From the attached procedures, make a table consisting of all the compounds and their respective molecular weight (g/mol), density (g/ml), volume (ml), weight (g), mole and ratio. Take caution to ensure that the molecular weight is the CORRECT literature value.
From the attached procedures, make a table consisting of all the compounds and their respective molecular weight (g/mol), density (g/ml), volume (ml), weight (g), mole and ratio. Take caution to ensure that the molecular weight is the CORRECT literature value.
Chapter31: Synthetic Polymers
Section31.SE: Something Extra
Problem 28AP
Related questions
Question
100%
From the attached procedures, make a table consisting of all the compounds and their respective molecular weight (g/mol), density (g/ml), volume (ml), weight (g), mole and ratio. Take caution to ensure that the molecular weight is the CORRECT literature value.
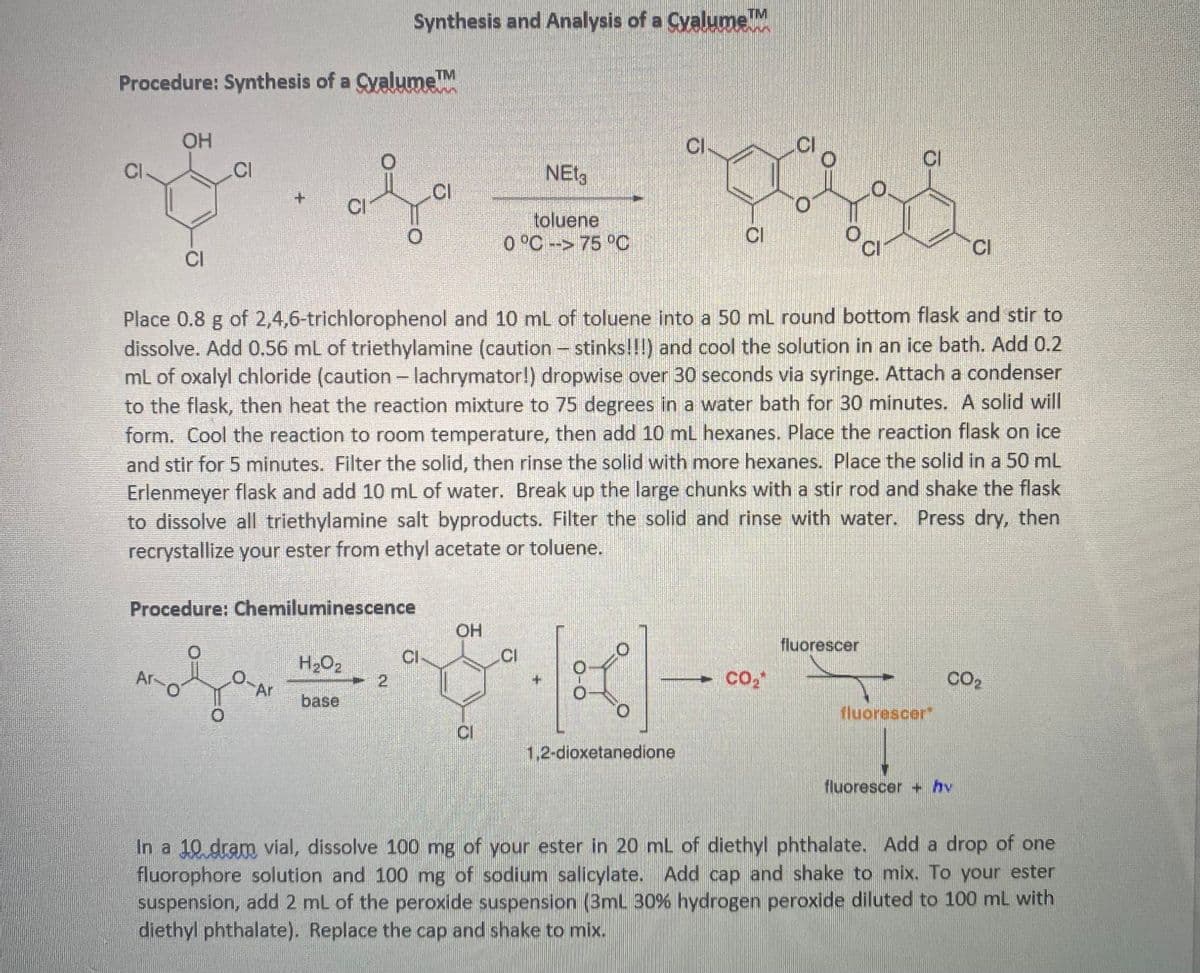
Transcribed Image Text:Procedure: Synthesis of a Cyalume™M
CI
OH
CI
Ar-
CI
O
Procedure: Chemiluminescence
0.
CI
Ar
TM
Synthesis and Analysis of a Cyalume ™M
H₂O₂
base
2
CI
OH
Place 0.8 g of 2,4,6-trichlorophenol and 10 mL of toluene into a 50 mL round bottom flask and stir to
dissolve. Add 0.56 mL of triethylamine (caution - stinks!!!) and cool the solution in an ice bath. Add 0.2
mL of oxalyl chloride (caution - lachrymator!) dropwise over 30 seconds via syringe. Attach a condenser
to the flask, then heat the reaction mixture to 75 degrees in a water bath for 30 minutes. A solid will
form. Cool the reaction to room temperature, then add 10 mL hexanes. Place the reaction flask on ice
and stir for 5 minutes. Filter the solid, then rinse the solid with more hexanes. Place the solid in a 50 mL
Erlenmeyer flask and add 10 mL of water. Break up the large chunks with a stir rod and shake the flask
to dissolve all triethylamine salt byproducts. Filter the solid and rinse with water. Press dry, then
recrystallize your ester from ethyl acetate or toluene.
CI
NET3
toluene
0 °C -> 75 °C
CI
O-O
0
CI
1,2-dioxetanedione
CI
2
CI
CO₂*
0
0
oc
fluorescer
CI
fluorescer*
CI
CO₂
fluorescer + hv
In a 10 dram vial, dissolve 100 mg of your ester in 20 mL of diethyl phthalate. Add a drop of one
fluorophore solution and 100 mg of sodium salicylate. Add cap and shake to mix. To your ester
suspension, add 2 mL of the peroxide suspension (3mL 30% hydrogen peroxide diluted to 100 mL with
diethyl phthalate). Replace the cap and shake to mix.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

