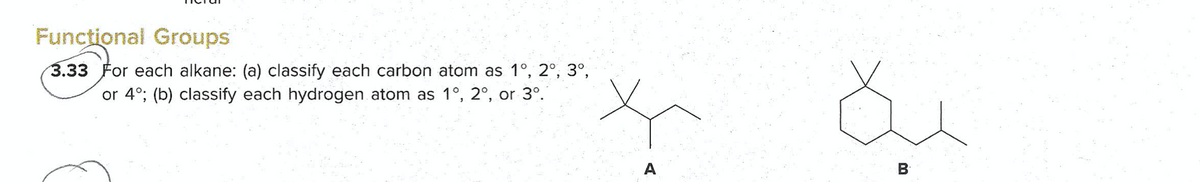
Functional Groups 3.33 For each alkane: (a) classify each carbon atom as 1°, 2°, 3°, or 4°; (b) classify each hydrogen atom as 1°, 2°, or 3°. A B
Q: Data Table, Introduction 9.) Classify the following compound as an alkane, alkene, or alkyne and…
A: Organic compounds which contain single, double and triple bonds between consecutive carbon atoms are…
Q: Use the IUPAC system to name the structural diagram shown above. О Н нн нн Н-С-С- Н- C - C - 0 Н Н н…
A:
Q: By hand, draw all of the isomers with the molecular formula C4H9F using skeletal structures. Name…
A: The molecular formula of the compound given is C4H9F.
Q: Draw the five constitutional isomers that have molecular formula C5H10 and contain one ring ?
A: The given molecular formula of compound is C5H10. It obeys the general formula for cycloalkanes,…
Q: give the full iupac name include isomerism
A: The given compounds are: The IUPAC names of the above-provided compounds have to be determined.
Q: Draw the seven constitutional isomers having molecular formula C4H1,0. Identify the functional group…
A: Functional groups present in the constitutional isomers are alcohol and ether. Constitutional…
Q: Provide the IUPAC name for the molecule below. Do not put spaces between commas or dashes.
A:
Q: Draw the structure of compound C4H9COCH3C4H9COCH3 to show all individual atoms and all covalent…
A:
Q: Name each alkane using the ball-and-stick model, and classify each carbon as 1°, 2°, 3°, or 4°.
A:
Q: CI. Molecular formula
A:
Q: ODD-ONE OUT: Considering possible isomerism, choose which one is not part of the group.
A: Square planner complexes: Four atoms are positioned at the corners of a square on the same plane…
Q: A. Expand the condensed structural formula written on the first column of the table to reveal the…
A: IUPAC nomenclature from structural formula Identify the longest carbon chain Identify the…
Q: r(s) of organic compounds is/are constitutional isomers? ... A D D and B, C B and C, D and D only…
A:
Q: (b) Constitutional isomers with different positions of functional groups
A: # First we have to know about constitutional isomerism : Consitutional isomerism : Those compounds…
Q: Give complete iupac name 2&3
A:
Q: How are the ball-and-stick models A and B related to each other? Do they represent constitutional…
A: Both models A and B represents an identical molecule. IUPAC name of molecule is hexane. In both…
Q: The structures Pentan-2-one and Pentan-3-one are isomers. A Positional B Stereoisomers Functional…
A: Answer :
Q: DIRECTIONS:Provide the required number of isomers ner molecular formula by illustrating them in…
A:
Q: Write condensed and skeletal structures for all the tertiary alcohols with molecular formula C6H14O…
A: A condensed structural formula is a system of writing organic structures in a line of text. It shows…
Q: 5.) Classify the following compound as an alkane, alkene, or alkyne and give the correct IUPAC…
A: Organic compounds which contain single, double and triple bonds between consecutive carbon atoms are…
Q: Describe how would you distinguish the following pairs, (a) Benzene and cyclohexane
A: (a) Answer - Cyclohexane - Cyclohexane is an alicyclic hydrocarbon comprising a ring of six carbon…
Q: Give any one difference between ethylene organic compound and benzene with respect to conjugation…
A: Interpretation: The difference between ethylene organic compound and benzene with respect to…
Q: What evidence can you cite to support the fact that although isomers have thesame molecular formula…
A: Isomers:- These are those compounds which have the same molecular formula but different structure
Q: Explain the structure of cyclic compounds (propane, butane, pentane and hexane) of the ring,…
A: Concept is based on Baeyers strain theory
Q: B. C. D. and ng are constitutional isomers? and and Br and CH₂=CHCH, QUESTION 18 hich amine has the…
A:
Q: Identify It tells about the total number of ring systems and pi-bonds in an organic compound.
A: It tells about the total number of ring systems and pi-bonds in an organic compound.
Q: 1. For functional group and hydrocarbon each: -Give definition: -Give classification: -Give…
A: Functional groups ad hydrocarbons are part of organic chemistry.
Q: 1. Give the IUPAC names of the following molecules. a) b) CH,CH,CHCH, CH, CH,
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: a Table, Introduction Classify the following compound as an alkane, alkene, or alkyne and give the…
A: Organic compounds which contain single, double and triple bonds between consecutive carbon atoms are…
Q: The structural formula of benzene is often represented as a ________. A. ring of six carbon…
A: The structural formula of benzene is often represented as a ring of six carbon atoms with a circle…
Q: Draw the seven constitutional isomers having molecular formula C4H10O. Identify the functional group…
A: Constitutional isomers A constitutional or structural isomer of a compound is another compound…
Q: What functional groupcharacterizes an alcohol? (c) Write a structural formula for1-pentanol, the…
A: There are so many organic compounds having variety of its application in daily life of human with…
Q: Answer the following question by referring to the ball-and-stick modelof fentanyl, a potent narcotic…
A:
Q: Quinapril (trade name Accupril) is a drug used to treat hypertension andcongestive heart failure.…
A:
Q: MCQ 13: Hydrocarbons contain A. carbon only B. hydrogen only C. oxygen D. carbon and hydrogen
A:
Q: For each alkane: (a) classify each carbon atom as 1°, 2°, 3°, or 4°; (b) classify each hydrogen atom…
A: (a)The given alkane is 2,2,3-trimethylpentane
Q: Which of the following does NOT belong to the group? a. alkenes b. alkanes c. alkynes d.…
A: The organic compounds that contain mainly carbon and hydrogen atoms are called hydrocarbons.…
Q: iii. CH3CH2COCH3 CH3CH2CH2OH iv. CH3CH2CH2CH2CH2COOCH3 CH3CH2CH2CH2CH2CH2COOH
A: A) CH3CH2COCH3 This is a ketone, as CO is a carbonyl functional group CH3CH2CH2OH This is an…
Q: shouln't there only be two isomers of trimethylcyclopropane? can you explain how it became six?…
A: An isomer is defined as the compounds having the same molecular formula but different structural…
Q: 2. There is a 63° difference in boiling points between heptane and pentane. What is this method to…
A: Here is the answer
Q: (b) Give the IUPAC name for the following compound. CI
A:
Q: b. Identify the IUPAC (International Union of Applied and Pure Chemistry) name of the following…
A: The IUPAC name for the given compound is propan-2-ol.
Q: what are the fncional groups presenm MAspirn which has the folbwing structural formula -CH3
A: Aspirine molecule is an aromatic compound containing benzene ring with two substituents on two…
Q: Draw at least THREE structural isomers for structure A AND structure F
A:
Q: Give 1 example each for Alkynes. Structure should contain 5 carbons with branching and substituents…
A: An organic compound that contains a carbon-carbon triple bond is known as alkyne. The general…
Q: The instructions are to 1. list the functional group that differentiates benzhydrol from biphenyl…
A: Functional group is an atom or a group of atoms that defines the chemical reactivity of a compound.
Q: Classify each of the following compounds as an alkane, alkene, alkyne, alcohol, aldehyde, amine, and…
A:
Q: Need asap provide the iupac name of each compound
A:
Q: Q2/Draw all isomer for structural formula and write the IUPAC name ? 1) CH. 2) C.H,O
A: 1) The isomers of C6H10 along with their IUPAC names are given below :
Q: Draw structures that t each description and name the functional group in each molecule: (a) two…
A: Isomers have same molecular formula but differ in arrangement of atoms within a…
Step by step
Solved in 3 steps with 3 images

- Bottled straight-chain alkanes can be used as fuels, including pentane and hexane . pentane hexane ΔΗ°comb (kJ/mol) -3535.0 -4163.0 Liquid Denisty (g/mL) 0.684 0.655needhelp finding two constitutional isomers that share the molecular formula C4H102 Q: Consider the following compound: CH3-CH2-CO-CH3 a. suggest a method for increasing the number of carbons by one: b. suggest a method for decreasing the number of carbons by one:
- Is it possible for a gasoline to have an octane numberexceeding 100? Explain.What functional groupcharacterizes an alcohol? (c) Write a structural formula for1-pentanol, the alcohol derived from pentane by making asubstitution on one of the carbon atoms.How many distinct dimethylcyclopropanes are there?
- Can you show the isomers and alchols present with a molecular formula?name and draw structural formulas for all cyclo-alkanes with the molecular formula C5H10. Be certain to include cis-trans isomers, as well as constitutional isomersC5H8Cl2 can you name and draw all isomers , make sure each has a 5 membered cyclic ring . And use E,Z when approp
- For the compound shown below, identify functional groups (A) – (L).Call out all functional groups and their approximate pKas (0-14) Does Curcumin belong to Phenol group?whwn naming a compound do ignore the di,tertri and the iso and juat look at the alphebets of the names, like dimethyl and isopropyl;

