For the following reaction in aqueous solution, identify all those species that will be spectator ions. Select all that apply. Na,SO4+1 + Hg(NO3)2 HgSO,+ 2NANO3 >
For the following reaction in aqueous solution, identify all those species that will be spectator ions. Select all that apply. Na,SO4+1 + Hg(NO3)2 HgSO,+ 2NANO3 >
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 147CP: The measurement of pH using a glass electrode obeys the Nernst equation. The typical response of a...
Related questions
Question
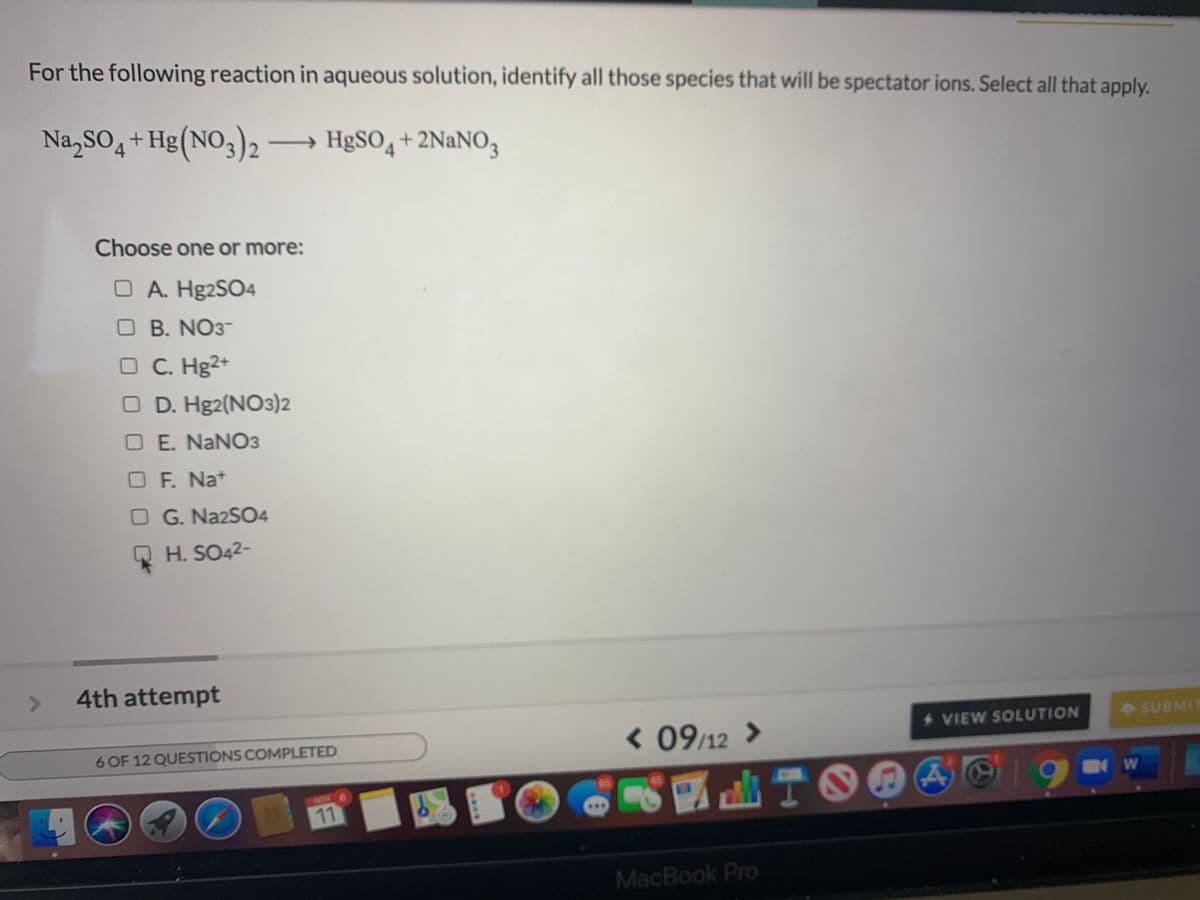
Transcribed Image Text:For the following reaction in aqueous solution, identify all those species that will be spectator ions. Select all that apply.
Na,SO4+Hg(NO3)2 → HgSO, + 2NaNO3
Choose one or more:
O A. H92SO4
O B. NO3-
C. Hg2+
O D. Hg2(NO3)2
O E. NANO3
O F. Na+
O G. NA2SO4
H. SO42-
4th attempt
SUBMIT
+ VIEW SOLUTION
< 09/12 >
6 OF 12 QUESTIONS COMPLETED
TOO4
19
415
NOV 6
11
MacBook Pro
....
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning