For each of the following pairs of compounds, choose the bond that would absorb at the higher wavenumber in IR spectroscopy. Pair one Pair two (H3C);C¬H (H3C)3Si-H A' B C A and C B and D A and D Band C
For each of the following pairs of compounds, choose the bond that would absorb at the higher wavenumber in IR spectroscopy. Pair one Pair two (H3C);C¬H (H3C)3Si-H A' B C A and C B and D A and D Band C
Chapter12: Structure Determination: Mass Spectrometry And Infrared Spectroscopy
Section12.SE: Something Extra
Problem 20AP: The hormone cortisone contains C, H, and O, and shows a molecular ion at M+=360.1937 by...
Related questions
Question
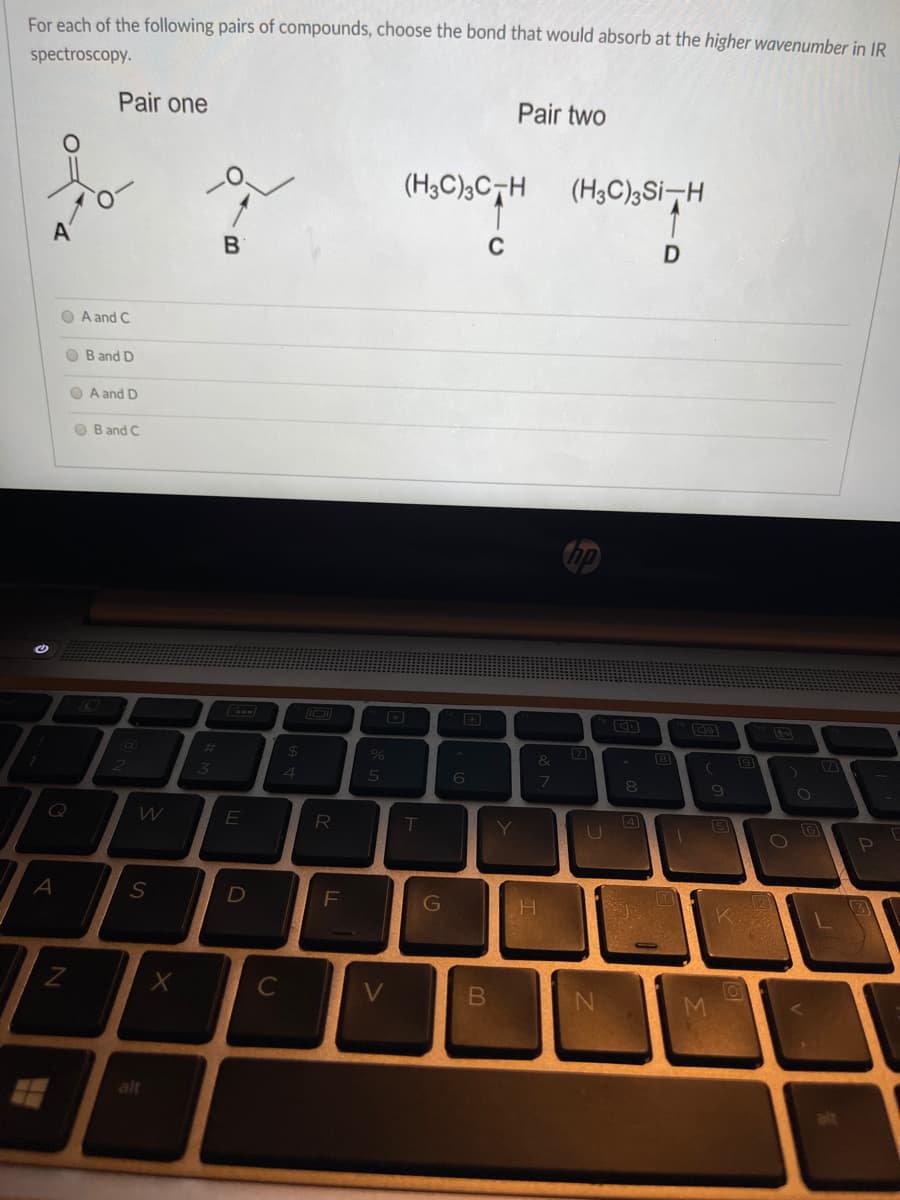
Transcribed Image Text:For each of the following pairs of compounds, choose the bond that would absorb at the higher wavenumber in IR
spectroscopy.
Pair one
Pair two
(H3C);C¬H
(H3C)3Si-H
A'
O A and C
O B and D
O A and D
O B and C
24
2
8
E
R
T
5
U
A
G
C
V
alt
alt
B
Expert Solution
Step 1
IR wavenumbers are decided by the bond strength it is directly proportional to bond strength. Stronger the bond higher the wavenumber.
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning