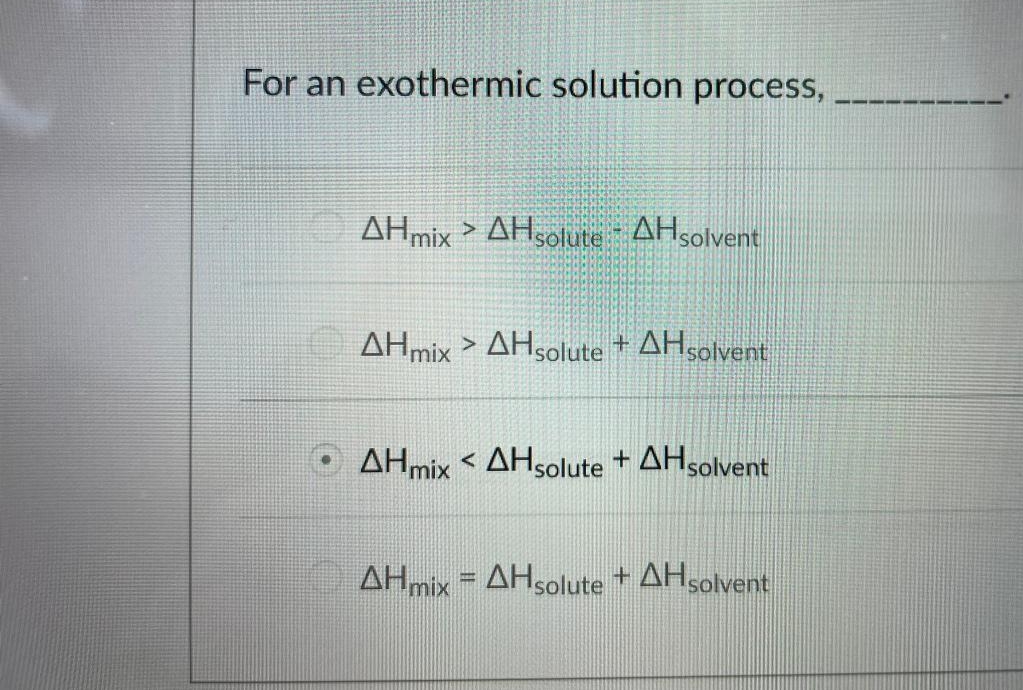
For an exothermic solution process, AHmix > AHsolute AHsolvent AHmix > AHsolute + AHsolvent O AHmix < AHsolute + AHsolvent AHmix = AHsolute + AHsolvent
Q: A student dissolves 12.5g of lithium chloride LiCl in 250.g of water in a well-insulated open cup.…
A:
Q: The heat of solution of potassium acetate (KC2H3O2) in water is -15.30 kJ/mol. If 14.40 g of…
A: Given data set: Mass of potassium acetate = 14.40 g Volume of water= 560 ml = 0.56 L Initial…
Q: Use the temperature and atmospheric pressure data to identify which of the following bodies of water…
A:
Q: How many calories of heat are produced or absorbed when a solution of HF and KOH are mixed resulting…
A: Given: Mass of solution= 42.0 gInitial temperature, Ti = 22.5°CFinal temperature, Tf =…
Q: What mass of silver nitrate will precipitate out of solution if 950.0g of silver nitrate is…
A: Given data,Mass of AgNO3=950gMass of water=225gInitial temperature=50oCFinal temperature=20oC
Q: What is the molarity of a solution in which 150g of CaO are dissolved in 1900mL of solution? (Use…
A:
Q: A. Freezing Point of Cyclohexane (Solvent) 1. Mass of beaker, test tube (g) 85.364g 2. Freezing…
A: 1. Mass of beaker + test tube = 85.364 g2. Freezing point from cooling curve = 5.68°C1. Mass of…
Q: The heat of solution of ammonium nitrate is 26.2 kJ/mol. When ammonium nitrate solid is dissolved in…
A: Endothermic reaction: The reaction in which heat energy is absorbed by the system from the…
Q: A student determines the heat of dissolution of solid cobalt(II) sulfate using a coffee- cup…
A: Given the mass of water inside the coffee-cup calorimeter = 105.00 g Mass of the solute, cobalt(II)…
Q: The total enthalpy change associated with forming a solution (ΔHsoln) can be broken down into three…
A: A solution is made up of two components: solute and solvent. To form a solution, the solute is added…
Q: In a coffee-cup calorimeter, 1.60 g of NH4NO3 is mixed with 75.0 g of water at an initial…
A: The amount of NH4NO3 added to 75.0 g of water is 1.60 g. The initial temperature and final…
Q: 13.6 g of solid ammonium nitrate (NH4NO3, molar mass 80.052 g/mol) is dissolved in 120 mL of water.…
A:
Q: A 10.5 g sample of an ionic compound with a molar mass of 163 g/mol was added to water to form 65.2…
A: Given data: Mass of ionic compound = 10.5 g Mass of solution = 65.2 g Density of solution = density…
Q: acid solution containing 572g of sulfuric acid per liter of solution has a density of 1.33g/mL.…
A: As per our guidelines, I can answer only first three sub-parts. Molarity = number of moles of solute…
Q: In an coffee cup calorimeter experiment, sodium hydroxide (7.17 g) was dissolved in water (86.6 g).…
A: Enthalpy of solution :- It is the amount of heat released or absorbed in the system…
Q: A 400-mL solution of 0.200 MHA, initially at 30.5°C, was placed in an insulated bottle. A 200-mL…
A:
Q: An automobile antifreeze mixture is made by mixing equal volumes of ethylene glycol (SG= 1.114; MW =…
A: Since you have posted multiple sub-parts, the answer for first three sub-parts are given below.…
Q: List the Enthalpy changes accompanying the solution process?
A: ANSWERE IS DISCUSSED BELOW :
Q: The enthalpy of solution for ammonium perchlorate NH4 CIO, is +33.47 kJ/mol. How many grams of…
A:
Q: When 1.18 g of KClO3 was added to 149.35 g of water in a calorimeter, the temperature dropped by…
A: Given:Mass of water = 149.35 g.Mass of KClO3 = 1.18 g.specific heat of the solution = specific heat…
Q: - 10.50g of CaCl2 is added to 100.0g of water at 25.0°C. When the salt is completely dissolved the…
A: · Mass of water mwater = 100.0 g· Mass of CaCl2 msalt = 10.50 g· Initial temperature Ti = 25.0 °C·…
Q: Magnesium chloride is often used to melt ice on sidewalks. Considering that the solubility of…
A:
Q: When 2.00 g of sodium hydroxide (NaOH) was dissolved in 150.00 g of water a value of 11.00oC was…
A: Given, mass of sodium hydroxide = 2.00g. Mass of solution = 150.00g. ∆T =…
Q: Using the temperature of 25°C, is 28.0 grams of KNO3 in 300.0mL of water: saturated, unsaturated or…
A: Temperature given = 25°C Amount of KNO3=28g Volume of water= 300mL
Q: heat absorbed or lost, in J, by the water = 5.89 j 1. a. The heat transferred from/to the NH4NO3…
A: a) Entropy is defined as the measure of disorder or randomness of system entropy. It is a…
Q: KAl3Si3O10(OH)2(s) + H+(aq) + H2O(l) -->Al2Si2O5(OH)4(s) + K+(aq) Balance the equation without…
A: In the balanced chemical equation, the total no of all the atoms should be same on both reactant as…
Q: It sodium perchlorate is soluble in water. When 7.41 g of NaCIO4 is dissolved in 108.00 g of water,…
A:
Q: 6.A fuel is burned with 50% excess air and the combustion characteristics of the fuel oil are…
A:
Q: 5. A substance is more soluble in cold water than in hot water so: a. The enthalpy of the solution…
A:
Q: Determine the freezing point of the following solution: A. 0.5 mole Br2 dissolved in 507g…
A: We have to predict the freezing point of solution.
Q: How many grams of a hypothetical salt X4Y5 of molar mass 54.00 g/mol would need to be added to…
A: Given : Freezing point of solution = -19.8 oC mass of water i.e solvent = 514 g = 0.514 Kg…
Q: small.unt of sodium chloride (NaCl) is dissolved in a large amount of water. Imagine separating this…
A: The process by which a solid substance breaks down to corresponding ions is called dissociation.…
Q: You have a container of 17.42 M conc. acetic acid (CH3COOH(aq)) with density 1.05 g/mL, find the…
A: Observing the given question it is clear that for given volume 150mL molality has to be calculated.…
Q: The hydration of a hydrocarbon, but-2-ene, C4H8 produces butan-2-ol, C4H100 as follows: Penghidratan…
A: The heat content of a system is known as enthalpy. The amount of energy gained or lost during a…
Q: 2. An unknown solid Y was dissolved in water resulting to a decrease in temperature of its…
A:
Q: student combines 0.19 g of Mg metal with HCl and gets a final V of 0.25 L which completely reacts…
A: The solution is as follows:
Q: You are given 10.0 grams of a mixture of NANO3 and KF. You dissolve this mixture in 100.0g of water…
A:
Q: An automobile antifreeze mixture is made by mixing equal volumes of ethylene glycol (SG= 1.114; MW =…
A: Volume percent: As there are equal volumes of two solutions. The volume percent would be 50% for…
Q: Determine the normality of the following solutions: 95g PO4(-3) in 100mL solution
A: To determine the normality of a 95g PO4(-3) in 100mL solution.
Q: In the following reaction calculate 1 point and find the normality when it is 1.0 M H3PO4? Given…
A:
Q: 0.9% (w/v) sodium chloride (MW = 58.44 g/mol) solution (Normal Saline Solution) has an osmotic…
A: 0.9% (w/v) MW = 58.44 g/mol osmotic pressure = 3.91 atm T = 25ºC
Q: Q) A student misreads the directions and adds 4.0 g of benzoic acid instead of 0.4 g. What do you…
A: Given that the student adds 4.0 g of the solute, benzoic acid instead of 0.4 g Mass of the solute,…
Q: Ethylene glycol [CH2(OH)CH2(OH)]is a common automobile antifreeze. The freezing point and boiling…
A: Freezing point = -8.09 oC Boiling point= 102.262oC
Q: A student determines the heat of dissolution of solid ammonium chloride using a coffee-cup…
A:
Q: Boiling point of distilled water 97℃ Boiling point of solution 99℃ Mass of flask 118.2g Mass…
A: Solution is made up of two components which are solute and solvent. Solute is present in the minor…
Q: The dissolution of barium hydroxide in water is an exothermic process. Which of the following…
A: Generate energy in the form of sound, light, cold and heat. Endothermic reactions absorb energy from…
Q: Dane is attempting to make experiments on her manufactured apple ciders. Lab analysis has shown that…
A: Given, Mass percentage of C6H12O6 = 35% Molal freezing point constant of H2O = kf = 1.86 oC/m…
Q: The hydration of a hydrocarbon, but-2-ene, CAHS produces butan-2-ol, C4H100 as follows: Penghidratan…
A: Th enthalpy change of the reaction is equal to the sum of the energies of the different steps. The…
Q: The heat of solution of potassium acetate (KC2H3O2) in water is -15.30 kJ/mol. If 15.90 g of…
A:
Q: first-aid hot packs, giving off heat when dissolved in water. A coffee-cup calorimeter at 25ºC…
A:
please answer clearly and explain.
For an exothermic solution process, ______
Step by step
Solved in 2 steps with 2 images

- 1. What is the molarity of glucose (mol wt =180) in a solution containing 6.70g of glucose and 150 g of water? The final solution has a specific gravity of 1.015. Round off to 4 decimal places. 2. What is the osmolality of 12.5% by weight solution of AlCl (MW=133 g/mole)? 3. What is the normality (N) by w/v solution of CaCl containing 5 g of CaCl and 105g of water (CaCl MW=111 g/mole)? The specific gravity of the final solution is 1.35.Osmotic pressures are often reported in units of atmospheres or mm Hg. The latter impliesthat the height of a column of liquid can be used as a measure of pressure. This is, in fact, thebasis of the torricellian barometer from which the units of “torr” come. The pressure, P, isrelated to the height of the column by:P = ρghwhere ρ is the density of the liquid, g is the acceleration due to gravity and h is the height. Whatwill be the height (in mm) of a column of aqueous solution with a density of 0.9987 g/mL, if theosmotic pressure of the solution at 25 °C is 1.4 mm Hg? The density of mercury at thistemperature is 13.8 g/mLIn the determination of molar mass of an unknown substance by ebullioscopic constant, 30 mL of acetone (C3H6O) was placed in a test tube with thermometer and glass tubing and subjected to water bath. Upon boiling, the temperature reads 56 deg C. For the boiling point of unknown-acetone solution, you prepared the solution by mixing 1.60 g of unknown solute in the 30 mL acetone and subjected it again to water bath. The boiling temperature of the solution is 56.68 deg C. The density of acetone = 0.9849 g/mL and the Kb of acetone = 1.67 degC/kg
- In the determination of molar mass of an unknown substance by ebullioscopic constant, 30 mL of acetone (C3H6O) was placed in a test tube with thermometer and glass tubing and subjected to water bath. Upon boiling, the temperature reads 56 deg C. For the boiling point of unknown-acetone solution, you prepared the solution by mixing 1.60 g of unknown solute in the 30 mL acetone and subjected it again to water bath. The boiling temperature of the solution is 56.68 deg C. The density of acetone = 0.9849 g/mL and the Kb of acetone = 1.67 degC/kg What is the value for delta Tb or the change in boiling temperature (in degrees Celsius)?In the determination of molar mass of an unknown substance by ebullioscopic constant, 30 mL of acetone (C3H6O) was placed in a test tube with thermometer and glass tubing and subjected to water bath. Upon boiling, the temperature reads 56 deg C. For the boiling point of unknown-acetone solution, you prepared the solution by mixing 1.60 g of unknown solute in the 30 mL acetone and subjected it again to water bath. The boiling temperature of the solution is 56.68 deg C. The density of acetone = 0.9849 g/mL and the Kb of acetone = 1.67 degC/kg What is the value for delta Tb or the change in boiling temperature (in degrees Celsius)? Final answer must be rounded off to 2 decimal places, and shall NOT have any unit.In the determination of molar mass of an unknown substance by ebullioscopic constant, 30 mL of acetone (C3H6O) was placed in a test tube with thermometer and glass tubing and subjected to water bath. Upon boiling, the temperature reads 56 deg C. For the boiling point of unknown-acetone solution, you prepared the solution by mixing 1.60 g of unknown solute in the 30 mL acetone and subjected it again to water bath. The boiling temperature of the solution is 56.68 deg C. The density of acetone = 0.9849 g/mL and the Kb of acetone = 1.67 degC/kg - What is the value for delta Tb or the change in boiling temperature (in degrees Celsius)? Final answer must be rounded off to 2 decimal places, and shall NOT have any unit. - How many moles of solute is present in the solution given in the video? Final answer must be rounded off to 2 decimal places, and shall NOT have any units. - What is the molal concentration of the solution? Final answer must be rounded off to 2 decimal places, and shall NOT…
- An experimental attempt was to convert 2000 cm^3 of a solution consists of 0.96 ethanol into another solution consisting of 0.56 ethanol. If the density of water at the experiment's temperature and pressure is 997 kg/ m^3, How much H2O should be added to the 2000 cm^3 solution? What is the solution's volume after conversion? Given data: In the 0.96 ethanol solution :V ethanol = 0.816 cm^3/g and Vwater = 1.273 cm/g In the 0.58 ethanol solution: V ethanol = 0.953 cm^3/g and :V water = 1.243 cm/gGiven the following data for Mass of test tube and stearic acid = 14.17 gMass of test tube = 11.40 gFreezing point of strearic acid = 69.59o CMass of weighing paper + naphthalene =1.230 gMass of weighing paper = 0.920 gFreezing point solution = 64.00o CKf = 4.5o C/m Determine the following1. mass of stearic acid in g (2 decimal places); _____2. mass of naphthalene in g (2 decimal places); _____3. freezing point depression (2 decimal places); _____4. molality of solution (3 significant figures); _____5. moles of naphthalene (3 significant figures); _____6. molar mass of naphthalene, experimentally (3 significant figures); _____7. % error if theoretical molar mass of naphthalene is 128.17 g/ mole, USE ABSOLUTE VALUE (3 significant figure); _____You have a container of 17.42 M conc. acetic acid (CH3COOH(aq)) with density 1.05 g/mL, find the molality of a 150ml sample of the solution. molality = moles solute/Kg solvent moles solute = (0.150 L)(17.42 mol/L) = 2.613 mol CH3COOH molar mass of CH3COOH = 60.06 g/mol grams of solution = (150mL)(1.05 g/mL) = 157.5 g solution grams of solute = (2.613 mol)(60.06 g/mol) = 156.9 g CH3COOH so the total grams of water in this solution would be: g H2O = g solution - g CH3COOH = 157.5g - 156.9 g = 0.6g H2O 0.6g H2O = 0.0006 Kg H2O so molality = (2.613 mol CH3COOH) /(0.0006 kg H2O) = 4355 molal this seems rather high to me but if its 'concentrated' acetic acid, I know that you can get it to about 98% as glacial acetic acid so perhaps there is 4355 moles of acetic acid per kg of water. Did I do this correctly? I know that typically the solvent is the larger component of a solution but the fact that it gives me CH3COOH(aq) would imply that H2O is the solvent still in this situation, I have…
- Stock hydrofluoric acid solution is 49.0% HF and has a specific gravity of 1.17. What is the molarity of the solution?You are wanting to apply a soil conditioner to your garden. 10mL of conditioner cover 3m^(2) of your garden. However, before you use it you must dilute the concentrate by placing 10ml of Seasol in a 9L bucket. How many litres of the diluted mixture do you need to cover a garden of 80m^(2).You weigh out 80g of NaOH pellets and dilute to 1 liter. MW = 40 g/mol. What is the normality of the solution?



