment of an a-bromo ketone with base to yield a ring-contracted product. For example, reaction of 2-bromocyclohexanone with aqueous NaOH yields cyclopentanecarboxylic acid.what is the mechanism of the reaction.
- Propose a synthesis strategy for the following. Show complete reaction mechanismIf bromobenzene is treated with HNO3/H2SO4 the major product should be 4-nitrobromobenzene. Please show the complete mechanism for the reaction and include any intermediates. Thanks!Drawing Hydrohalogenation with a Carbocation RearrangementDraw a stepwise mechanism for the following reaction.
- Write the products for (A-I). Label products with the mechanism (Sn2, Sn1, E1, E2) that produced it and write out and label major vs minor elimination products.The following compound undergoes Benzilic Acid Rearrangementto yield ahydroxyacid salt. Proposea mechanism for the reaction, writethe major product,and provide an explanation as tothe preference of migration of one R group over the other.Draw a stepwise mechanism for the attached reaction that illustrateshow two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reactionconditions, even though it is a 1 ° alkyl halide.

