larpip Place the conformations of cis-1-isopropyl-4-methyleyclohexane that are drawn below in order of increasing stability deast stable to most stable). B E
larpip Place the conformations of cis-1-isopropyl-4-methyleyclohexane that are drawn below in order of increasing stability deast stable to most stable). B E
Chapter4: Organic Compounds: Cycloalkanes And Their Stereochemistry
Section4.4: Conformations Of Cycloalkanes
Problem 11P: Two conformations of cis-l, 3-dimethylcyclobutane are shown. What is the difference between them,...
Related questions
Question
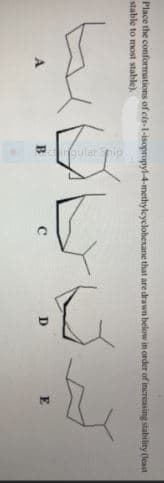
Transcribed Image Text:larpip
Place the conformations of cis-1-isopropyl-4-methyleyclohexane that are drawn below in order of increasing stability deast
stable to most stable).
B
E
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning