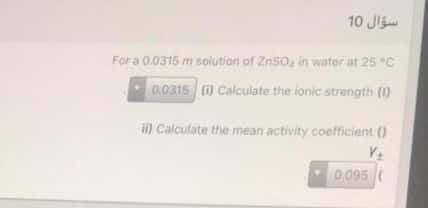
For a 0.0316 m solution of ZnSo, in water at 25 "C 0.0315 ) Calculate the ionic strength (1) ) Calculate the mean activity coefficient () 0.095
Q: Consider a binary solution for which the vapor pressure (in torr) of one of the components (say…
A:
Q: Many ions do not have tabulated activity coefficients nor can the activity coefficient be estimated,…
A:
Q: Calculate the FECO3 molar solubility in a Fe(NO3)2 desolution whose ionic strength (u) is 0.05M. The…
A: Molar solubility is defined as the number of moles of solute that can be dissolved per liter of the…
Q: When methanol is dissolved n water a conducting solution results. Justify your answer.
A: Ionic substances dissolve in water and conduct electricity due to freedom of motion of ions.
Q: Calculate the activity coefficient, y, of Mn²+ when the ionic strength of the solution, 44, is 0.077…
A:
Q: Determine the solubility of lead(II) phosphate in a solution that is 0.0500 M in NaNO,. Ignore ion…
A: Solubility of lead(II) sulphate is calculated as follows
Q: 1) Calculate the molar conductance of KCI solution (0.1M), if the specific conductivity for the…
A:
Q: Calculate for the equilibrium or solubility constant of the dissolution of 0.065 m M2SO4 with th…
A:
Q: Calculate activity coefficient for the binary solutions at 20°C and 1 atm; (a) for which an enthalpy…
A:
Q: using the debye-huckel limiting law, calculate the activity coefficient of sulfate ion with u =…
A:
Q: By using Debye-Huckel limiting law calculate activity value (a) of the La3+ ions in the following…
A: According to Debye Huckel limiting law we can calculate the ionic activity coefficient value by…
Q: Calculate the Ionic strength of solution prepared by mixing of 0.2 M NaNO3 and 0.02M of K2Cr2O7: a)…
A: We can calculate the ionic strength of solution by using the formula of ionic strength : I = ionic…
Q: An equimolar solution of ammonium sulfate, sodium chloride and magnesium nitrate has ar ionic…
A: Suppose the concentration of each solution is M since solutions are equimolar. Hence concentration…
Q: For a 0.0924 m solution of CaCl, in water at 25 °C (iii) Calculate the mean ionic molality (m.) (iv)…
A: (iii) Calcium chloride would dissolve as, CaCl2→Ca2++2Cl- The concentrations of calcium ions and…
Q: Calculate the ionic strength of 0.0079 M NaOH. Calculate the ionic strength of 0.00027 M La(IO,),.…
A:
Q: The ionic strength of an aqueous solution containing 0.0087 M NaCI and 0.0020 M La(IO3)3 is: O a.…
A:
Q: The ionic strength of an aqueous solution containing 0.0087 M NaCl and 0.0020 M La(103)3 is: O a.…
A: We will calculate Ionic strength
Q: Calculate the mean activity coefficient y, for the Ba2+ and SO2 ions in a saturated solution of…
A:
Q: 24. Use the extended Debye Huckel limiting law to calculate the mean ionic activity coefficient…
A:
Q: An aqueous solution is prepared by mixing 20.0 mL of 0.0065 M K3PO4, 30.0 mL of 0.015 M NaCl, and…
A: [K3PO4] = 0.0065 M Vol of K3PO4 = 20.0 mL Millimoles of K3PO4 =…
Q: To prepare a stock solution of EDTA, 73 grams of EDTA were mixed to 500 mL of distilled water. What…
A:
Q: ity of saturated solution of of BaSO4 is 3.48 x 10-4 S/m and the conductivity of pure water is 0.50…
A: The ions present in a solution contribute to the total conductivity of a solution. The Ksp for BaSO4…
Q: culate Yso,2- and YM+ using the given mean activity coefE ecies Mean activity coefficient 0.71 CI…
A:
Q: Calculate the ionic strength of a solution that is 0.20 M in FeCl3 a 0.10 M in FeCl2 ? IS
A:
Q: Calculate the [Cu2+] and [IO3–] in a solution made by saturating pure water with solid Cu(IO3)2. Use…
A:
Q: Calculate the ionic strength of 0.01818 M Na2CO3 + 0.0505M NaCl + 0.0202 M K2SO4
A: The formula used to calculate the ionic strength is as follows,
Q: The disolufion of unknown subslances in 825 g of berzene, C, of 298 Klowen the heezing point by…
A: The depression in freezing point ∆Tf is given by: ∆Tf=Kf×m······(1) Here, Kf is the freezing point…
Q: For a solution in which u = 8.0 x 10, calculate K', for s. Pbl,. The thermodynamic constant of the…
A:
Q: For a 0.067 m solution of ZNSO4 in water at 25 °C. Calculate the ionic strength (1) O a. 0.4020 Ob.…
A: Ionic strength basically measures the concentration of ions that is present in the given solution. C…
Q: For a strong electrolyte when C = 0.04 M then Am = 250 S cm2 /mol. And when C= 0.09 M then Am = 200…
A:
Q: The table shows the value of the activity coefficient of Ag* Ionic strength at different values of…
A: The activity coefficient is related to ionic strength by the debye huckle limiting law which is log…
Q: Find the isotonic coefficient for a 0.2M solution of an electrolyte, if it is known that one liter…
A: Isotonic coefficient is also called Vant Hoff's coefficient which is the ratio between the actual…
Q: Determine the equilibrium constant (solubility constant) when a total of 0.025 m of M2SO4 wa…
A: Given reaction: M2SO4(s)↔2M++SO42- (Note that the reaction given in the question…
Q: How do you calculate for the equilibrium or solubility constant of a dissolution reaction given the…
A: To Calculate equilibrium or solubility constant of a dissolution reaction given the ionic strength…
Q: For a solution in which u = 8.0 x 10, calculate K, for Pbl,. The thermodynamic constant of the…
A:
Q: Calculate for the equilibrium or solubility constant of the dissolution of 0.065 m M2SO4 with th…
A: From the mean salt method the mean activity coefficient of M+ and SO42- can be calculated as:…
Q: A solution of Na SO, has an ionic strength of 0.24 m, then the molality of this solution is:
A: The ionic strength of the solution can be determined by using the formula given below as; I=12ΣmiZi2…
Q: Activity is the value that replaces concentration in a thermodynamically correct equilibrium…
A:
Q: 3. Consider a 0.10m solution of Mg(SO4) а. Calculate the ionic strength of this solution b. Use the…
A: Introduction : We have to calculate ionic strength and mean activity coefficient for the given…
Q: 1.38 g of NaCI (molar mass 58.44 g/mol) as dissolved in 43.3 mL of water. Assuming molar…
A: Molarity = no. of moles/vol. of soln. =(1.38g/58.44gmol-1)/43.3cm3…
Q: To verify Beer’s Law for solution of KMnO4 or K2Cr2O7 using colorimeter.
A: Spectrophotometer is a device consists of monochromator that produces a light beam containing a…
Q: Find the activity coefficient using the Debye-Huckel equation for Be²* if µ = 0.075. Assume the…
A:
Q: Which of the following statement/s is/are TRUE? I. The Arrhenius Theory cannot be applied to account…
A: The answer is given below
Q: From the given information in the table calculate for both titration1 and 2: 1)moles of thiosulphate…
A:
Q: Given that p*(H2O)=0.02308 atm and p(H2O)=0.02238 atm in a solution in which 0.122 kg of a…
A: Activity of H2O = 0.97 Activity Coefficient of H2O = 0.98
Q: The ionic strength of a solution containing 0.3 M sucrose and 0.6 M sodium Phenobarbital (1:1)…
A: Ans Ionic strength is given by the formula (I)= 12MiZi2 where the M= concentration of the solution,…
Q: Taking the activity coefficient effect into consideration, please calculate the in a solution that…
A: Given: ionic strenth = 0.001 M Z+ = +3 and Z- = -4 formula used: activity coefficient = -[0.5 ×…
Q: Using the Debye-Huckel limiting law equation, Please compute ( not qualitatively) the hydroxide ion…
A:
Step by step
Solved in 2 steps

- Calculate the activity coefficient, γ , of Cu 2 + when the ionic strength of the solution, μ , is 0.069M by linear interpolation of the data in the table. \[ \gamma_{\mathrm{Cu}^{2}+}= \] Calculate the activity coefficient, γ , of Cu 2 + when the ionic strength of the solution, μ , is 0.069M by using the extended Debye-Hückel equation at 25 ∘ C , where the ion size is 600pmThe mean ionic activity coefficient for 0.1 M H2SO4 is 0.265. Calculate the activity of H2SO4Which has an acitivity coeeficient closest to 1 with ionic strength = 0.001 M? a. Lithium ion b. Magnesium ion c. Lead ion d. Aluminum ion
- How can I calculate the activity coefficient and the resulting activities of a mixture of 0.001M NaCl with 0.05 NaNO2 using Debye-Hückel approximation? Is the resulting activity coefficient only for one ion species or the whole salt?if the mean ionic activity coefficient ,? ± of KIO4 in the saturated solution 0.85, what Ksp of KIO4? the information is above in the paper.Average activity coefficient for HBr at 25oC for three different diluted solutions is 0.930 (at m = 5.0 mmol kg-1), 0.907 (at m = 10.0 mmol kg-1), and 0.879 (at m = 20.0 mmol / kg-1) has been given. Calculate the approximate value of the constant B in the expanded Debye Hückel law.
- Calculate the gravimetric factor of the following. 2 Fe3O4 is sought(Analyte), 3 Fe2O3 is weighed (Precipitate)6 The ionic strength of a solution containing 0.00100 F KCl, 0.0048 F K3PO4, and 0.0070 F Pb(NO3)2 is .0508 M Find the activity coefficient and activity of potassium in problemAllowable recovery from limiting salt calculations Determine the limiting sait and allowable recovery for a brackish water RO system containing the following solutes: calcium 74 mg/L. barium 0.008 mg/L. and sulfate 68 mg/L. Assume 100 percent rejection of all solutes and & polarization factor of 1.15 and ignore activity coeficients (i.e., activity = concentration).
- A 10mL solution contains dissolved PbCl2. There are 0.02575 moles Pb2+ and 0.0515 moles Cl-. What is the math expression for the activity coefficient of an ion according to Extended Debye-Hückel approach?An equimolar solution of ammonium sulfate, sodium chloride, and magnesium nitrate has an ionic strength of 0.15M. What is molar concentration of the solution wiht respect to each solute? a. 0.010 b. 0.015 c. 0.020 d. 0.025Calculate the gravimetric factor of the following. 2 Fe is sought(Analyte), 1Fe2O3 is weighed (Precipitate)

