For the reaction: 6 COx + 6 H;O CH12O6«9 + 6O) (photosynthesis)| a.)Calculate the ASmiy, and classify whether or not the reaction is spontaneous at 25ºC s° (J/K.mol) AH°; (kJ/mol) -393.50 AG° (kJ/mol) CO20 H;O 213.60 -393.40 69.90 -285.80 -237.20 205.00 212.10 -1274.50 -910.56
For the reaction: 6 COx + 6 H;O CH12O6«9 + 6O) (photosynthesis)| a.)Calculate the ASmiy, and classify whether or not the reaction is spontaneous at 25ºC s° (J/K.mol) AH°; (kJ/mol) -393.50 AG° (kJ/mol) CO20 H;O 213.60 -393.40 69.90 -285.80 -237.20 205.00 212.10 -1274.50 -910.56
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter16: Spontaneity Of Reaction
Section: Chapter Questions
Problem 6QAP: On the basis of your experience, predict which of the following reactions are spontaneous. (a)...
Related questions
Question
P.S. Answer carefully and show your solution.
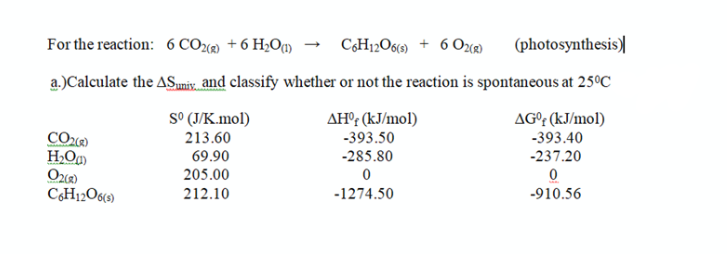
Transcribed Image Text:For the reaction: 6 CO208) + 6 H2Oa)
C6H12O6(s) + 6 Ox®)
(photosynthesis)
a.)Calculate the ASumis, and classify whether or not the reaction is spontaneous at 25°C
s° (J/K.mol)
AH°; (kJ/mol)
AG° (kJ/mol)
213.60
-393.50
-393.40
69.90
-285.80
-237.20
Oz)
CH12O6«)
205.00
212.10
-1274.50
-910.56
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning