Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter29: Supercritical Fluid Chromatography And Extraction
Section: Chapter Questions
Problem 29.11QAP
Related questions
Question
How do you solve chemical equations
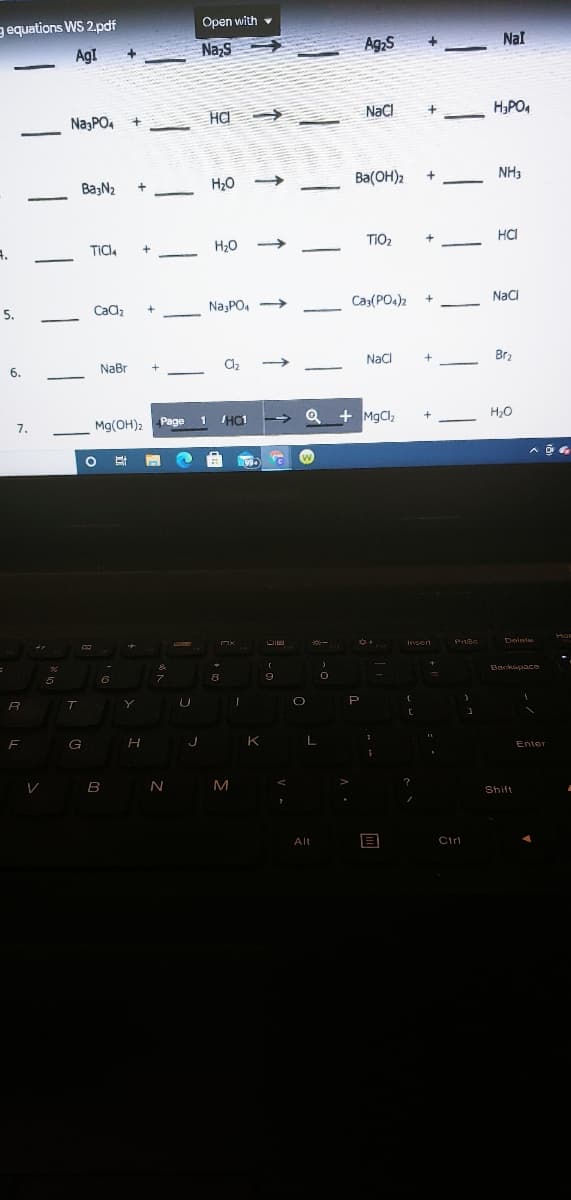
Transcribed Image Text:gequations WS 2.pdf
Open with v
AgI
Na,s
Ag;S
Nal
NazPO, +
HA
NaCI
H,PO4
BaşN2
H20
Ba(OH)2
NH3
-
-
ㅋ.
H20 >
TIO2
на
TICI,
5.
Cadz
Na,PO, >
Ca3(PO4)2
NaCl
NaBr
Naci
Brz
6.
Q + MgClz
H20
1
7.
Mg(OH)2 Page
DID
Hor
Insen
Delete
8
Backapace
R
T.
F
G
H J
K
Enter
V
N M
B
Shift
Alt
Ctri
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning