Q: Translate the following reaction completely into English words using no symbols: 4NH3 + 5O2 -->…
A: Translate the following chemical reaction into english words using no symbols : 4NH3 + 5O2 --->…
Q: Consider the reaction below, the thermodynamically controlled product in this reaction is Br Br HBr…
A: Ans a Reaction takes place via free radical mechanism
Q: If exactly 3 moles of propanol (C3H8O) is combusted in the presence of excess oxygen, how many moles…
A:
Q: The product distribution of the reaction below will consist of... QH HBr, A Br 4. Compound A…
A: Alcohol reacts with an alkyl halide to form an alkyl chloride. The reaction proceeds with an SN1…
Q: 3. Give the reactants for the following products. а. H- -H b. CH,CH, Br с. CH3 H;C. Br HBr
A: Reactants of following products
Q: 2. Write the name(s) of the product(s) for the following reactions. If no reaction can occur, write…
A: Here we are required to find the product of the reaction.
Q: What is the product distribution of the reaction below? Br2 Br (A) (B) hv (C) (D) Br almost…
A:
Q: (g) 1) OsO4 2) NaHSO3
A: Alkenes can be converted to diols by treating with OsO4 and KMnO4 . Both reagents gives cis-diols.…
Q: 1. O3 2. Zn
A: The above reaction is the Ozonolysis in which O3 (ozone) reacts with double in the presence of Zn…
Q: Predict the products of oxidizing the molecules shown in this problem. In each case, identify the…
A: a.
Q: Cl2 (low conc.)
A:
Q: Give the minor product of the following reaction. CO,Me heat CO,Me CO,Me cO, Me cO, Me co, Me H.…
A:
Q: 1. Br2, hv 2. NaOCH3 NaBH4 МеОн
A: o Br2 in light give benzylic bromination product o NaOCH3 acts as a base o NaBH4 in methanol reduces…
Q: Develop a general equation for the standard heat of reaction as a function of temperature for one of…
A:
Q: Consider the reactions below. In which cases is product formation favored by decreased temperature?…
A: When energy is released in an exothermic reaction, the temperature of the reaction mixture…
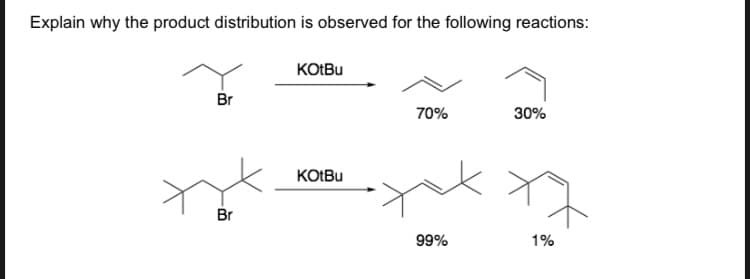
Q: Explain why the product distribution is observed for the following reation: Explain why the product…
A: The reaction of alkene with HBr proceeds via carbocation intermediate. As carbocation is planar in…
Q: For the reaction run at room temperature, compound I is formed. However, over prolonged reaction…
A: Here we have to determine the correct statement among the following from the given Diel's alder…
Q: нн (а) Н—Ҫ—С—О—Н— Н-С—о—ҫ—н H H H H Ethanol Dimethyl ether но H (b) H-C-C-H → H-C-C-H I I H H…
A:
Q: Give the major product(s) of the following reaction. 1) KMNO4, OH", heat ? 2) Hзо A OH В но
A:
Q: Draw structural formulas for the major organic product(s) of the reaction shown below. CN + Cl₂…
A: CN is an deactivating group which withdraw electron density from the ring,so the reaction will occur…
Q: d. HO, CO2H 1. LIAIH4 2. H3O* е. HO, PCC f. OH PPH3
A: Predicting Reagent , Product for the multiple Organic Transformation.
Q: NON H. H,N a) HC CH3 b) H,C CH,
A: Organic reaction mechanisms
Q: For the Reaction Scheme below provide the following (i) list all the reagents and conditions needed…
A: The reaction shows the formation of…
Q: Predict the product/s and write the structural formula for the organic product/s of the following…
A:
Q: 3. Give the product for the reaction below. H-Br
A: The question is based on the concept of organic reaction. this is an electrophilic addition…
Q: Which of these statements is true about this reaction coordinate diagram for the reaction of A with…
A: Since the energy of reactant is greater than the energy of products, the reaction is exothermic.
Q: Tell whether each reaction favors product-formation or reactant-formation. A. 2NO) + O2e)= 2NO2) K,…
A: We write partial pressures of gaseous products and gaseous reactants in the expression of Kp.
Q: (C=C) (C-H) (O-H) (C-O) (C-C) (C=0) (CI-CI) (Н-Н) (C-CI) (Н-C) 614 413 463 358 348 799 242 436 328…
A:
Q: 1) HNO,, H2SO4 2) Br2. FeBr3 3) H2 / Pd Br- NO2
A: 1. Nitration of acetophenone:
Q: What is the major product produced in the given reactions? A 1. В D 1. Mg ,Et,0 2. H,C CH, major…
A:
Q: What is the product distribution of the reaction below? (1) O3/CH2C2, -78°C (2) (CH3)2S HO (A) (B)…
A: Ozonolysis of alkene: One of the important reactions of alkene is ozonolysis. In the ozonolysis…
Q: What is the correct product or products for this reaction?
A: The hydrogenation reaction of alkenes takes place in the presence of H2 and Pd to form alkanes. It…
Q: CH2 1| C CH 11 HCl- CH
A:
Q: What is the structure of the main product produced by the following reaction? но- -CO2H 1) heat ? 2)…
A:
Q: Product of the reaction Choose from the following reactants: B A E
A:
Q: Draw structural formulas for the major organic product(s) of the reaction shown below. CN +…
A:
Q: Which of the following is one of the products formed in the reaction below? IV OA V O B. II OC. I O…
A:
Q: PCC (CH),CHCH,CH,OH
A: PCC is an weak oxidizing agent..it oxidize alcohol into aldehyde or ketone.
Q: What is the major product produced in the following reactions? A 1. B 3. D 4. 1. NANH, / NH, major…
A: NaNH2 being a strong base abstracts acidic proton from given alkyne in first step leading to…
Q: Consider the following reaction: CH3CH2CO0 If the concentration of propanoic acid is 1.7E is the…
A: The equilibrium constant, K, describes the correlation between both the products and reactants of…
Q: this
A: The above reaction is an example of redox reaction
Q: Give the major organic product for the reaction. H₂O A -Br Br
A: As H2O is a polar protic solvent, it will follow SN1 reaction and moreover the positive charge…
Q: >...SH N3 N3 Cl +
A: The conversion of alcohol to thiol can be carried out in a number of steps involving tosylate…
Q: سمر a H₂N. HBr 1) 03, -78 °C 2) H₂O2 1) NaNO₂, HCI, 0 °C 2) CUCN
A:
Q: Halogenation is typically done with symmetric dihalides (Cl2 or Br2 ), but mixed dihalogens can also…
A: The reaction of alkenes with interhalogens (I-Cl) follows Marfovnikov’s rule and forms a single…
Q: 1 BH, 2 H,O, NaOH
A:
Q: What is the major product of each of the following reactions? Assume that there is an equivalent…
A: The given reactions are represented as follows:
Q: Draw the structure of the major organic product(s) of the reaction. CH3 1. DIBAH, toluene 2. Hо"…
A: Diisobutyl aluminium hydride is a reducing agent, it reduces ester functional group in to aldehyde…
Q: Which of the following is a product of the reaction of K2CO3(aq) with Zn(NO3)2 (aq)? O Aqueous K2NO3…
A: The state of the substances in chemical reactions can be obtained from the Balanced chemical…
Explain why the product distribution is observed for the following reactions:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Compounds Mass 3-nitrophthalic acid used 200 mg = 0.2g 8% aqueous hydrazine used 0.4mL 3-nitrophthalhydrazide obtained 130 mg = 0.13 g sodium hydrosulfite dihydrate 0.6 g luminol obtained 70 mg = 0.07 g compute yield for nitrophthalhydrazide in the first step (assume nitrophthalic acid is limiting reagent) compute yield for luminol in the second step (using nitrophthalhydrazide as limiting reagent) compute yield for the overall reactionNa2SO3Na2SO3 reacts with HClO4HClO4 according to the reaction Na2SO3(aq)+2HClO4(aq)⟶2NaClO4(aq)+H2O(l)+SO2(g)Na2SO3(aq)+2HClO4(aq)⟶2NaClO4(aq)+H2O(l)+SO2(g) You wish to provide a 50% excess of 6.466.46 wt% aqueous HClO4HClO4 to react with 50.050.0 mL of 0.07040.0704 M Na2SO3Na2SO3. Calculate the mass of the 6.466.46 wt% aqueous HClO4HClO4 solution required to provide the 50% excess.Weight of crude acetanilide = 2.2 grams Weight of pure acetanilide = 1.5 grams Melting point of recrystallized acetanilide = 114.3 C (a) What is the % yield? (b) What is the % purity?
- The oxidizing and reducing agent is: A) Sodium benzoate B) Sodium nitrite C) Sodium thiosulfate D) Sodium bicarbonate E) Sodium chlorideGive the balanced equation of acetylene and the following reactions • Combustion • dilute KMnO4 • Ammoniacal CuCl / Ammonical AgNO3Give reactants and explain selectivity
- a reaction was preformed in which 0.550 mL of p-anisaldehyde was reacted with a slight excess of benzyltriphenylphosphonium chloride to make 0.646 g of p-methyoxystilbene. calculate the theoretical yield and percent yield for this reaction. 388.9 g/mol benyzyltriphenylphosphonium chloride 136.2 g/mol anisaldehyde d= 1.12 g/mL 210.1 g/mol p-methoxystilbeneWhich one of the following compounds produces 3 M solution when 1 mole of compound is dissolved in 1 L of solution? A. H2SO3 B. Hg2Br2 C. Ag2SO4 D. Cu2S E. SrCl2when 2.0 of NaOHCO3 was heated the weight of the reside product after heating was 1.0g calculate the percent weight loss on heating
- Some people have an allergic reaction to the food preservative sulfite (SO32−). Sulfite in wine was measured by the following procedure: To 50.0 mL of wine were added 5.00 mL of solution containing (0.8043 g KIO3 + 5 g KI)/100 mL. Acidification with 1.0 mL of 6.0 M H2SO4 quantitatively converted IO3− into I3−. The I3− reacted with SO32− to generate SO42−, leaving excess I3− in solution. The excess I3− required 12.86 mL of 0.04818 M Na2S2O3 to reach a starch end point. lO_3^- + 8l^- + 6H^+ rightarrow 3l_3^- + 3H_2O l_3^- + SO_3^2 - + H_2O rightarrow 3l^- + SO_4^2- + 2H^+ l_3^- + 2S_2O_3^2- rightarrow 3l^- + S_4O_6^2- Find the concentration of sulfite in the wine. Express your answer in mol/L and in mg SO32− per liter._____mol/L HINTS: Don't use scientific notation, do put zero before decimal point, carefully evaluate significant figures.The density of acetic anhydride (C4H6O3) is 1.08 g/mL. Calculate the moles of acetic anhydride present in 3.60 mL of acetic anhydride.Calculate the moles of the limiting reagent for the following reaction: oxidation of cyclododecanol to cyclododecanone. 0.2093 grams of Cyclododecanol 0.5 mL of acetone 0.20 mL of Glacial acetic acid 2.0 mL of bleach Final weight of cyclododecanone = 0.0578 g

