For each of the following reactions, draw the skeletal structure(s) of the most stable conformer for the major expected product(s) for the reaction. KMNO4 a) H2O/MEOH, 0°C ОН Br Охопе Br HO. b) Acetone/H2O, 0° ОН OH KMNO4 c) Н2О/MеОН, 0°С OH
For each of the following reactions, draw the skeletal structure(s) of the most stable conformer for the major expected product(s) for the reaction. KMNO4 a) H2O/MEOH, 0°C ОН Br Охопе Br HO. b) Acetone/H2O, 0° ОН OH KMNO4 c) Н2О/MеОН, 0°С OH
Chapter11: Reactions Of Alkyl Halides: Nucleophilic Substitutions And Eliminations
Section11.SE: Something Extra
Problem 75AP: In light of your answer to Problem 11-74, explain why one of the following isomers undergoes E2...
Related questions
Question
plz avoid using other ONLINE SOUCES
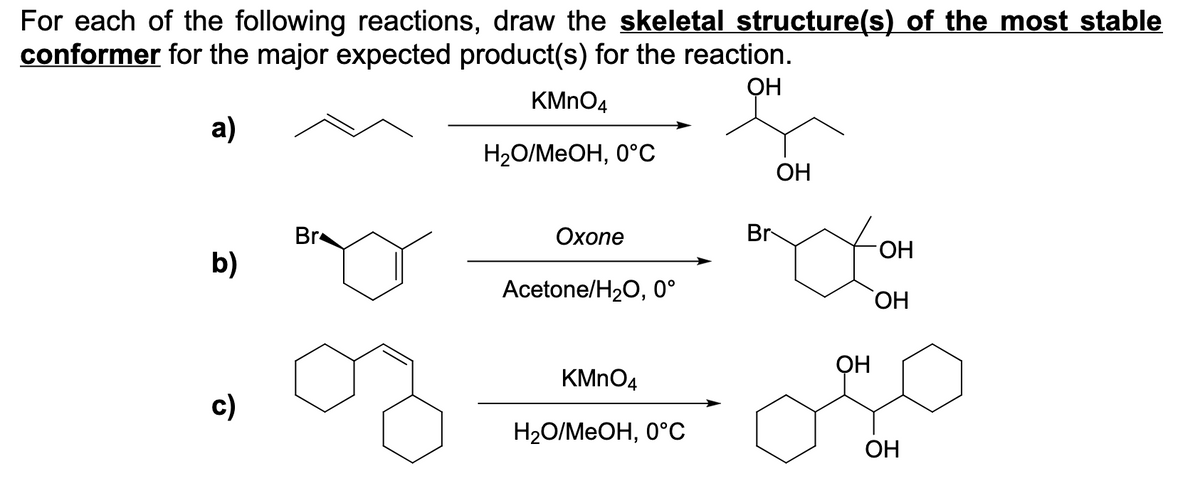
Transcribed Image Text:For each of the following reactions, draw the skeletal structure(s) of the most stable
conformer for the major expected product(s) for the reaction.
ОН
KMNO4
a)
Н2О/МеОН, 0°С
OH
Br
Охопе
Br
b)
Acetone/H20, 0°
ОН
OH
KMNO4
c)
Н2О/МeОН, 0°С
OH
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

