Formation of soluble silver-ammonium complex, in an AgCl solution, occurs on a 3-step process: Based on the chemical equation provided, what do you think is equivalent to K2? a. [Ag(NH3)2+] / {[Ag(NH3)+][NH3]} b. [Ag(NH3)2+] / {[Ag+][NH3+]2} c. [Ag+] [Cl-] d.[AgNH3] / {[Ag+][NH3]}
Formation of soluble silver-ammonium complex, in an AgCl solution, occurs on a 3-step process: Based on the chemical equation provided, what do you think is equivalent to K2? a. [Ag(NH3)2+] / {[Ag(NH3)+][NH3]} b. [Ag(NH3)2+] / {[Ag+][NH3+]2} c. [Ag+] [Cl-] d.[AgNH3] / {[Ag+][NH3]}
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.10QAP
Related questions
Question
Formation of soluble silver-ammonium complex, in an AgCl solution, occurs on a 3-step process:
Based on the chemical equation provided, what do you think is equivalent to K2?
a. [Ag(NH3)2+] / {[Ag(NH3)+][NH3]}
b. [Ag(NH3)2+] / {[Ag+][NH3+]2}
c. [Ag+] [Cl-]
d.[AgNH3] / {[Ag+][NH3]}
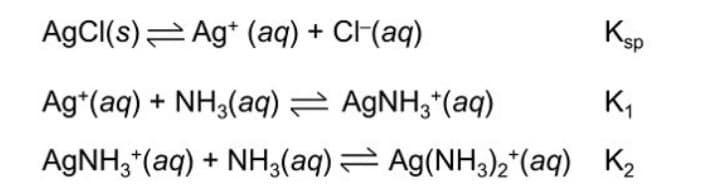
Transcribed Image Text:AgCI(s)= Ag* (aq) + CF(aq)
Ksp
Ag*(aq) + NH3(aq) = AGNH3*(aq)
K,
AGNH,*(aq) + NH3(aq)= Ag(NH3)2*(aq) K2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole



Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole