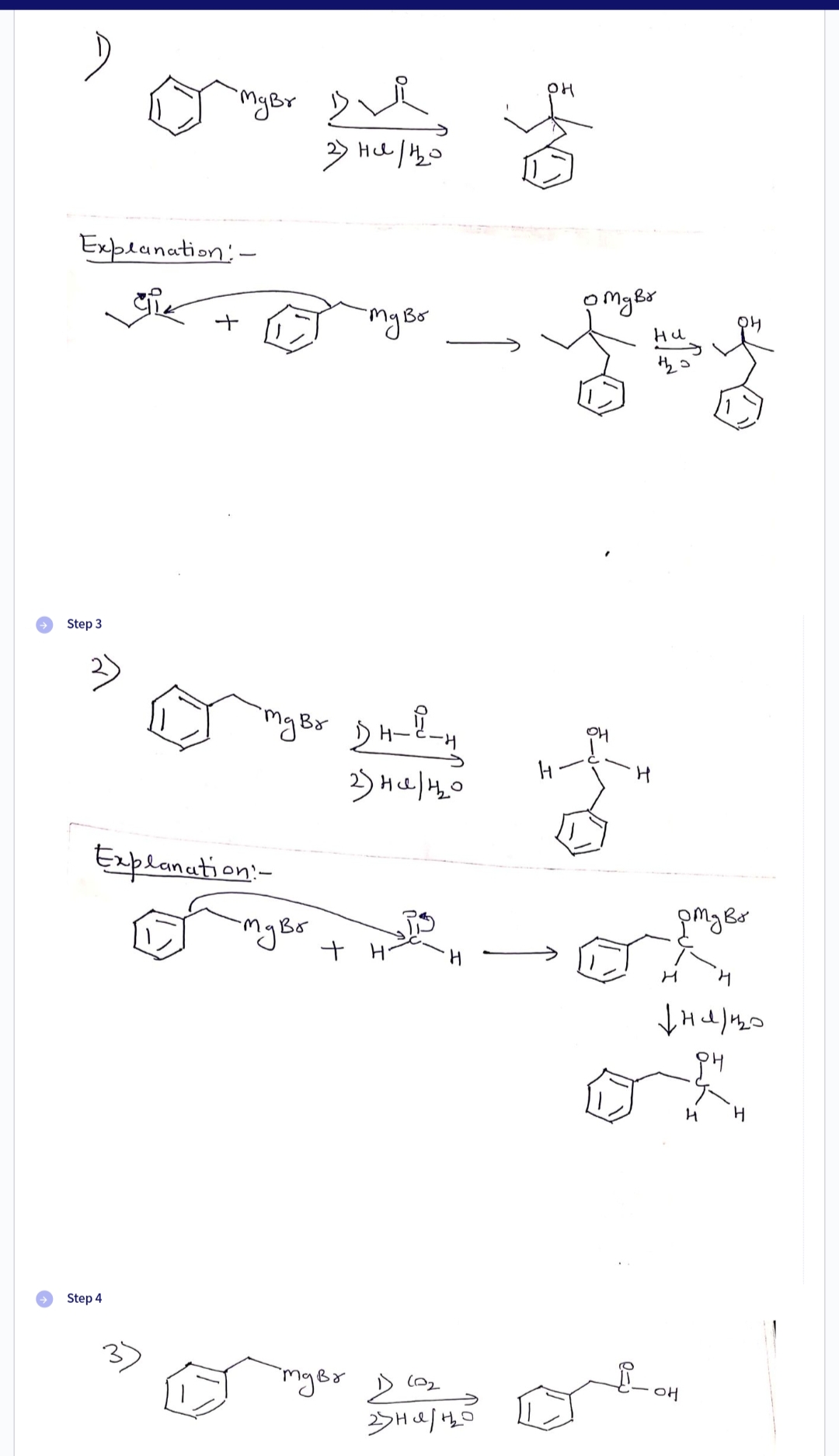
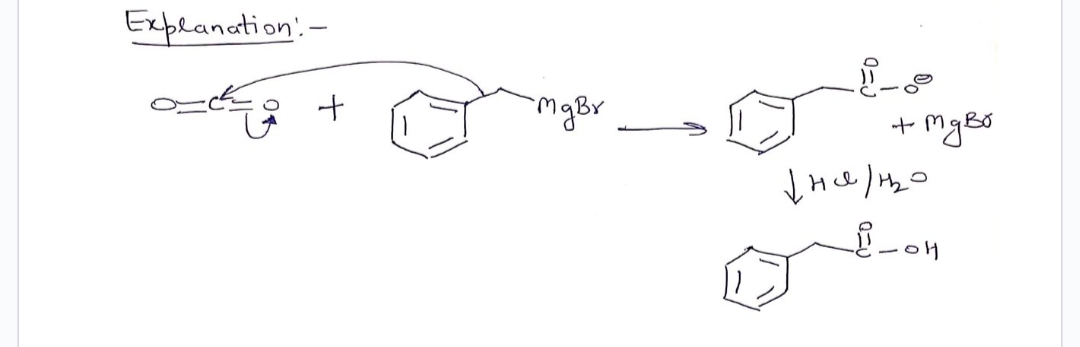
) Explanation' - як Step 3 "My Br Step 4 + Explanation:- 3) нано с кое mger 2н-2-н знано мува + + стува едвил -mg Br 1) (0₂ отдых ни дни значно он ртува н стигна н но н Н Н
Q: discharge tube containing an unknown gas emits light with a wavelength of 485 nm. Given the line…
A: You have to identify the gas that emits light with a wavelength of 485nm. From the line spectra of…
Q: The following materials are reacted together according to the equation below 5.00 moles of KReO4…
A:
Q: Read each description in the first column of the table below. If any chemical element with atomic…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Describe, quantitatively, how you would make 250 mL of a 40 mM HEPES buffer at pH 7.60. You have…
A: Introduction: A buffer solution is a solution in which small addition of strong acid or base does…
Q: Next the calcium hydroxide reacts with magnesium ions, Mg2 () in the seawater to produce magnesium…
A:
Q: D D Question 3 Shown below is an arrow pushing mechanism. CI: Which atom was the nucleophile in this…
A: In nucleophilic substitution reactions, an electron rich nucleophile bonds with or attacks an…
Q: 46) The bonding character of silicon carbide, a ceramic material, is: a) Purely ionic b) Purely…
A: Silicon carbide (SiC) is a ceramic material composed of silicon (Si) and carbon (C) atoms.
Q: = 몸
A:
Q: For X please- 1) 10n + 2713Al → X + 42He 2) 11H + 189F → 158O + X 3) 42He + X → 2512Mg +…
A:
Q: Read each description in the first column of the table below. If any chemical element with atomic…
A: Solution- an element is period 3 and group 2A As magnesium is places in group 2A and period 3 so…
Q: 2. You are provided with KIO3, how much do you need to weigh out and in what volume to give you what…
A: To prepare a solution of potassium iodate (KIO3) with a molarity of 4.167 x 10-3 M, you would need…
Q: the hydrogen bonding intermolecular force is and when it is created. Use H2O and CH4 in your answer…
A: We have been asked when intermolecular force hydrogen bonding is created.We have been asked to use…
Q: 30. A single displacement reaction occurs between copper and silver nitrate. When 63.5 g of copper…
A:
Q: 11) Synthesize 2-methyl-3-hydroxycyclohexone from cyclohexone, methyl iodide, and inorganic…
A: We have to carry out the synthesis of 2-methyl-3-hydroxycyclohexanone from cyclohexanone, methyl…
Q: Name the three alkenes using systematic names. A. H₂C B. H3C CIC The name of compound A is: H3C CH3…
A: IUPAC rule-The ene suffix (ending) indicates an alkene use it by replacing e from alkane 2. The…
Q: For the bond-line drawings below, draw the Newman projection on the template provided. You must draw…
A: The representation of a three dimensional figure in two dimensions with the help of perspective is…
Q: Quantum Numbers and Orbitals: Insert the appropriate values in the chart below. Principal Quantum #…
A: The principal quantum number (n) has integer values from 1, 2, 3 ....... The angular momentum…
Q: B. Write the complete and abbreviated electron configurations for the following elements. 4. Ag 5.…
A: Since, Electronic configuration represent the electron present in orbital in which way. Arrangement…
Q: Write the empirical formula of at least four binary ionic compounds that could be formed from the…
A: ionic compound: ionic compounds are a chemical compounds formed with ions held together by…
Q: JICN "L P, 1) MgBr 2) HC1/B₂0 2) HCI/HO OH НО
A:
Q: green color is seen when a metal salt is heated in a flame. The wavelength of the light emitted is…
A: Flame tests can be used to identify the metal ion based on the color of the flame. Each element has…
Q: Order these chemical species by increasing pH of an 0.1 M aqueous solution of each. That is, imagine…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: B. Write the complete and abbreviated electron configurations for the following elements. 1. Mg 2. K…
A: The elements given are Mg, K, Co.
Q: Write the chemical formula for this molecule: I Drag the slider to get a different view of the…
A: Since, Molecules formed by combination of bond with correct valency. Thus,
Q: UNIT 1 2. Which atom has six valence electrons? a. lithium d. neon b. carbon e. boron c. sulfur 3.…
A: 2) valence electrons: valence electrons are electrons which are present in the outermost shell of an…
Q: Synthesize: OH & its enantiomer Synthesize: 3-methyl-1-pentene From: (E)-3,4-dimethyl-hex-2-ene…
A:
Q: 1) Provide the name for each structure shown. NO₂ E H₂C NH CH3 CH₂ H₂N CH 3
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: The vapor pressure of the liquid HF is measured at different temperatures. The following vapor…
A: Given, Temperature, K Pressure, mmHg T1 = 272.9 K P1 = 356.6 mmHg T2 = 289.9 K P2 = 684.0…
Q: ving reaction. 1. Hg(OAc)2, H2O 2. NaBD4 ОН D _OH D OH
A: The given reaction is oxymercuration demercuration reaction. It is an example of electrophilic…
Q: Draw the structure of the molecule and upload it here. Molecular Formula: C,H₁002 -1 FTIR: Strong…
A:
Q: You found a sphere composed of an unknown pure substance. What is the density (in g/cm³) of the…
A:
Q: Consider the following rate data for this reaction: NO (g) + H₂ (g) HNO2₂ (9) Experiment NO (mol/L)…
A:
Q: D. Calculate the mass (g) of the following samples. 25) 238 moles of lead 26) 1.54 x 109⁹ moles of…
A: D) Calculating the mass (in grams) of the following sample: 25) 238 moles of lead 26) 1.54 × 10-9…
Q: Be sure to answer all parts. The equilibrium lies to the right in the reaction below. Following the…
A: In the Brønsted–Lowry definition of acids and bases, a conjugate acid–base pair consists of two…
Q: A discharge tube containing an unknown gas emits light with a wavelength of 485 nm. Given the line…
A:
Q: The molecule is beta-carotene. Tell me everything i should know about thi molecule. please describe…
A: Beta-carotene is a naturally occurring pigment that belongs to the carotenoid family of compounds.…
Q: Consider the reaction shown below: 2 MnCl₂ (aq) + 5 NaCIO (aq) + 6 NaOH (aq) →→→2 NaMnO4 (aq) + 3…
A:
Q: Draw a resonance structure that places a pi bond in a different position. Include all lone pairs in…
A: Resonance Structure : Resonance structures are described as a group of two or more Lewis…
Q: A certain atom in a molecule has a formal charge of 0 and when illustrated in a covalent bonding,…
A: Halogens have 7 valance electrons and hence these can form 1 covalent bond by sharing 1 electron…
Q: For this problem, the molecular orbitals of vitamin C are represented as thick lines. It takes light…
A:
Q: You are planning to run a gel using a Tris-HCl buffer. You have 0.75 M Tris-HCl, pH 7.30 buffer, but…
A:
Q: Calculate the formal charge on each of the atoms in the Lewis structure given. Be sure to answer all…
A:
Q: What volume, in milliliters, of fluorine gas at 30.0 °C and 725 mmHg is needed to produce 2.20 g of…
A:
Q: You weighed 200 g of NaOH (M = 40 g.mol-¹ and purity = 50%) that you dissolved in a final volume of…
A: You weighed 200 g of NaOH (M = 40 g.mol-1 and purity = 50%) that you dissolved in a final volume of…
Q: The chemical formula for hydrogen bromide is HBr. A chemist measured the amount of hydrogen bromide…
A: Here mass of hydrogen bromide (HBr) is given. We have to convert mass to number of moles. Mass and…
Q: What is the hybridization of each labeled atom below? A: sp²; B: sp; C: sp²; D: sp3 A: sp²; B: sp³;…
A: We are given a structure of a large organic molecule and asked to write the hybridization of the…
Q: Click the "draw structure" button to launch the drawing utility. Draw the Lewis structure of a more…
A: A Lewis structure is a way to represent the valence electrons of an atom or molecule.
Q: A chemist determines by measurements that 0.050 moles of hydrogen gas participate in a chemical…
A: Moles can be calculated using the formula: Moles = Mass/ Molar mass
Q: Compare the glucose levels obtained from (a) whole blood and plasma/serum; (b) arterial…
A: Glucose levels obtained from whole blood and plasma/serum are generally similar.
Q: 4. X4 X4 Find out the Miller Indices of the planes. Z4
A: As per guidelines, we can answer only for three subparts. The question is about Miller indices of a…
just draw the same in chemdraw asap
Step by step
Solved in 3 steps with 2 images

- Give a clear handwritten answer of all sub parts asap..?Given below (c): Standard gibb's free energies (∆Gf0 kJ mol-1 ): UO2 = -962.7 UO22+ = -953.5 U4+ = -579.1 Fe2+ = -78.9 Fe(OH)3 ferrihydrite = -692.07 Mn2+ = -288.1 MnO2 pyrolusite = -465.1 HS- = 12.1 H+ = 0 H2O = -237.1 S0 = 0 Given: U(VI) as uraninite; UO2 (where Mn2+ = reductant; MnO2 pyrolusite = product): ∆ Gr0 = -21.3 KJ/mol E0 (emf) = 0.110 V n = 2 F = 96.42 QUESTION: Calculate Eh equation below to calculate at different pH: – Eh = E0 + (RT/nF) * lnK For U(VI) as uraninite; UO2 (where HS- = reductant; S0 = product): UO22+ + Hs- ---- > UO2 + S + H+ [A] pH 3 [B] pH 7Provided the following information determine Δ?ΔHformation of C2H6(g). C(graphite) + O2(g) ⟶⟶ CO2(g) Δ?=−393.5??ΔH=−393.5kJ H2(g) + 1212O2(g) ⟶⟶ H2O(l) Δ?=−285.8??ΔH=−285.8kJ C2H6(g) + 7272O2(g) ⟶⟶ 2CO2(g) + 3H2O(l) Δ?=−1560.7??
- https://m.youtube.com/watch?v=vM1SP346XBc&list=PLeJOSNLNZfHubfLdq0kOayASeUllMOGn4&index=4 I watched this the lecture video over and over and I am allowed to work with someone but I am having trouble with part B and I provided the YouTube link of the data or video attached to this labGeneral Chemistry (Quesion -6) =====================9. Calculate the Go in kJ/mol. 3Fe2+ (s) + 2Cr(aq) ⟶2Cr3+(aq) + 3Fe (s); Eocell=0.30 V Group of answer choices -170 +170 +87 +58 -195 10. Calculate Ecell in volts. Zn(s)|Zn2+(2.50x10-4M)||Sn2+(1.50M)|Sn(s) Eocell=+0.624 V Group of answer choices 0.736 0.635 0.512 0.848 no correct answer
- Please give clear handwritten answer of all subparts of question no. 5 i will give you upvote.What is ΔHsys for a reaction at 51.4 °C with ΔSsurr = 749 J mol-1 K-1 ? Express your answer in kJ mol-1 to at least two significant figures.Consider the theoretical bases below and use the Kb values provided (at 25 oC) to determine the Ka for H2XO42- at 25 oC. XO44- Kb = 1.5 x 10-3 HXO43- Kb = 2.0 x 10-6 H2XO42- Kb = 1.8 x 10-9 H3XO4- Kb = 2.5 x 10-12

