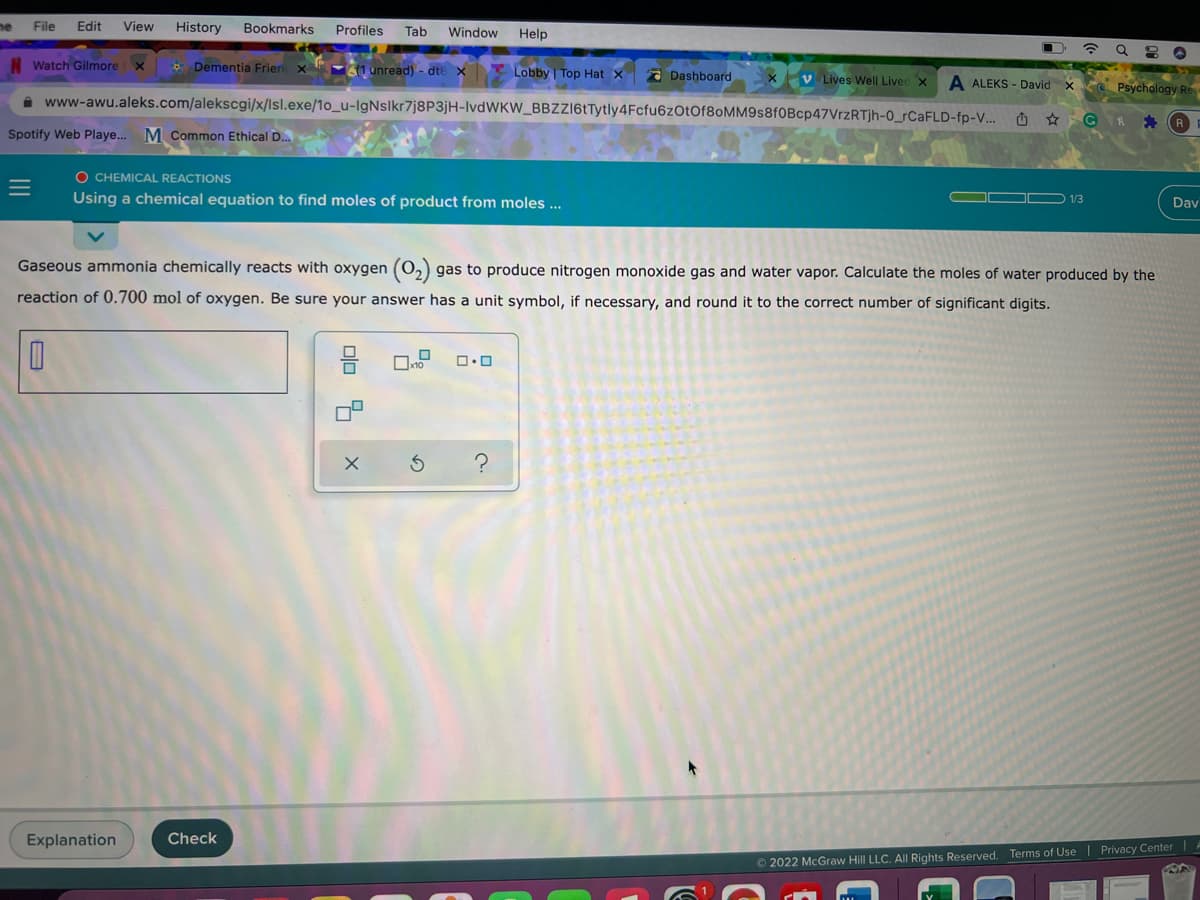
Gaseous ammonia chemically reacts with oxygen (0,) gas to produce nitrogen monoxide gas and water vapor. Calculate the moles of water produced by the reaction of 0.700 mol of oxygen. Be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits.
Gaseous ammonia chemically reacts with oxygen (0,) gas to produce nitrogen monoxide gas and water vapor. Calculate the moles of water produced by the reaction of 0.700 mol of oxygen. Be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits.
Chapter82: Physical Constants Of Liquids: The Boiling Point And Density
Section: Chapter Questions
Problem 5P
Related questions
Question
Transcribed Image Text:me
File
Edit
View
History
Bookmarks
Profiles
Tab
Window
Help
Watch Gilmorex
* Dementia Frien x
1 unread) - dte X
Lobby Top Hat X
2 Dashboard
xV Lives Well Livec x
A ALEKS - David
x Psychology Re
i www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IVDWKW_BBZZ6tTytly4Fcfu6zOtOf8oMM9s8f0Bcp47VrzRTjh-0_CAFLD-fp-V...
Spotify Web Playe..
M Common Ethical D..
O CHEMICAL REACTIONS
Using a chemical equation to find moles of product from moles ...
Dav
Gaseous ammonia chemically reacts with oxygen (0,) gas to produce nitrogen monoxide gas and water vapor. Calculate the moles of water produced by the
reaction of 0.700 mol of oxygen. Be sure your answer has a unit symbol, if necessary, and round it to the correct number of significant digits.
Explanation
Check
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT