Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
ChapterA1: Evaluation Of Analytical Data
Section: Chapter Questions
Problem A1.14QAP
Related questions
Question
Transcribed Image Text:Ma 9 uta
Bb Co Bb 70 Bb Or
Ch
O Int O Dr O Hc G pe b Se E Lal 4 Pa O Da B Cla G BH
O
Di +
WP
A education.wiley.com/was/ui/v2/assessment-player/index.html?launchld=28461bef-2a57-4092-8156-ef4e43a30d2a#/question/8
E Chapter 1 (modified)
Question 9 of 26
< >
0 / 1
Show Attempt History
Current Attempt in Progress
Incorrect.
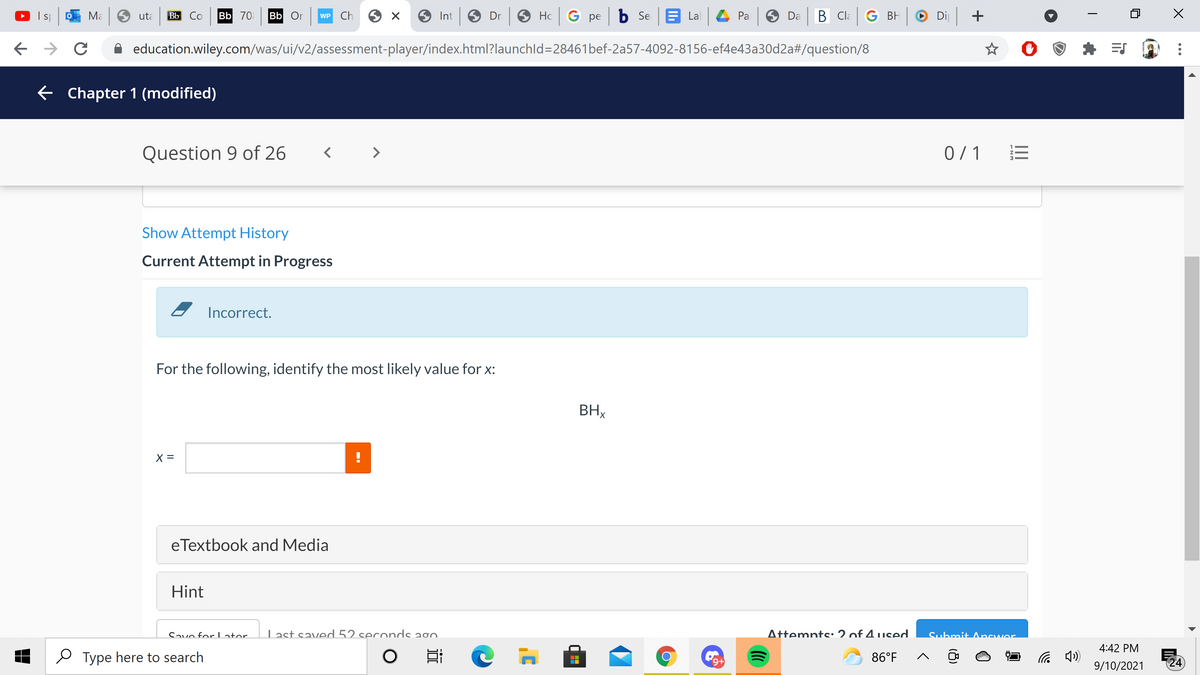
For the following, identify the most likely value for x:
BHx
X =
eTextbook and Media
Hint
Savo forLator.
Last saved 52 seccnds a0.
Attemnts:2 of 4 used.
Submit Ancwer
4:42 PM
e Type here to search
86°F
24
+6,
9/10/2021
ENM
18)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning