Q: (a) Group 1A positive + The elements in group 1A configuration. an electron (obtain a charge) in…
A: Concept: Group I A is the alkali metal group. It includes Lithium, sodium, potassium, rubidium,…
Q: When an alkali metal forms an ion, what is the charge of the ion? What is the charge of an alkaline…
A: Alkali metals are those metals which are present in the first group of the periodic table. The…
Q: Write the symbol and name for the elements located in the perio a Belongs to group VIA(16) and…
A: We can take reference of periodic table , and see the element according to direction provided in…
Q: Which of these is the smallest? Arsenic Selenium Bromine Krypton Iodine
A: Answer:- This question is answered by using the simple concept of variation of atomic size across…
Q: Determine whether each element is a main-group element.a. tellurium b. potassiumc. vanadium d.…
A: Main group elements are the elements of s and p block. In s-block , group 1 and group 2 are present.
Q: Order these elements from highest to lowest ionization energy 1 | Chlorine, CI | Fluorine, F 3 |…
A: The ionization energies (in kJ/mol) of F,Cl,Br and I are 1681,1255,1140 and 1008 respectively. So…
Q: Ca+ (g) + e- --> Ca2+ (g) is the correct representation of the second ionization of calcium.…
A: Given representation, Ca+ (g) + e- --> Ca2+ (g)
Q: Rb+
A: Ionization energy is the minimum energy required to remove an electron from outer most shell in…
Q: Arrange in order of increasing nonmetallic character. (a) the Period 3 elements Cl, Si, and Na (b)…
A: All the chemical elements of the periodic table can be broadly divided into three categories,…
Q: Element X combines with phosphate ion to form X3(PO4)2. Element X is A. Ca+2 B. Al+3 C. N-3 D.…
A: Given: Element X combines with phosphate ion to form X3(PO4)2 To identify Element X
Q: Determine the balanced reaction of element A (white circles) with element B (gray circles) as…
A: In the given problem, Element A with white circles is diatomic molecule, therefore it will be…
Q: To what group number does each element belong?(a) germanium(b) nitrogen(c) sulfur(d) carbon(e) boron
A: An atom is the basic and fundamental unit of matter. An atom consists of electrons and the nucleus…
Q: - - Identify the lowing families of elements. (a) boron group (c) nickel group group number…
A: To find group number we must know total valence electrons of atom.
Q: Reactivity Trends on the Periodic Use the drop-down menus to complete each sentence. Elements in the…
A: Introduction: We have to fill the followings.
Q: Group the elements into pairs that would most likely exhibit similar chemical properties. It does…
A: We have to pair the elements as per their properties
Q: Draw electron dot diagrams of the following elements: 6. Hydrogen 7. Chlorine 1. Carbon 2. Nitrogen…
A: Lewis’s structure: Lewis's structure is based on the octet rule. The Lewis structure is a simplified…
Q: Which group of elements has the largest atomic radii? Halides Alkali metals Alkaline earth metals…
A: Atomic radii varies with the group of periodic table and with this we will answer the question.
Q: 1. The potassium ion Kt is isoelectronic with which of the following? a) 02- b) F- c) S2- d) Na + e)…
A: Isoelectronic species means same number of electron present in both the elements. K+ and S2- are…
Q: Group the elements into pairs that would most likely exhibit similar chemical properties. It does…
A: Chemical properties of any element depends on the number of valence electron the element is having.…
Q: What alkaline earth metal is located in period 4 of the periodic table of elements? اختر احدى…
A: The question is based on the concept of inorganic chemistry. We have to identify the alkaline earth…
Q: Arrange Na+, Ca, O2− in increasing atomic/ionic radius (smallest to largest). Group of answer…
A:
Q: Which is NOT a property of Fluorine? a) pale-yellow gas b) strong nonmetallic c) has 7 valence…
A: Fluorine is a halogen that belongs to 17th group of periodic table. Furthermore, it is present in…
Q: The diagram below shows the energy levels and electron configuration for fluorine Which of the…
A: F has a total of 7 valence electrons with a valence electron configuration of 2s2 2p5.
Q: Rank the elements from highest to lowest ionization energy. To rank items as equivalent, overlap…
A: • The given elements are - F, N, O, B , Li and Na • We need to rank the given elements from…
Q: What is the name given to group 7A alkali metals alkaline earth metals…
A: An atom is mainly composed of three sub-particles; electrons, protons and neutrons. These particles…
Q: Fill in the Blank. Identify the what is asked. 11. A representation that shows valence electrons as…
A: 11. Lewis dot structure Lewis structure is the representation of a molecule by using only its…
Q: Identify the appropriate atoms by selecting each atom and assigning it a map number of 1 until all…
A: A carbon molecule is chiral when it is attached to 4 different atoms or groups.
Q: Period ale to eldat bibohaq to train Characteristics of element belong to every period. Give at…
A: Below attached file showing the details answer. All the best.
Q: Amazing news! Chemists have just discovered a brand new element on an asteroid. Calculate the…
A:
Q: Use Coulomb's law to calculate the energy of a copper ion and an oxide ion at their equilibrium…
A: The energy of given two ions can be calculated by using Coulomb's law as follows :
Q: Species Number of electrons Number of electrons Number of core in valence shell electrons Ca Cs+ p3.…
A: A neutral atom consists of same number of protons and electrons. When the atom loses electron, it…
Q: Which of the following pairs of ions represent isoelectronic species? A) Na* and N²- B) A13+ and 02-…
A: The paiss of ions that are isoelectronic species is as following -Among the given options , B is…
Q: 2Mg (s ) + O (g ) 2MGO (s) Magnesium (Mg) reacts with oxygen gas (O) to produce magnesium oxide…
A: Given Reaction2 Mg (s) + O(g) → 2 MgO(s)Correct movement of electron = To be determined
Q: Identify the highlighted element in the periodic table and give its: [1] element name and symbol;…
A: Various elements are arranged in 7 periods and 18 groups in the periodic table. The elements of…
Q: Classify each element. Note that another term for main group is representative, another term for…
A: the element is classified into different categories according to their behaviour is:
Q: 1) Which of the following make an isoelectronic pair? A) Ca and Fe³+ B) O and F C) F and CI D) Cl…
A: Isoelectronic pair are the species which have same number of electrons.
Q: 1. Which of the following elements would you expect to behave most like magnesium? A) Sodium B)…
A: Since you have posted multiple questions, we will solve only first question for you. If you want…
Q: What is the charge of the ion most commonly formed by S?a) 2+ b) +…
A: Given: Common charge of sulfur
Q: E is a made up element and contains 6 valence electrons Y is a made up element and contains 7…
A:
Q: Which of the following is Not isoelectronic with Argon A. K+ b. P3- C. Ca+2 D.…
A: We have to predict the ion isoelectronic with the Argon.
Q: Classify each element. Note that another term for main group is representative, another term for…
A: the classification of the element in the following categories is described in the table below:
Q: Discuss the general chemical reactivity of noble gases.
A: In the18th group of the periodic table, noble (inert) gases are placed.
Q: Classify each element. Note that another term for main group is representative, another term for…
A:
Q: Elements in Group 7A are known as the A) alkaline earth metals B) alkali metals C) chalcogens E)…
A: Periodic table represents the systematic arrangement of elements in which the properties of these…
Q: Which of the elements listed below has a lower ionization energy? Strontium Silicon
A: Ionization energy is the energy required to eliminate one electron from the valence shell from an…
Q: Rank the following isoelectronic species in order of decreasing size: Argon atom (Ar), Calcium ion…
A: Anions are formed by the gain of electrons and cations are formed by the loss of electrons. The…
Q: Formulas of oxides only are given in a line: а. Na20, CaO, НСІ O b. K20, BaO, N205 c. Na202, NH3,…
A:
Q: Choose Metals, Metalloids ,Non-metals ( Os , N , Ge , Si , Te , Cs , S , He )
A: We will Choose Metals, Metalloids ,Non-metals ( Os , N , Ge , Si , Te , Cs , S , He ).
Q: Identify each statement as true or false: (a) Cations are largerthan their corresponding neutral…
A: Given statements, (a) Cations are larger than their corresponding neutral atoms (b) Li+ is…
Q: X is a made up element and contains 4 valence electrons E is a made up element and contains 6…
A: Given that, Number of valency electrons in X = 4 Number of valency electrons in E = 6 Charge on the…
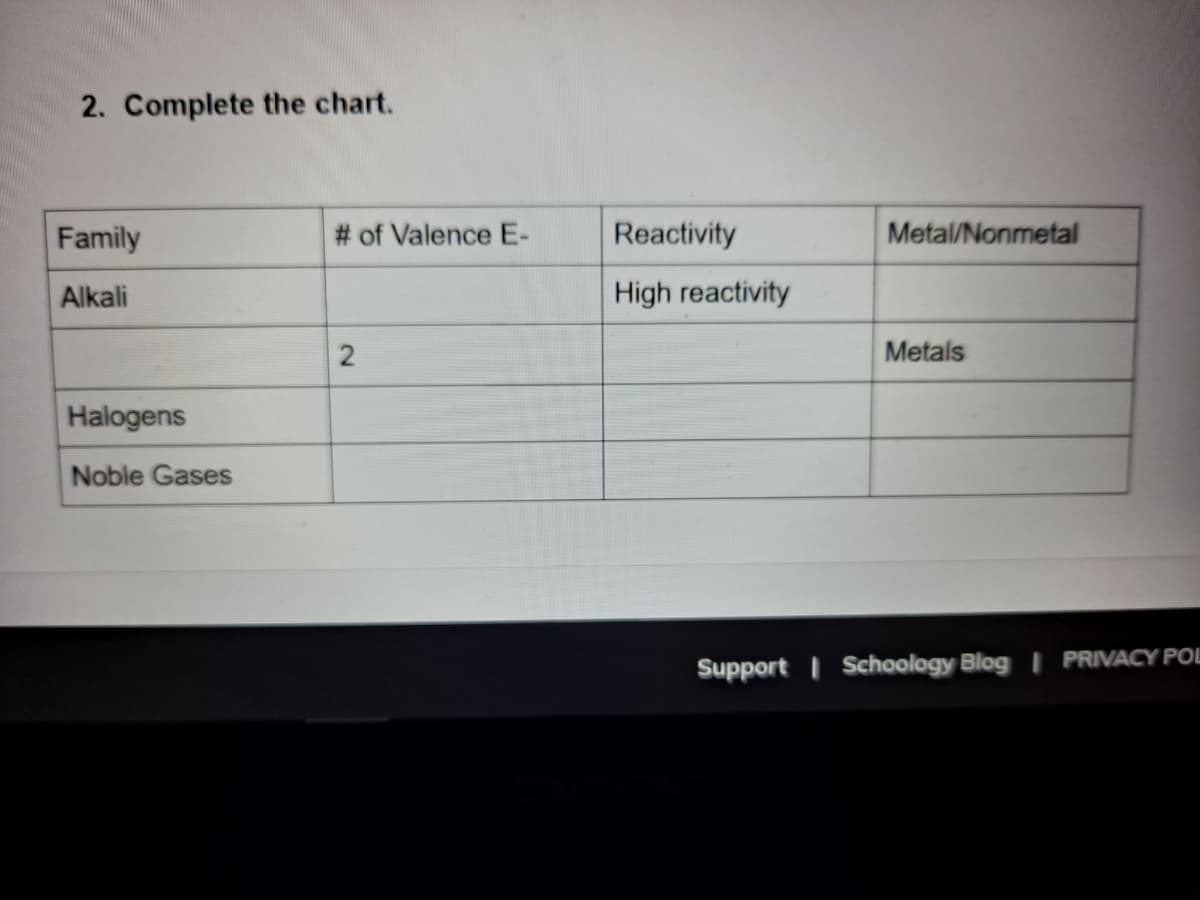

Step by step
Solved in 2 steps with 2 images


